Q: A chemistry student must write down in her lab notebook the concentration of a solution of sodium th...
A: Given: Mass of cylinder = 5.0 g. Mass of cylinder with thiosulfate = 61.167 g. And the volume of the...
Q: The four bonds of carbon tetrachloride (CCI4) are polar, but the molecule is nonpolar because the bo...
A: Electronegativity decreases from F > Cl > Br
Q: Use a table of Standard Reduction Potentials to predict if a reaction will occur between Fe metal an...
A: For the reaction to occur , or cell will form then standard cell potential for the reaction should b...
Q: 2. A 1.00 g sample of X and Y enantiomers has a % optical purity of 65.5. What is the mass % of each...
A: koi
Q: Whether or not the process is observed in nature, which of the following could account for the trans...
A: electron capture positron emission alpha decay beta decay All these are nuclear recations.
Q: [References] The overall reaction and standard cell potential at 25°C for the rechargeable nickel-ca...
A:
Q: The second law of thermodynamics states... A that energy cannot be created equally. B that energy ca...
A: Interpretation : We have to explain second law of thermodynamics.
Q: The equilibrium constant, Kc, for the following reaction is 1.20×10-2 at 500 K. PCl5(g) PCl3(g) + C...
A: Given: Kc = 1.20×10-2 At t= 0 Moles of PCl5 : 0.259moles
Q: Given the following data table, indicate which are organic compounds and which are inorganic accordi...
A: Explanation: It's because of the difference in chemical bonds. Inorganic compounds are mostly made u...
Q: Use ANY needed reagents to effect the following transformation rting terial
A: Given are series of organic reactions.
Q: The normal boiling point of a liquid is 282 °C. At what temperature (in °C) would the vapor pressure...
A:
Q: . Propose structures for compounds that fit the following description: a. An alkyl halide that does ...
A: The chemical reaction that involves the moving out of two groups from a molecule and that leads to t...
Q: A physician orders a drug to be administered to a patient weighing 36 lbs. The dose is 55 mg/Kg TID ...
A:
Q: The director of a chemical analysis laboratory is told to expect a large number of samples for which...
A: Fajan's method is an analytical method to determine quantity of halogen ion in the sample. In this, ...
Q: Compound Melting point Boiling point Density (g/ml) Answer A -81,8°C -84°C 0,906 -79,3 126,2 0,747 C...
A: Organic compounds have lower melting and boiling points and higher density as compared to the inorga...
Q: When exposed to UV light, phosgene (COCL2) decomposes according to the reaction COCL2(g)->CO(g) + C...
A: The balanced reaction given is, => COCl2 (g) → CO (g) + Cl2 (g) Given: Initial pressure of COCl2 ...
Q: The pOH of an aqueous solution of 0.354 M nitrous acid is .
A: Given: Concentration of HNO2 = 0.354 M
Q: When exposed to UV light, phosgene (COCL2) decomposes according to the reaction COCL2(g)->CO(g) + C...
A: The balanced reaction given is, => COCl2 (g) → CO (g) + Cl2 (g)
Q: The molar solubility of silver chloride in a water solution is M.
A: Molar solubility of AgCl in water Ksp = [Ag+][Cl-] = 1.8 x 10^-10 let x amount of AgCl is in sol...
Q: Write a balanced nuclear equation for the decay of radium-226 to radon-222
A: radium has atomic number 88 with symbol Ra radon has atomic number 86 with symbol Rn
Q: e electrolysis of BiO* produces pure bismuth. How long would it take to produce 14.0 g Bi by the ele...
A:
Q: Provide the name of the functional group attached to the aromatic ring. "Do not include aromatic in ...
A:
Q: A factory wants to produce 1.00 × 10 kg barium from the electrolysis of molten barium chloride. What...
A: Electrolysis is a chemistry and industrial process that uses direct electric current (DC) to drive a...
Q: 1. The diagram shown gives the molecular orbital scheme for 1,3-butadiene. How many electrons are av...
A: Since you have posted multiple questions as per guidelines we can not answer more than one question ...
Q: the change in enthalpy for ammonium nitrate dissolving in water is 25.7 kJ/mol. determine the amount...
A: We have to calculate the mass of Ammonium nitrate dissolved.
Q: identify whether an ice cube melting has a negative or positive change in enthalpy
A: Given: to identify whether an ice cube melting has a negative or positive change in enthalpy.
Q: A 4.74 g sample of C2H3X3 (g) has a volume of 891.0 mL at 769 mm mercury and 35.0 degrees C. What is...
A:
Q: If the temperature is maintained constant, a gas sample has 2 atm pressure and occupies 5 L. The vol...
A: At constant temperature volumn of gas is directly proportional to pressure
Q: DNA contains the information to generate all of the proteins in an organism. This information is imp...
A:
Q: [References] The blood alcohol (C2H5 OH) level can be determined by titrating a sample of blood plas...
A:
Q: [References] A study was made of the effect of the hydroxide concentration on the rate of the reacti...
A: Use the table equation. Let order w.r.t [I]- be x. Order w.r.t [OCl]- be y. Order w.r.t [OH]- be z.
Q: sample of gas has a pressure of 638 mmHg at a temperature of 28oC and occupies1.75 L. If the condit...
A:
Q: Complete and balance the following redox reaction in basic solution MnO₄⁻(aq)+C₂O₄²⁻(aq)→MnO₂(s)+CO₂...
A: The reaction given is, => MnO4- (aq) + C2O42- (aq) → MnO2 (s) + CO2 (g)
Q: Give the systematic name for the following coordination compound or complex ion: (NH,),[Ni(C,O,),(H̟...
A: Naming of coordination compound 1. In coordination compound always write the name of cation before ...
Q: An electrochemical cell consists of a silver metal electrode immersed in a solution with Ag+| = 1.0 ...
A: Since Ag+(aq) has a higher reduction potential, Ag+(aq) will undergo reduction to Ag(s) and act as c...
Q: Show that CP = VT α (∂P/∂T) S
A: We have given that Show that CP = VT α (∂P/∂T)S
Q: How many moles of O₂ come from 5.0 moles of KClO₃ in this equation? 2KClO₃(s) → 2KCl(s) + 3O₂ (g)
A: We have to calculate the moles of oxygen gas.
Q: A voltaic cell is constructed from a standard Hg2+|Hg half cell (E°red = 0.855V) and a standard Cr3+...
A: We have to predict the anode reaction, cathode reaction, cell reaction and cell voltage.
Q: Select all major products of the following reactions. Me H2, Raney Ni EIOAC A B D E Me O A O B
A: This is reduction reaction.
Q: An ethanol water solution is prepared by dissolving 10.00 mL of ethanol, CH3CH2OH (density = 0.789 g...
A:
Q: Osmosis is the process responsible for carrying nutrients and water from groundwater supplies to the...
A: Given : Temperature = 350C Pressure = 18.1 atm
Q: (1.75x101) mL of 0.150 M NaOH. if the Ka of HPeO3 is (7.1100x10-7) what is the pH at the equivale...
A: Ka = 7.11 × 10-7 pka = 6.15 At equivalence point , weak acid-strong base type salt is formed. Concen...
Q: Complete and balance the following reactions. Include phases. a. _____ CH4(g) + _____ O2(g) → b. ___...
A: The reactions given are, a) CH4 (g) + O2 (g) → b) HF (aq) + KOH (aq) → c) K2SO4 (aq) + Ba(NO3)2 (aq)...
Q: A standard galvanic cell is constructed in which a Cu2+ | Cu+ half cell acts as the cathode. Which o...
A: From the electrochemical series, standard reduction potentials (E°reduction) are E°(Cu+2|Cu+) = 0.1...
Q: lons formed in a mass spectrometer will be separated by magnetic deflection. The radius of curvature...
A: Smaller the m/z ratio, higher the deflection in the mass spectrometer. Ions with lower mass and high...
Q: Which of the following species can exhibit geometric isomerism? A) [Mn(H,O),Br]* B) [Co(ox),J** C) P...
A: We have to identify the following given compounds exhibit geometric isomerism as follows in step 2:
Q: ased on nuclear stability, what is the symbol for the most likely product nuclide when iron-59 under...
A: Interpretation: We have to product the decay reaction for iron-59.
Q: 4. Can you predict the melting points of naphthalene and 1-naphthol? Structures provided if noed be ...
A: The structure of Naphthalene is
Q: What is ∆G° for a redox reaction where 6 moles of electrons are transferred and E° =-2.80 V? (F = 96...
A: Given = Number of moles of electrons (n) = 6 mol E° = -2.80 V F = 96,500 J/(V・mol) To calculate ...
Q: A 5.0 L sample of gas at 300. K is heated to 600. K. What will the new volume of the gas be?
A:
Please provide a step by step list explaining the mechanism for the reactions
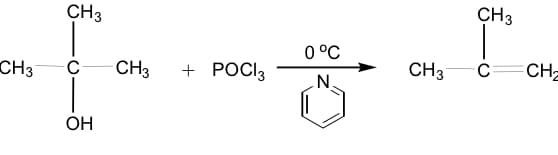
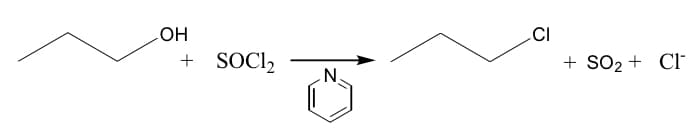
Step by step
Solved in 2 steps with 1 images

- Briefly explain the following observation. S can have an expanded octet and thus SF6 exists(~$1.00 per gram), but O cannot have an expanded octet and thus OF6 does not exist as a stablecompound.(a) The ionization energy of potassium (K) is 4.34 eV and the electron affinity of chlorine (Cl) is -3.61 eV. The Madelung constant for the KCl structure is 1.748 and the distance between ions of opposite sign is 0.314 nm. On the basis of these data only, compute the cohesive energy of KCl per ion pair. (b) The observed cohesive energy of KCl is 6.42 eV per ion pair. On theassumption that the difference between this figure and that obtained in (a) is due to the exclusion principle repulsion, find the exponent n in the formula Br^-n for the potential energy arising from this source.Zinc in its 2+ oxidation state is an essential metal ion for life. Zn2+ is found bound to many proteins that are involved in biological processes, but unfortunately Zn2+ is hard to detect by common chemical methods. Therefore, scientists who are interested in studying Zn2+- containing proteins will frequently substitute Cd2+ for Zn2+ since Cd2+ is easier to detect. Based on the properties from this chapter and their positions in the periodic tables, provide one pro and one con of using Cd2+ as a Zn2+ substitute?

