Q: Corrosion is a redox reaction that is responsible for the failure of many water mains. These are the…
A: Some of the useful methods used to improve the situation are :
Q: -CH3 HBr -Br =CH₂ CH3 Draw curved arrows to show the movement of electrons in this step of the…
A:
Q: < Question 14 of 25 Submit What monomer is used to make the addition polymer shown here?
A:
Q: What is a correct name of the following molecule? C1 4-chloro-1,2-dimethylcyclopentane…
A: Correct ans is D) cis-1-chloro-trans-3,4-dimethylcyclopentane
Q: Explain the basis for the selective formation of the product shown over the alternative product. H₂B…
A: N atom has a lone pair of electorns which it can donate to act as a lewis base. Whereas in BH3 the…
Q: Draw the expected major product and the complete reaction mechanism for the following reaction. i.…
A: Ketones are the organic compounds that contain carbonyl group in a compound.
Q: When benzene loses one of its hydrogen atoms, it is called phenylene True Error
A:
Q: Octahedral complexes usually undergo nucleophilic substitution reactions through a dissociative or…
A: To design an octahedral complex following associative mechanism we need to find the difference…
Q: Objective 9.2 (bolded objective) Use the following set of equations: a) Metallic tin reacts with a…
A: a) When metallic tin reacts with lead (II) nitrate Solution then metallic lead and a Solution of tin…
Q: Calculate the concentration of HC6H606 in an aqueous solution of 0.0337 M ascorbic acid, H₂C6H606…
A: Ascorbic acid, H2C6H6O6(aq) is a weak diprotic acid with pKa1 = 4.17 and pKa2 = 11.6 Given the…
Q: How much heat is required to raise the temperature of a 1.15 kg piece of copper metal from 25.0 °C…
A: Given :- Mass of Copper metal = m = 1.15 kg = 1150 g Initial temperature of Copper metal = Ti =…
Q: oxidation of toluene Permanganate leads to acid formation be yourself True Error
A: Given statement is : Oxidation of toluene with permanganate leads to acid formation. Whether this…
Q: 8. Below you will find a molecule with protons labeled a-j, match the corresponding peak to its…
A:
Q: Alkene reacts with chlorine to form alcohol chloride 1-Error 2-true
A: Alkene reacts with halogens ( Cl, F, Br, I ) to form haloalkanes.
Q: Chemical waste is considered hazardous if it has one of the following properties except: Flammable
A: Hazardous waste is waste that has harmful effect on environment or health.
Q: Select the alkyl halides which reacts fastest in an SN1 reaction. Br A. CH3 CH₂CI B. C. D. CH3 CH₂…
A: We have to determine the alkyl halide that will react fastest
Q: The Lewis Dot Structure rule states that S=N-A. Recall that S represents the TOTAL number of valence…
A: The Lewis Dot Structure rule states that S=N-A. Recall that S represents the TOTAL number of valence…
Q: NAMING AND DRAWING ORGANIC MOLECULES Drawing a skeletal structure from a simple condensed structure…
A: IUPAC nomenclature is used for naming the organic compound as per recommended by international union…
Q: What is the oxidation state on the central carbon in the following compound? CH₁ H₂C-C-CH₂ 0-2 0-1…
A: Here, we have to find the oxidation states of the central carbon atoms of the given compounds.
Q: Draw the Lewis structure and determine the molecular geometry for SCN. Upload an image of your…
A: We have to determine the Lewis structure and molecular geometry of SCN-
Q: What class of nitrogen containing compounds does this structure represent? e O primary amine O…
A: Solution : Let us first discuss about the various terms included here : Primary amine : When one…
Q: Equilibrium & Solutions Test Page Which of the following would NOT represent an INCREASE in entropy?…
A: Entropy is measure of randomness of the particles. If product formed has more randomness than the…
Q: (a) the concentration gradient of hydrogen and (b) the flux of hydrogen through the foil.
A:
Q: What is the number of grams of aluminum sulfate needed to prepare a standard solution of 0.15…
A:
Q: Lorion belove Structure define aromatic, een harmedic monaromatic Explain O
A:
Q: Calculate the concentration of HC6H606 in an aqueous solution of 0.0337 M ascorbic acid, H₂C6H6O6…
A: The weak acid considered is ascorbic acid i.e. H2C6H6O6. Its conjugate base is ascorbate, HC6H6O6-.…
Q: [Review Topics] [References] Draw structural formulas for the alkoxide ion and the…
A: -> Williamson synthesis is an organic reaction forming an ether from an organohalide and a…
Q: 4. Write the product and the mechanism for reaction. Write the names of starting material and…
A: Alcohol and carboxylic acid reacts under acid medium to give ester, this reaction is often called as…
Q: A system of only gases was at equilibrium (concentrations were given) and then the volume was…
A: When the volume of the system decreases, the equilibrium shifts in favour of the direction that…
Q: 5. Write at least 4 different oxidation reactions for any hexose.
A: Hexose is defined as a monosaccharide having 6 carbon atoms.They are simple sugar molecule. Hexose…
Q: Which of the following compounds will undergo radical bromination (using Br₂ and light) with the…
A:
Q: As the alkane chain increases by CH2, the boiling point increases by 0 10-20°C 20-30°C 30-40°C
A:
Q: Its concentration is 0.1 molar and has a function of BX. solution is HX acidic, equal to 5.7. If you…
A:
Q: Determine the total hardness as ppm CaCO3 of a sample of water that has calcium content of 56 mg/L…
A: We have to find the total hardness as ppm (mg/L) CaCO3 of a sample of water. Given - Calcium content…
Q: Three moles of each reactant are placed in a reaction vessel roduct? (75%)
A:
Q: Draw a structure for the following compound: 2-Heptyne Draw a structure for the following compound:…
A:
Q: Question 9 has been shown to be an efficient support for the asymmetric 1,2-Trans-cyclohexanediol…
A:
Q: Ionic Equation: 2Na+ (aq) + SO2 (aq) + Ca2+ (aq) + 2Cl- (aq) - CaSO4(s) + 2Na+ (aq) + 2Cl- (aq)…
A: Ans: Na+ and Cl- Explanation: The spectator ions, which do not participate in chemical reactions…
Q: one liter of a 5.0% solution of NaCl. b. 10 liters of a 6.0 M NaOH solution. (MM of NaOH is 40.00…
A: The explanation is given below-
Q: Use your graph equation to calculate the expected mass loss for your Epson salt sample, then use…
A: Mass of the sample (x) 1.03 3 1.06 3.16 Mass loss on heating (y) 0.59 0.56 0.47 1.62
Q: Fill the missing in the following table : Class AB3 AB4 AB6 Number of atoms bonded to the central…
A: The molecular geometry depends on the surrounding densities around a central atom. The electron…
Q: a. Add all implied hydrogen atoms to the skeletal structure. b. How many tetrahedral carbons are in…
A:
Q: d) e) OH НО,
A:
Q: How a functional group will change the physical properties of an alkene? Explain with examples
A: Alkene are the hydrocarbons having carbon - carbon double bond ir we can say they have sp2…
Q: The glycolysis pathway is shown. Place the enzymes used in each of the ten labeled steps of the…
A:
Q: 4. Write the product and the mechanism for reaction. Write the names of starting material and…
A:
Q: Calculate the molarity of 28.0% ammonia if you know that its specific gravity is 0.898 and that the…
A: Given that - Percentage of Ammonia NH3 = 28.0% Specific gravity of the Solution = 0.898 Atomic…
Q: Draw the structural condensed formula of 4,4-dimethyl-2-hexene.
A:
Q: y isomers can be formed in each case? (2) Addition of HBr to 3,3-dimethyl-1-butene gives a mixture…
A: The addition of particular group without any substitution. For eg., HBr addition in a double bond.
Q: Introduction to Volumetric Analysis A 0.095g sample of oxalic acid dihydrate is titrated with NaOH…
A: Given the mass of oxalic acid dihydrate, H2C2O4.2H2O(s) = 0.095 g Molar mass of oxalic acid…
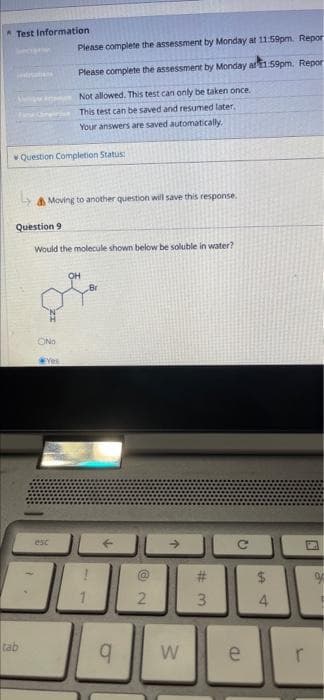
Is this molecule soluble in water?
Step by step
Solved in 2 steps

- The procedure is as follows: Weigh out around 0.3 g of cobalt chloride hexahydrate, CoCl2•6H2O, and place it in a clean, dry beaker. Dissolve it in 20 mL of 95% ethanol (95% ethanol contains about 5% water). Record your observations. Place about 3 mL of the ethanolic cobalt chloride solution in each of five dry test tubes that you have labeled A-E. Place B in a hot water bath. (See figure and instructions below.) Place C in an ice-water bath. Wait a few minutes for them to equilibrate to the temperatures of the baths. Compare the tubes with the control (the ones still at room temperature, i.e. A, D, and E), and record your observations. Add 5 drops of a saturated aqueous sodium chloride solution to D. Observe the result and compare with A. NOTE: any clear or white precipitate that forms in the bottom of this test tube is sodium chloride coming out of solution and is not involved with the chemical reaction that we are studying. Add 3 drops of distilled water to E. Compare with A and D.…Q No 10 Kindly solve this question correctly in 30 minutes and get the thumbs up please show me neat and clean work for Kindly provide correct solutionwhich of the following statements regarding purification by recrystallization is false? i. activated charcoal removes insoluble impurities. ii. water is the best recrystallization solvent for organic compounds. iii. slow cooling permits the exclusion of the solvent from the recrystallized solid. a. i, ii, and iii b. i and ii c. i only d. ii and iii
- Write TRUE if the underlined word/phrase makes the statement correct. Otherwise, write the correct WORD/PHRASE that will make the statement true. If there are two bold words/phrases in a number, write your answer for EACH of the bold words/phrases 1. 6-hydroxy-1-naphthoic acid is soluble in 5% NaOH and soluble in 5% NaHCO3. 2. The addition of excess charcoal would decrease the % recovery in the purification by recrystallization. 3. If the target compound is contaminated with impurity, the melting point will be higher than the theoretical value and the melting range is large.COMPLETE THE STATEMENT: 1. Before manufacturing process begins the sources of suitable fragrances are ______ in the manufacturing centre. 2. Oils are ______ from plants and other substances by several methods 3. During ________, flowers are spread on glass sheets coated with grease. 4. _________is the oldest and least complex method of extraction. 5. During Aging, the_____ tests whether the perfume has the specific smell or not.Answer only the one question below. This is about a labwork in chemistry, i need your help. These are the solute to be dissoolve in water and vegetable oil: Acetic acid (vinegar) Sucrose (sugar) Monosodium glutamate (vetsin) Cellulose (cotton fiber) Isopropyl or ethyl alcohol Fatty acid salts (soap) Kerosene Naphthalene balls (NOTE: So this 10 will be mixed with water and vegetable oil) Question: 1. What do you think is the main difference of the outcome of the solution of those who are mixed with water and those who are mixed the mixed with vegetable oil? Please show the difference and similaries(if theres any).
- I NEED THIS ASAP PLEASE. With step by step solution. Please, pleasevisual differences between the hydrated sample and the dried, anhydrous form?(3) GUIDE:- Substituted working formula- From the general formula, directly present your answer with substituted values already- Format: "F=_+_-_" (examples: F=2+1+1 , F=1+2+2 ... )- Spaces and other formats will invalidate your answer- Degrees of freedom- Multiple choice, each item is equivalent to one QUESTIONS TO ANSWER: 3a. What is the number of components for the formulation: "Mineral oil + isopropyl alcohol"? CHOICES: A. ONE, B.ZERO, C.TRHEE, D. TWO 3b. Present your working formula for the formulation: "Mineral oil + isopropyl alcohol" 3c. What is the degrees of freedom for the formulation: "Mineral oil + isopropyl alcohol"? CHOICES: A. ONE, B.ZERO, C.TRHEE, D. TWO
- #1. Part A. in a drug discovery program, a hit compound________. A. Shows activity in an animal model of disease B. Is active in target/ whole-cell assays in vitro C. Is purchased from a commercial vendor D. Is derived from screening natural product libraries Part B. When using TLC to monitor an organic chemistry reaction, which could be an indication that your desired reaction has gone to completion? A. the stationary phase changes colors B.the mobile phase runs to the top of the tlc plate C.the starting material spot disappears and a new spot appears D,the starting material spot remains on the tlc plateFor this statement, answer True or False and explain your answer briefly. Polarity is the only characteristic used in liquid-liquid extraction to isolate a poison from the biological matrix.Using an isopropyl alcohol, put a few drops on your palm then rub the area for a fewseconds. What is your observation? Apply the same procedures using any vegetableoil and water. What property of alcohol was exhibited relative to the vegetable oil and water? Elaborate your answer.

