MISSED THIS? Watch KCV 8.2: Read Section 8.3. You can click on the Review link to access the section in your e Text. For the reaction shown, calculate how many moles of NO₂ form when each amount of reactant completely reacts. 2 N₂Os (g) →4 NO₂ (g) + O₂ (g) ▾ Part A 1.7 mol N₂Os Express your answer using two significant figures. ΜΠΙ ΑΣΦ Submit ▾ Part B Request Answer 6.1 mol N₂ Os Express your answer using two significant figures. 5 ΑΣΦ Submit (900) ? Request Answer ? mol mol
MISSED THIS? Watch KCV 8.2: Read Section 8.3. You can click on the Review link to access the section in your e Text. For the reaction shown, calculate how many moles of NO₂ form when each amount of reactant completely reacts. 2 N₂Os (g) →4 NO₂ (g) + O₂ (g) ▾ Part A 1.7 mol N₂Os Express your answer using two significant figures. ΜΠΙ ΑΣΦ Submit ▾ Part B Request Answer 6.1 mol N₂ Os Express your answer using two significant figures. 5 ΑΣΦ Submit (900) ? Request Answer ? mol mol
Chapter3: Stoichiometry
Section: Chapter Questions
Problem 142AE: ABS plastic is a tough, hard plastic used in applications requiring shock resistance. The polymer...
Related questions
Question
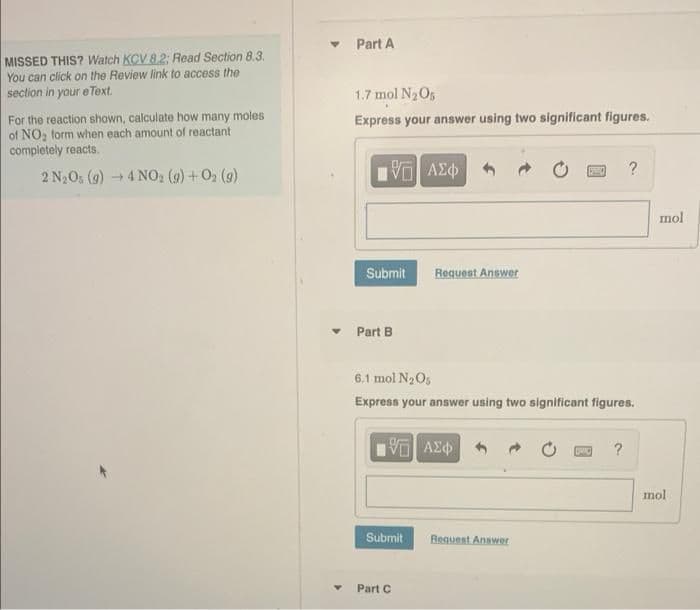
Transcribed Image Text:MISSED THIS? Watch KCV 8.2: Read Section 8.3.
You can click on the Review link to access the
section in your eText.
For the reaction shown, calculate how many moles
of NO₂ form when each amount of reactant
completely reacts.
2 N₂Os (g) → 4NO₂2 (9) + 0₂ (9)
Y
Part A
1.7 mol N₂O5
Express your answer using two significant figures.
15. ΑΣΦΑ
Submit
Part B
Submit
Request Answer
6.1 mol N₂O5
Express your answer using two significant figures.
5 ΑΣΦ
Part C
Request Answer
?
?
mol
mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning