Light was measured to have a frequency of (8.09x10^3) Hz. What was the wavelength in mm? c = 3.00 x 108 m/s 1 Hz = 1 s 1 Be careful with your units!!! For full credit: • Enter your answer with 3 significant figures . Put it in scientific notation. • Include the correct units Note: Your answer is assumed to be reduced to the highest power possible. Your Answer: Answer x10 units
Light was measured to have a frequency of (8.09x10^3) Hz. What was the wavelength in mm? c = 3.00 x 108 m/s 1 Hz = 1 s 1 Be careful with your units!!! For full credit: • Enter your answer with 3 significant figures . Put it in scientific notation. • Include the correct units Note: Your answer is assumed to be reduced to the highest power possible. Your Answer: Answer x10 units
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter1: Introduction To Chemistry
Section: Chapter Questions
Problem 1.71QE
Related questions
Question
plz answer with clear handwriting correctly in white paper
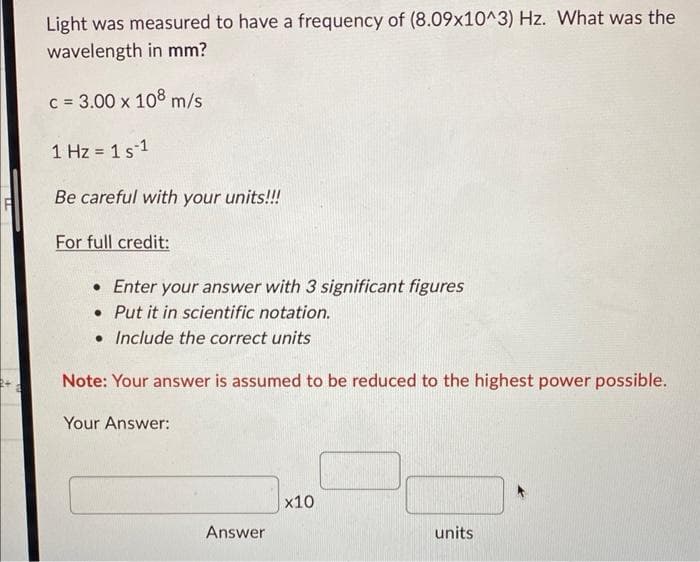
Transcribed Image Text:Light was measured to have a frequency of (8.09x10^3) Hz. What was the
wavelength in mm?
c = 3.00 x 108 m/s
1 Hz = 1 s 1
Be careful with your units!!!
For full credit:
• Enter your answer with 3 significant figures
. Put it in scientific notation.
• Include the correct units
Note: Your answer is assumed to be reduced to the highest power possible.
Your Answer:
Answer
x10
units.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
