(Intermediate) Reactant Xoda + Tip: Add curved arrows in this sketcher to represent the electron flows for the next mechanism step Apply Mechanism Hint Solution 1 remaining step(s) can be solved (Intermediate) Product
(Intermediate) Reactant Xoda + Tip: Add curved arrows in this sketcher to represent the electron flows for the next mechanism step Apply Mechanism Hint Solution 1 remaining step(s) can be solved (Intermediate) Product
Chapter8: Reaction Rates And Equilibrium
Section: Chapter Questions
Problem 8.20E
Related questions
Question
1
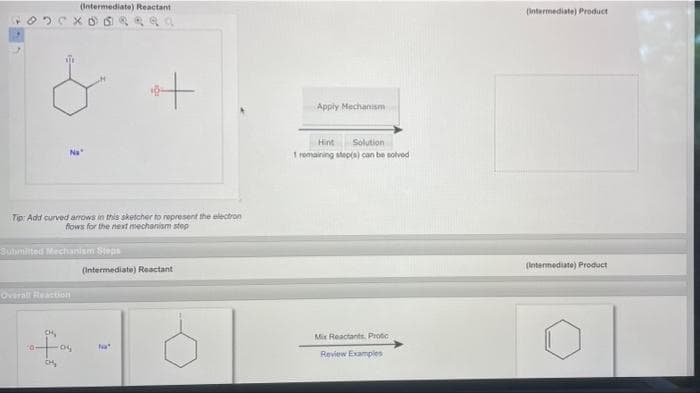
Transcribed Image Text:Tip: Add curved arrows in this sketcher to represent the electron
flows for the next mechanism step
Submitted Mechanism Steps
Overall Reaction
(Intermediate) Reactant
Xoda
CH₂
-04
+
(Intermediate) Reactant
Apply Mechanism
Hint Solution
1 remaining Mep(s) can be solved
Mix Reactants, Proto
Review Examples
(Intermediate) Product
(Intermediate) Product
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

