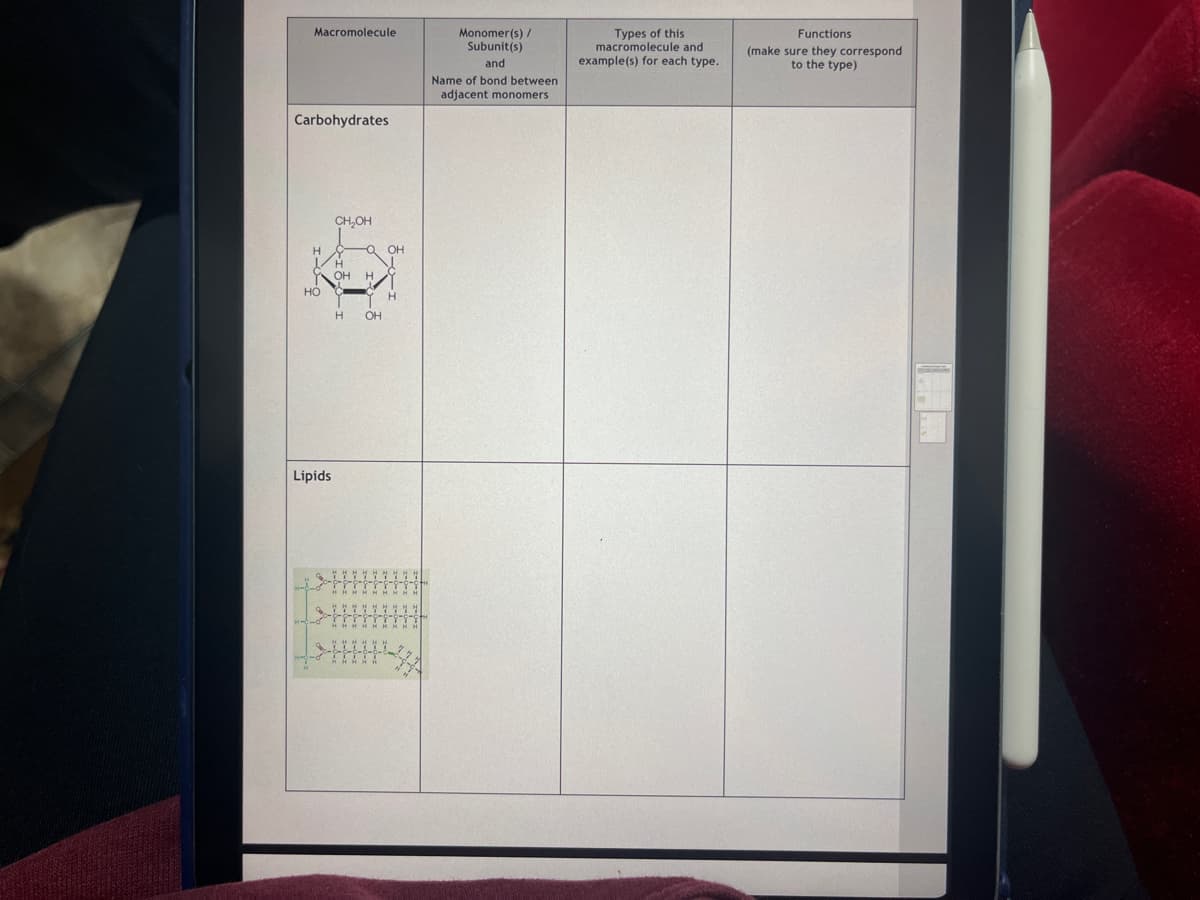
Macromolecule Carbohydrates CH₂OH H OH H H OH H HO Lipids QOH HHHH HH My HH Monomer(s)/ Subunit(s) and Name of bond between adjacent monomers Types of this macromolecule and example(s) for each type. Functions (make sure they correspond to the type)
Q: 7. Discuss the modes of actions by which the following three compounds disrupt DNA- associated…
A: Whether its a cancer cell trying to multiply inorder to generate more cancer cell or a pathogen…
Q: During Krebs cycle, the conversion of isocitrate to alpha-keto glutarate involves all of the…
A: Isocitrate to alpha-keto glutarate: Via enzyme isocitrate dehydrogenase
Q: Using the genetic code, indicate which polypeptides would be synthesized if poly-UGG were used in a…
A: Polypeptides are the linera chain of Amino acids Synthesised according to the codons on mRNA.…
Q: 1. Which of the following statement/s is/are TRUE for the protein sample?* The sample will give a…
A: Part 1. The Biuret test is used for testing the presence of peptide bonds and can also tell the…
Q: 1. Which of the following rules apply to the synthesis of nucleic acids? A. Nucleotides are added to…
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: QUESTION 2 True or False. tRNA anticodons are antiparallel and complimentary in nucleotide sequence…
A: Introduction: tRNA (transfer RNA) is a small single-stranded nucleic acid whose structure appears as…
Q: in the processing 1 molecule of pyruvate what will the atp yield is complex II of the electron…
A: Generally, 14 molecules of ATP are produced in processing one molecule of pyruvate. In the first…
Q: Determine the net ATP and total NADH produced upon complete oxidation of two molecules sucrose.…
A: Introduction: The production of ATP occurs in two ways: substrate-level phosphorylation: It…
Q: dentify any five junk foods you have taken from the last weekwhat lind of good foods can be the…
A: Introduction: A healthy diet is one that helps us to maintain or improve our health. It is necessary…
Q: 12. What are the steps in catabolism?
A: Catabolic process gives the chemical energy necessary for maintenance, and the growth of cells.…
Q: Accuracy in the translation of MRNA into the primary structure of a protein mainly depends on…
A: The translation is a process in which mRNA is transcribed into protein sequences. mRNA has universal…
Q: what are the buffers used for biological and clinical measurements? state the properties of each…
A: A buffer is a solution that can resist change in pH upon the addition of an acidic or basic…
Q: Choose your favorite drug (so far), and research its IR spectrograph, mass spectrometric…
A: In IR spectroscopy, the Infra red spectrum of the electromagnetic spectrum is used for investing…
Q: Why is it that Individuals with thiamin deficiency have high levels of pyruvate in their blood?
A: Thiamine, often known as thiamin or vitamin B1, is a vitamin that the body cannot produce. It is an…
Q: The ultimate acceptor of electrons in the elctron transport chain is ____.
A: At cellular level, the process of aerobic respiration of glucose involves glycolysis,…
Q: What is the reason why the transition state of a catalyzed reaction is lower has lower energy…
A: Enzyme is basically biocatalyst that increase the rate of chemical reaction without itself being…
Q: 2. Evaluate the secondary structure of proteins. Which of the following differentiates alpha…
A: Protein primary structures are amino acid sequences and the spatial arrangement of these sequences…
Q: what will happen (increase, decrease or no effect) to the activity of enzyme or rate of the…
A: Aleration of The activity of enzyme or rate of the metabolic pathway for given conditions are :
Q: The first step in glycogenesis is the attachment of a-D-glucose to In this process, glucose…
A: Blank 1 - Phosphate group. - In the first step of Glycogenesis Glucose is converted to…
Q: Ruhemann’s purple/blue-violet coloration in Ninhydrin test
A: Ninhydrin test is used for the detection of proteins and amino acids in a a sample. Ninhydrin…
Q: Indicate what will happen (increase, decrease or no effect) to the activity of enzyme or rate of the…
A: Glycogen synthase is an enzyme essential for the synthesis of glycogen from glucose residues.…
Q: If the pH of a voledronic acid solution is 5.8, and the voledronate concentration is 9 mM, what is…
A: Henderson Hasselbalch equation can be used to determine the concentration of a weak acid in an…
Q: Which letter statements are false
A: According to the expert answer some explaination are not complete, the false answer are given below…
Q: BIOMOLECULES - Please answer the questions properly. - Multiple choice 1. One of the following…
A: Introduction: All proteins are made up of basic secondary structure units, which can be either…
Q: The difference between prokaryotic and eukaryotic protein initiation is that eukaryotes' initiator…
A: Translation is the process of Synthesis of proteins from amino acids by peptide bond formation. It…
Q: Explain why glucose delivery is essential to health: The Impact of Diabetes.
A: Glucose is an essential metabolite which provides energy to a cell. The metabolism of glucose inside…
Q: Statement I: Glucose, galactose & fructose are absorbed in the stomach Statement II: Galactose and…
A: Metabolism is the set of chemical reactions in organisms that includes anabolism and catabolism.
Q: The mannose 6-phosphate (M6P) receptors are crucial for delivering lysosomal proteins to lysosomes,…
A: Vesicular transport is responsible for molecular traffic between specific membrane-enclosed…
Q: 10. Which reaction is convergent? Why?
A: Metabolism are consist of biochemical reactions which involved both catabolism (breakdown) and…
Q: Explain briefly but concisely 2. How would riboflavin deficiency affect the functioning of the…
A: Riboflavin is also called vitamin B2. It is a water-soluble vitamin. Through its coenzymes, it…
Q: Draw the Fischer projection of galactcose and show its catabolism until it releases carbon dioxide…
A: Galactose is an aldohexose. It is a C-4 epimer of glucose. The source for the galactose is a…
Q: 2) When a certain freon used in refrigeration was expanded adiabatically from an initial pressure of…
A: Joule Thomson coefficient is also called as Kelvin-Joule effect. It is the change in temperature…
Q: The amino acid in Hemoglobin that is most found to be most sensitive to pH changes, and hence…
A: Hemoglobin is present in RBC which carries Oxygen from lungs to cell, tissue and transports carbon…
Q: draw the structure of the substrates of the pentose phosphate pathway and write down the enzymes and…
A: The pentose phosphate pathway (or the hexose monophosphate shunt HMP Shunt pathway) is a metabolic…
Q: The definitive host of human malaria parasites are certain species of Anopheles mosquitoes. True…
A: Malaria is caused by a parasite known as Plasmodium, which is normally spread through infected…
Q: Draw the two dipeptides possible when glutamine and cysteine undergo dehydration to form a peptide…
A: Dipeptide is a peptide which contains two amino acids. Amino acids are the basic unit of polypeptide…
Q: Steroid derivatives like cholesterol are also part of the lipid family. Name three useful…
A: Cholesterol- Belongs to steroid family of lipid compounds and is a type of fat in the body
Q: 3. How is cytochrome c related to apoptosis?
A: In multicellular organisms programmed cell death is refer to as apoptosis. It is a method by which…
Q: For each genotype below, indicate whether it is heterozygous or homozygous. Write PAUL if its…
A: A particular sequence of nucleotides that encodes a product (RNA or protein) is referred to as a…
Q: The mannose 6-phosphate (M6P) receptors are crucial for delivering lysosomal proteins to lysosomes,…
A: The process of vesicular transport is an important mechanism for exchange of proteins and lipids…
Q: describe the importance of Vitamin and Mineral supplementation to human health
A: Vitamins and minerals are one of the major nutrients required by the body important for human…
Q: how many atp can a palmitic acid yield from nadh?
A: A fatty acid is made up of a straight chain of an even number of carbon atoms with hydrogen atoms…
Q: Consider the peptide EAHIVR. Write out the sequence of the peptide from N to C in three letter code.…
A: The pKa of the ionizable groups of the amino acids constituting a peptide determines the net charge…
Q: Refer to the statements below: 1. An amino acid is considered glucogenic if it has a…
A: There are twenty standard amino acids that make up all the proteins present inside the cell. Amino…
Q: Absorbance spectrum of Ninhydrin curve 1,6 1,4 1,2 0,8 0,6 0,4 0,2 100 200 300 400 500 600 700 800…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: How can enzyme inhibition be used in the treatment of certain diseases? Cite examples
A: Introduction: Inhibitors are substances that bind to an enzyme and inhibit its activity. It will…
Q: How is glycogenolysis being regulated?
A: Glycogenolysis is very important metabolic process , glycogenolysis is basically a breakdown of…
Q: The patient had been vomiting for several minutes, he threw up almost all his gastric content. What…
A: The present condition is observed by vomiting by the patient. The vomiting could be due to stomach…
Q: In a brief discussion, why is it dehydration can cause increase albumin level?
A: Plasms is the fluid portion of the blood, which consists of water, electrolytes, nutrients,…
Step by step
Solved in 2 steps

- Steroids are cholesterol-derived, polycyclic, lipid-solublemolecules that are very insoluble in water. Reaction withglucuronic acid makes a steroid much more water solubleand enables transport through the blood. What structuralfeature of the glucuronic acid increases the solubility?Thetealeavesthatcontainthecaffeinetobeisolatedarepredominantly composed of cellulose, a natural and extremely abundant polymer of glucose. Look up the chair structure of β-D-glucose (β-D-glucopyranose, the monomer from which the cellulose polymer is composed). Draw this structure, draw the flipped chair, and indicate which conformer you think is more stable, if either. BRIEFLY explain your choicewhy doesn't hexanes differentiate between a saturated and unsaturated fat or a cis/trans unsaturated fat? Explain how solubility plays a role in the effectivenss of a solvent to extracgt a particular compound. what are two different things that you can do to imporove the amount of compound extracted?
- Vasopressin is a hormone that plays an important role in social behavior, sexual motivation, and maternal responses to stress. Estimate its isoelectric point (ip) if it is known that the aminoacid sequence of vasopressin is: Cys-Tyr-Phe-Gln-Asn-Cys-Pro-Arg-GlyStructure activity relationship of pindololSteroids are cholesterol-derived, polycyclic, lipid-soluble molecules that are very insoluble in water. Reaction with glucuronic acid makes a steroid much more water-soluble and enables transport through the blood and excretion in urine. What structural feature of the glucuronic acid increases steroid solubility?
- Structural formula of a fat molecule (triglyceride) made of myristic acid, oleic acid, palmitic acid, and glycerolThe chemical basis of blood-group specificity resides inthe carbohydrates displayed on the surfaces of red bloodcells. Carbohydrates have the potential for great structuraldiversity. Indeed, the structural complexity of the oligosaccharides that can be formed from four sugars is greaterthan that of the oligopeptides that can be formed from fouramino acids. What properties of carbohydrates make thisgreat structural diversity possible?Amino acid isomerism (name types). Isomerism of the carbon skeleton of amino acids - give a few examples.
- structure of valine at pH of 12Because of their tendency to avoid water, nonpolar aminoacids play an important role in forming and maintaining thethree-dimensional structure of proteins. Can you suggesthow these molecules accomplish this feat?Disulfide bonds help to stabilize the three-dimensional structure of proteins. What amino acids are involved in the formation of disulfide bonds? Does the formation of a disulfide bond increase or decrease entropy (ΔS)?

