nabled: Exam 2 G Saved Enter your answer in the provided box. Consider the reaction: 2NO(g) + O2(g) → 2NO,(g) Suppose that a particular moment during the reaction nitric oxide (NO) is reacting at a rate of-0.010 M/s (with the rate expressed as a negative number since NO is a reactant). At what rate is molecular oxygen reacting? Proctorio is sharing your screen. Stop sharing Hide < Prev 9 of 17 Next
nabled: Exam 2 G Saved Enter your answer in the provided box. Consider the reaction: 2NO(g) + O2(g) → 2NO,(g) Suppose that a particular moment during the reaction nitric oxide (NO) is reacting at a rate of-0.010 M/s (with the rate expressed as a negative number since NO is a reactant). At what rate is molecular oxygen reacting? Proctorio is sharing your screen. Stop sharing Hide < Prev 9 of 17 Next
Chapter12: Chemical Kinetics
Section: Chapter Questions
Problem 5ALQ: Consider the following statements: In general, the rate of a chemical reaction increases a bit at...
Related questions
Question
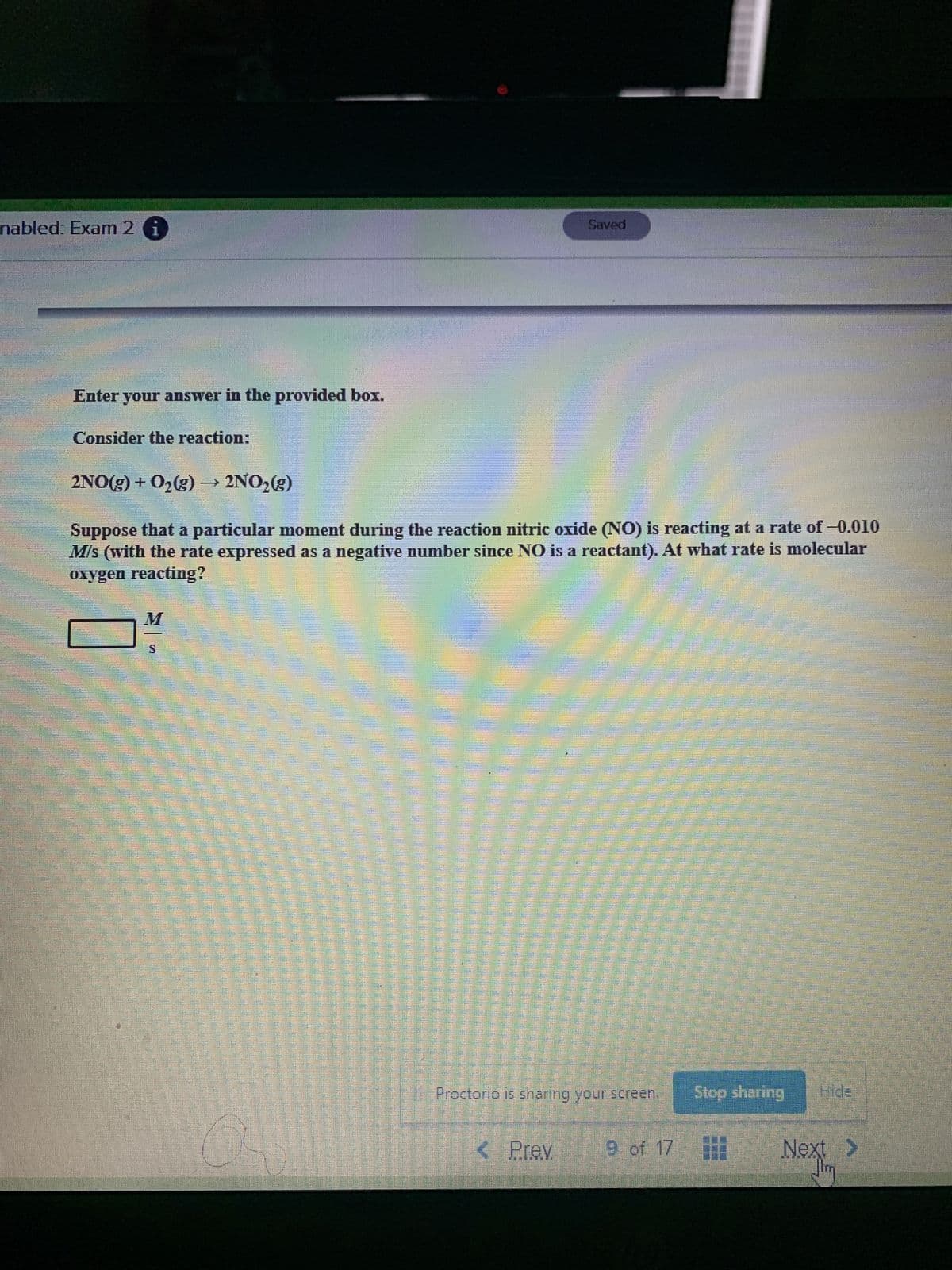
Transcribed Image Text:nabled: Exam 2 G
Saved
Enter your answer in the provided box.
Consider the reaction:
2NO(g) + O2(g) → 2NO,(g)
Suppose that a particular moment during the reaction nitric oxide (NO) is reacting at a rate of-0.010
M/s (with the rate expressed as a negative number since NO is a reactant). At what rate is molecular
oxygen reacting?
Proctorio is sharing your screen.
Stop sharing
Hide
< Prev
9 of 17
Next
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning