KMN04 converted to MnO with 5 electron * transfer T. FO Solutions of weak acids always have lower concentrations of H+ than solutions of * .strong acids T O FO The weight of a millimole of (NH4)2HPO4 is * .0.132 mg TO FO
KMN04 converted to MnO with 5 electron * transfer T. FO Solutions of weak acids always have lower concentrations of H+ than solutions of * .strong acids T O FO The weight of a millimole of (NH4)2HPO4 is * .0.132 mg TO FO
Chapter10: Effect Of Electrolytes On Chemical Equilibria
Section: Chapter Questions
Problem 10.11QAP
Related questions
Question
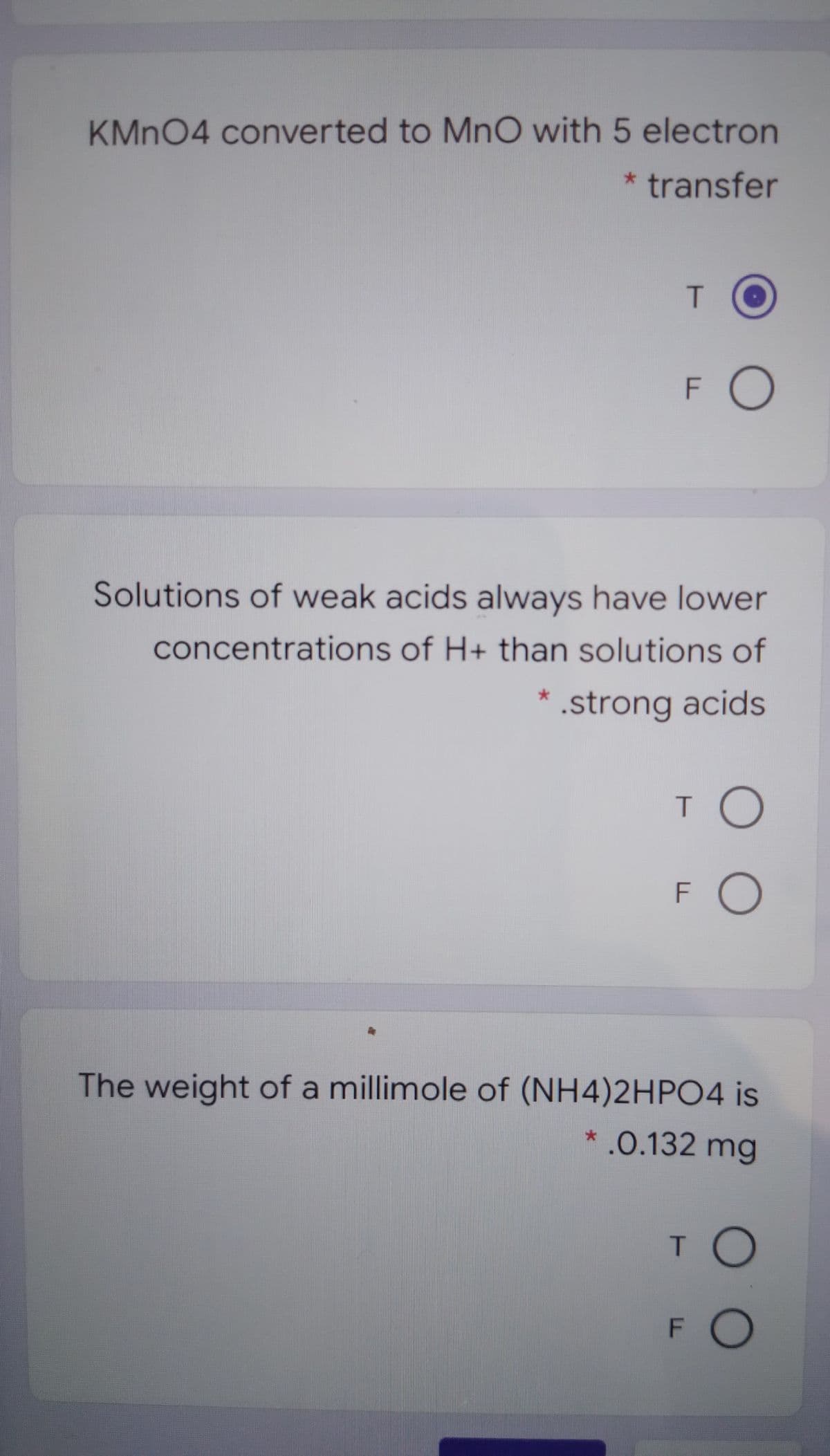
Transcribed Image Text:KMN04 converted to MnO with 5 electron
* transfer
F O
Solutions of weak acids always have lower
concentrations of H+ than solutions of
* .strong acids
T O
FO
The weight of a millimole of (NH4)2HP04 is
*.0.132 mg
FO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning