List the following substances in order of increasing normal boiling points Tb, and explain your reasoning: CH3OH, OF2, Ar, MgO Boiling point 1=lowest substance Reason 4=highest CH3OH OF, Ar MgO Is an ionic compound: forces are stronger than forces between neutral molecules. Has hydrogen bonds: stronger than other dipole-dipole interactions.
List the following substances in order of increasing normal boiling points Tb, and explain your reasoning: CH3OH, OF2, Ar, MgO Boiling point 1=lowest substance Reason 4=highest CH3OH OF, Ar MgO Is an ionic compound: forces are stronger than forces between neutral molecules. Has hydrogen bonds: stronger than other dipole-dipole interactions.
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter15: Gases,liquids, And Solids
Section: Chapter Questions
Problem 25E
Related questions
Question
Transcribed Image Text:evo/index.html?deploymentld%3D55750828934189288909969212&elSBN=9781305657571&snapshotld...
INDTAP
Q Search this c
Use the References to access important values if needed for this question.
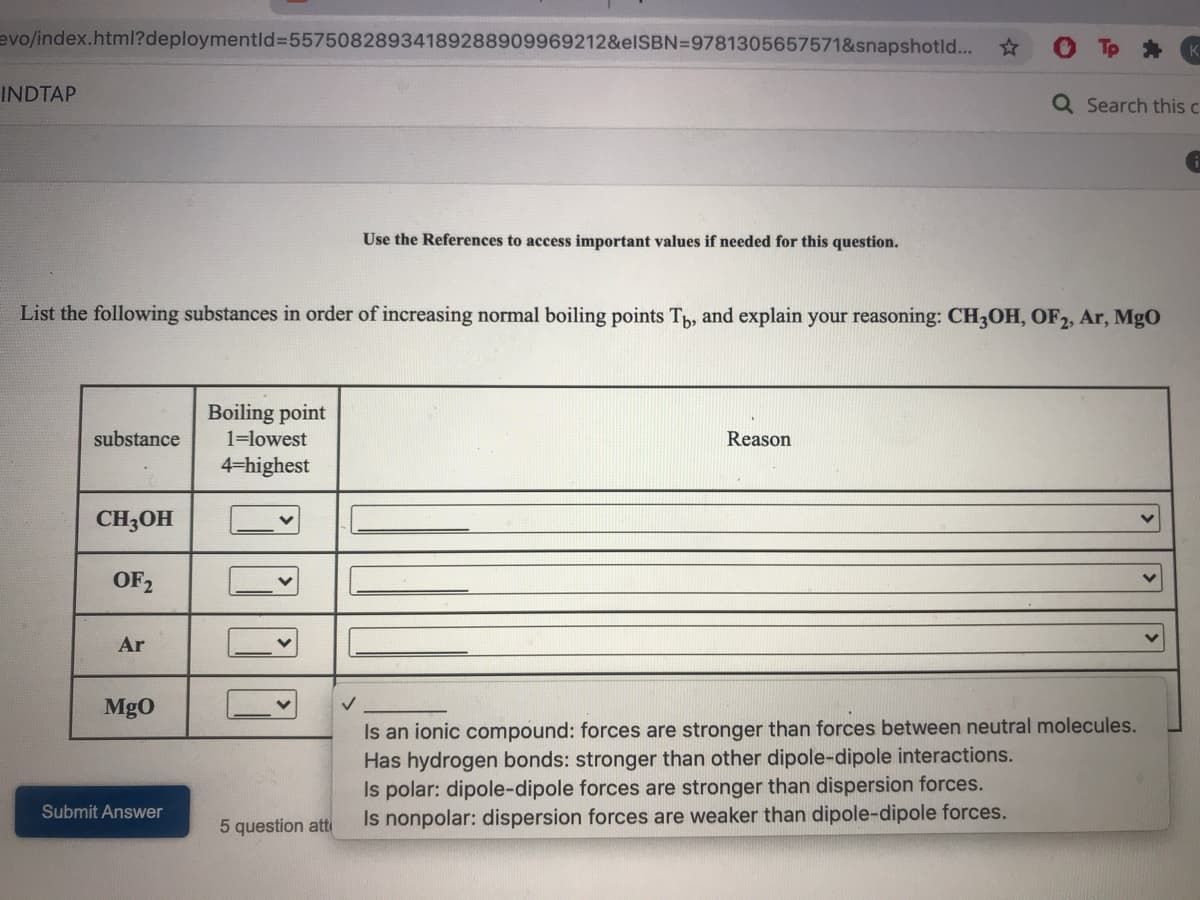
List the following substances in order of increasing normal boiling points T, and explain your reasoning: CH3OH, OF2, Ar, MgO
Boiling point
substance
1=lowest
Reason
4-highest
CH3OH
OF2
Ar
MgO
Is an ionic compound: forces are stronger than forces between neutral molecules.
Has hydrogen bonds: stronger than other dipole-dipole interactions.
Is polar: dipole-dipole forces are stronger than dispersion forces.
Is nonpolar: dispersion forces are weaker than dipole-dipole forces.
Submit Answer
5 question att
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning