Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter23: Enzymes
Section: Chapter Questions
Problem 23.65P
Related questions
Question
just draw all this same in chemdraw asap plz
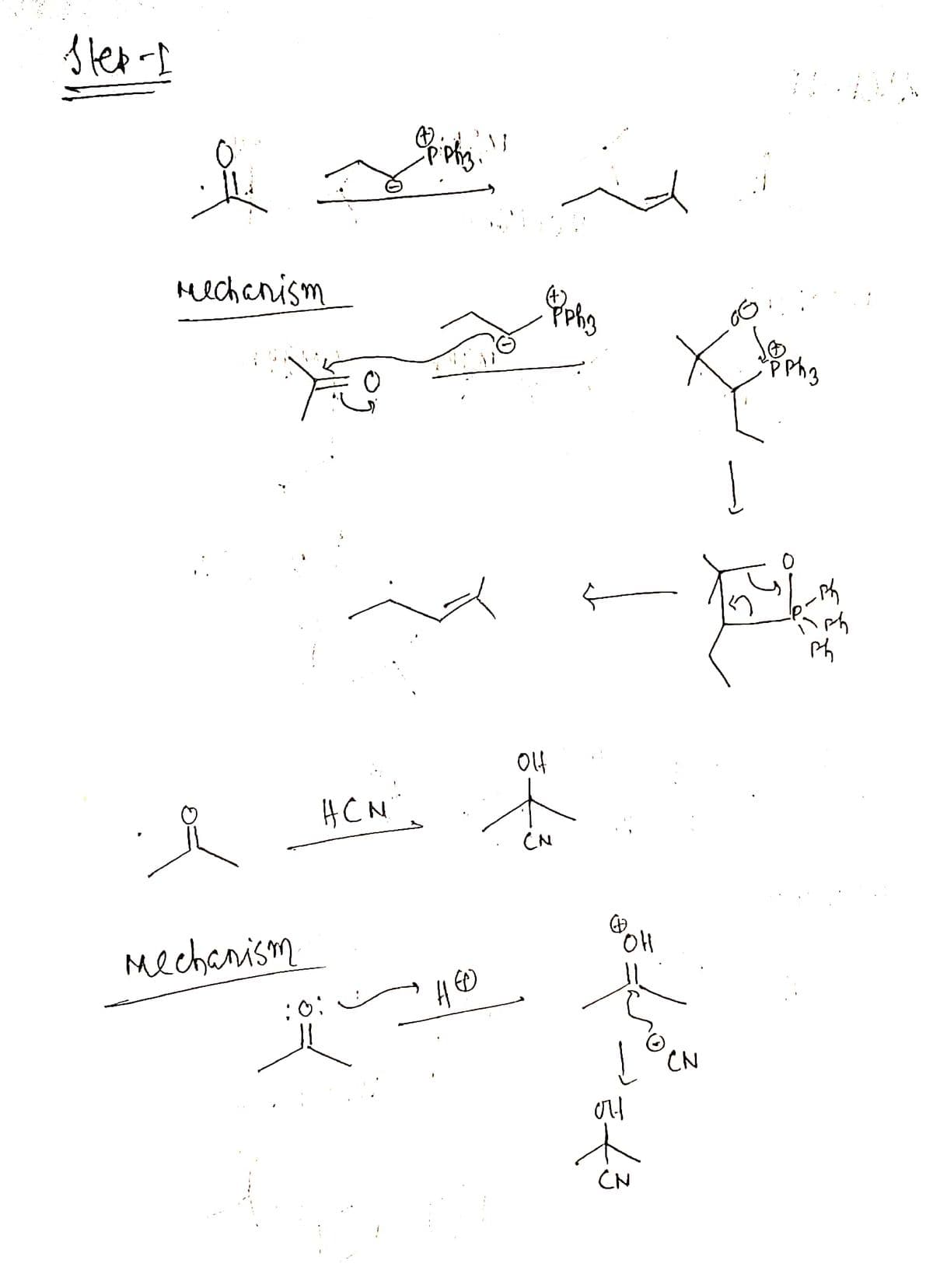
Transcribed Image Text:Slep - I
mechanism
Mechanism
HCN"
10:
ppg"
Oppho
the
CN
(P)
تھی کہ مجھ
에
X de
ㅅ
CN
PPh 3
파
먹
먹
먹
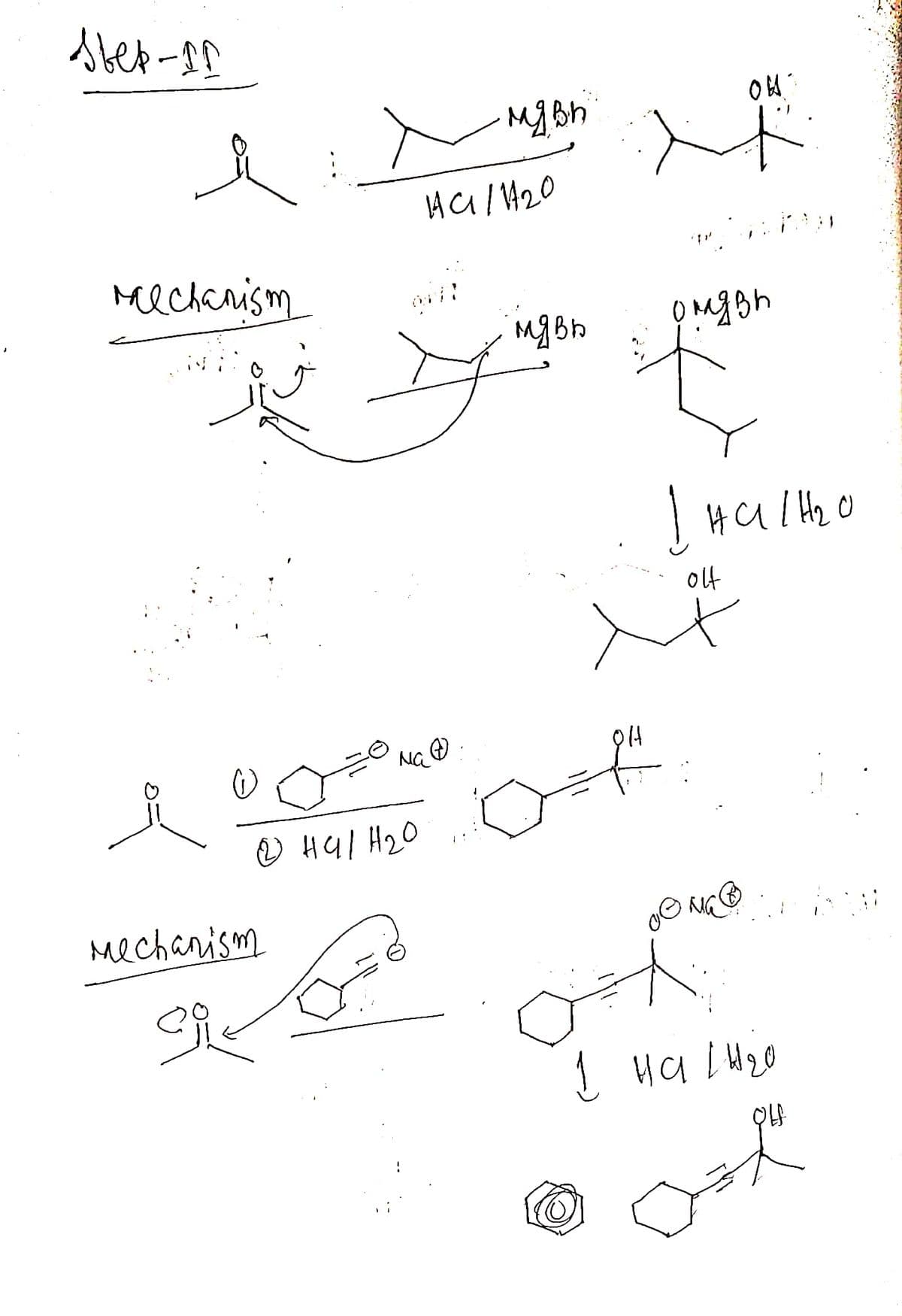
Transcribed Image Text:ввер-да
Mechanism
о на н20
Mechanism
сл
на/ 1420
мас
До
мя вы
мявь
он
Он
олдви
в
К
І нало
olt
ма
1 на снго
ook
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning