Q: Of particle size techniques, which technique can identify the 3-D dimensions of the particles? The…
A: A multiple choice question based on introduction to chemistry that is to be accomplished.
Q: A B C D
A: Relationship between these given organic molecules are like enantiomers , Diastereomers ,identical…
Q: Ethylene glycol freezes at -5 deg C in water. What is the boiling point of the solution?
A: Given→ Freezing point of ethylene glycol in water = -5 deg C. We have been asked to find…
Q: Half En. Eivy Ni +2e → Ni 0.23 Agt te → Ag o 0.80 Calculate the Equilibrium constant for this com…
A:
Q: 01 02 03 04 10--0 НО 2 2x OH HO OH OH O HO OH OH OH
A:
Q: Which of the following is the repeating unit in polyvinyl chloride (PVC)?
A: Polymer: Polymer is a Greek words. Poly means many mer means unit or part Polymer are large…
Q: A chemist knows that the equilibrium constant is 0.0049 for the following reaction: . CO₂ (g) + H₂…
A:
Q: How are the compounds in each pair related to each other? Are they identical, enantiomers,…
A: A question based on stereo-chemistry of organic compounds that is to be accomplished.
Q: Calculate the Carnot efficiency of a heat engine that has fluid temperature of 2,100 K down to 600…
A: The Carnot engine efficiency is calculated by the two temperatures. It will have a higher…
Q: Does adding sweetener and stabilizers to strawberry juice result to the decrease in temperature for…
A: When the sugar is added to the strawberry juice it lowers the freezing point because when sugar…
Q: ower! (a) (B) fo fa in a 488 m2 sample collected over water at 0.745 alm at 288.15K2 How many moles…
A: Given: Volume of sample = 488 mL Pressure = 0.745 atm Temperature = 288.15 K
Q: Consider the disulfide bond formation reaction via the oxidation of thiols along with the H- NMR…
A: In the given Spectrum the Thiol proton shows broad singlet at 1.5 ppm.
Q: When CA is 0.023 mol/L, the rate of a particular second-order reaction (in A) is 3.42 x 10-3…
A:
Q: emistry A 110.0-mL buffer solution is 0.105 M in NH3 and 0.135 M in NH4Br. The Kb value for NH3 is…
A: The answer is as follows:
Q: H3CO CI ARO NHCH3 MAT OCH 3 NH₂ TIC OCH 3 CI NO₂ HYD # CH3 ROC
A: When substitution at position-1 and position-3 in benzene ring called meta-substitution . When…
Q: 36) Identify the formal charge on the indicated atoms in the following A) SO4²; S and O B) CN- ; C…
A:
Q: A 2.89 g sample of limestone (CaCO3) was dissolved in 0.12M (HCI) solution according to the…
A: " Since you have posted a question with multiple sub-parts , we will solve first three sub-parts for…
Q: How long would it take a current of 1 ampere to reduce completely 80 cm3 of 0.10 M Fe2(SO4)3 to…
A: We can solve this problem by using the faraday law of electrolysis W= (E×i×t)/96500 W= mass of a…
Q: For the electrolysis of a molten mixture of NaCl and KBr, which of the following are the correct…
A: At anode oxidation takes place while at cathode reduction takes place.
Q: Determine the concentration of ferric ion that can exist in a solution at pH 3. The Ksp of ferric…
A: Given that : pH = 3 pH + pOH = 14 pOH = 14 -3 = 11
Q: III. Find the number of moles of the following: A.56.1 g of Zn(C₂H302)2 B.232 g of Na3PO4
A:
Q: 21) Consider the chemical reaction that occurs when 55 g of Al(OH)3(s) was added to 250 mL of 0.50M…
A:
Q: need the the step by step solution What is the volume in cubic meter of a 2 pound water?
A: Now here we need to do the unit conversion that is from pounds to cubic meter Now 1 pound =…
Q: Which way would the equilibrium shift if the temperature was decreased for this reaction (AH = -542…
A:
Q: N-p-hydroxyphenylethanamide is commonly known as a. acetaminophen b. acetamide c. acetanilide d.…
A: A question based on general chemistry that is to be accomplished.
Q: a. Based on the E°values, which metal is the most easily oxidized? b. Which metal on the list is…
A: Given - E0(Cu2+/Cu) = +0.34V E0(Fe2+/Fe) = -0.44V E0(Al3+/Al) = -1.66V…
Q: Arrange the steps of gravimetric analysis in chronological order
A: Gravimetric analysis is a quantitative method of the measurement of the substance on basis of their…
Q: Gas particles have random _____. speed speed and direction mass temperature direction
A: We can answer the above question by using Kinetic theory of gases.
Q: 4000 100 IR Spectrum (quid film) 88888 80 Få 60 40% 8 3000 CO 80 40 13C NMR Spectrum (50.0 MHz.…
A: 1H-NMR spectrum gives complete information about structure of the molecule. Form the position of the…
Q: The electrochemical cell notation for the non-standard cell (assumed to be voltaic cell) below is…
A: Here we have to predict whether the given cell notation is written correct or not.
Q: for the reaction NH3 C991+ H20 (1) - Nhu caalt of cour which of the following is a conjugate…
A: When base accept a proton it forms conjugate acid and when acid donates a proton it forms conjugate…
Q: The conductance of several salts in a binary mixture of water and a dipolar aprotic solvent ethylene…
A: Given data:
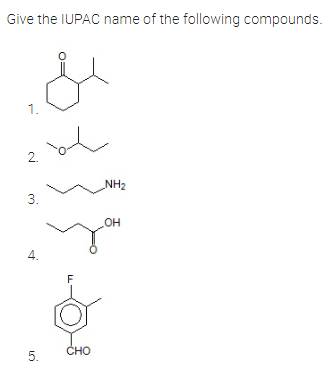
Q: Give the 9) (b) of the name Compounds given below LOH 애
A: Write IUPAC name of the given structure---
Q: (b) Suggest a reasonable biosynthesis for the naturally occurring alkaloid coniine (isolated from…
A:
Q: CI H-CH3 CH3 OLE CI H₂CC Hạc"C-CH, CH3 BIM J-G CI HIGH H LAR CI C-H HI.C CH3 CUL
A: The reactivity towards SN2 reaction decreases when the nucleophile is sterically inhibited . Steric…
Q: A solution was prepared in the lab from equal volumes of lithium fluoride and zinc chloride. If the…
A: The concentration of lithium fluoride = 4.00×10−4 M The concentration of zinc chloride= 1.00×10−4 M…
Q: OH Pool of choices: PI3 concentrated H₂SO4, heat, PRODUCT A PRODUCT B
A:
Q: Propose a mechanism to account for the following reaction: CH3 CH₂Cl H₂C AICI H₂C
A: We have to predict the mechanism for the given reaction.
Q: Consider the following alcohols: - produces symmetric ketone after oxidation? -resistant to…
A:
Q: Under certain conditions the rate of this reaction is zero order in ammonia with a rate constant of…
A:
Q: Q5. Give one condition for the denaturation step employed in blotting
A: As per the guideline, since you have asked multiple questions, we have solved the first question for…
Q: Select among the following series of univalent cations an ion with the highest coagulative power…
A: The coagulation power of cation increase with increasing valency of cation this is called Hardy…
Q: MON TUE WED THU FRI SAT SUN Brom-3-methylpenbare has three isomers 7 - Bromo-3-methylppent- ane…
A:
Q: sample of gas at 1045.9 mmHg and 21.64 degree celsius has the pressure increase to 159.59 kPa. What…
A: Pressure is directly proportional to temperature
Q: At what position, and on what ring, would you expect the following substance to undergo…
A: In this question, we will see that which ring and what position will give expected electrophilic…
Q: What is the percent of each halogen in a 0.4712g mixture of sodium chloride and barium chloride…
A:
Q: 7. If 10 kg/min of air are compressed isothermally from p₁ 96 kPa and V₁ = 7.65 m³/min to p₂ = 620…
A:
Q: What are the two types of membrane usually employed for western blotting?
A: Membranes used in western blotting are Nitrocellulose and PVDF that is Polyvinylidine Difluoride…
Q: 5) Synthesis: Show how you would carry out the following synthesis using inorganic or organic…
A:
Q: A piece of metal thought to be either chromium, nickel or lead was weighed and found to have a mass…
A: A piece of metal Chromium/Nickel/Lead has a mass of 47.4 grams. Volume of water in a graduated…
Step by step
Solved in 2 steps


