Q: Is the experimental value exactly equal to the theoretical value? If not, what could be the…
A:
Q: How many V atoms are there in 4.51 moles of V? atoms
A:
Q: Chemistry f) Glucose is the primary sugar in your bloodstream, supplying energy to your body.…
A: • Explanation:- Given , A constant-volume calorimeter showed that the heat generated by the…
Q: At 432 °C, Keq = 0.00309 for the reaction: NO(g) + 1/2 Br₂(g) = NOBr(g) (a) What is the value of Keq…
A:
Q: When 426. mg of a certain molecular compound X are dissolved in 30. g of benzene (C6H6), the…
A:
Q: If your answer is from the internet include the references and write your own understanding.…
A:
Q: A precipitant will form when Mg(NO3)2 is treated with which of the following? I. K2CO3 II. NaOH III.…
A: To check the precipitation, we would write the reaction between the compound and check the state of…
Q: An oxide of nitrogen contains 30.45 % N by mass. What is the empirical formula of the oxide? (MW for…
A: Molecular formula: In a molecular formula we describe the number of atoms of all the elements…
Q: Write the empirical formula for at least four ionic compounds that could be formed from the…
A: Given, Fe2+, NH4+, PO43- , CrO42- Write the empirical formula for at least four ionic compounds that…
Q: Use the following data to determine the value of F. Comment on the data and any assumptions you…
A: F Test is a Statistic test which has an F distribution under the null hypothesis. This test is used…
Q: How do the following factors affect the antiseptic activity of alcohols: concentration, structure…
A: The following factors affect the antiseptic activity of alcohols a-concentration b-structure…
Q: Select all of the correct statements about reaction quotients and equilibrium constants from the…
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical…
Q: 31. Identify the amino acid sequence by writing the One Letter Abbreviation of the amino acids in…
A:
Q: Sketch the general structure of a triglyceride molecule, name the characteristic chemical groups…
A: TRIGLYCERIDES:- Trigkycerides are formed of glycerol with long chain fatty acids.
Q: In a redox reaction, explain why the species that gets oxidized is called the reduction agent
A: In this question, we have to explain why the species that gets oxidized is called the reduction…
Q: What is the half-life of a first-order reaction if k =0.050 s¨¹? Your Answer: Answer units
A: Given that The rate constant of the first order reaction, k = 0.050 s-1 The half-life period of the…
Q: 3 Serotonin (C10H12N₂O) is a neurotransmitter that when balanced is responsible for there in 12.40 g…
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: ccess important values if needed for this question. with the charge The element chlorine forms a(n)…
A: Chlorine is a non - metal and it is denoted by the symbol Cl. Its atomic number is 17. It has 7…
Q: Which of the following represent the proper short hand form electron configuration of silver, Ag,…
A: The atomic number of Ag is 47 and it comes in 5th period of periodic table the nearest noble gas…
Q: Figure 11 BH, 2 H,O,, NaOH ?
A: We have to write the product of the given reaction. Introduction: Here the given reaction is an…
Q: Calculate the ionic strength of 0.00041 M La(IO3)3. Assume complete dissociation at this low…
A: Given 0.00041 M La(IO3)3 Complete dissociation of compound
Q: Label each reactant and product in this reaction as a Brønsted acid or base. HCN + NH₂CN + NH3 DO Or…
A:
Q: The intensity of red light (680 nm) and sorted by a solution of green dye was found to be 1.99 V and…
A:
Q: What is the activity coefficient of H+ in a solution containing 0.070 M HCl and 0.010 M Ca(C1O4)2?…
A: First, we find the value of miu (u) for this. This would be the ionic strength u = (0.070) × 12 +…
Q: What is the pH of these solutions? 0.100M sodium acetate CH3CH2O2Na, Kb=5.6x-10 A solution that is…
A: Given solutions are 0.100M sodium acetate CH3CH2O2Na, Kb=5.6x10-10 A solution that is both 0.050M…
Q: HN
A:
Q: Q2 State the number of significant figures in each of the following measurements: 4.5 m 0.0004 L 805…
A: Rule of significant digit-1) In non decimal number, 0 comes after non-zero not count in significant…
Q: C8Hg(s) + Brag) → C8Hs Bra(s) Balanced Styrene Brominc Dibromostyrene 0.50g 1.009
A:
Q: 5. Give the IUPAC name for each of the following alcohols. a. Н b. С. HO ОН она НО- CH3 CH3 -OH
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: It takes 5 days for the delivery of 198 Au grains for the implantation in carcinoma of the prostate.…
A:
Q: 48.1 mL of a 0.18 M Ca(OH)2 solution was consumed to neutralize a 0.12 M HCl solution. How many mL…
A:
Q: 11. What is the net charge on the peptide Ala-Arg-His-Ser-Lys-Glu-Thr-Pro-Tyr-Gln-Asn-Arg-His-Ile at…
A: Given peptide : Ala-Arg-His-Ser-Lys-Glu-Thr-Pro-Tyr-Gln-Asn-Arg-His-lle pH = 7.0
Q: In your experiment you have 0.2 M solutions of HCl, NaOH, and the uncharged free base form of…
A: Given Data: Volume of TEA = 2.00 L Concentration of TEA = 0.040 M pKa of triethanolamine = 7.8 pH =…
Q: Assume that we used a solute with a larger molar mass than salicylic acid, would this change deltaTf…
A:
Q: A sphere has a density of 13.6 g/mL. Calculate the volume in mL if the sphere weighs 5.0 g
A:
Q: Period: 1. The atomic number tells you the number of proton's element. It also tells you the number…
A: Please note: As per our company guidelines we are supposed to answer only three parts of one…
Q: Make atleast three combination that will result jonic bonding. What What kind of element force…
A: Solution: We know the simple rule that is opposites always attracts. Means opposite charged species…
Q: In one titration, 48.1 mL of a 0.18 M Ca(OH)2 solution was consumed to neutralize a 0.12 M HCl…
A:
Q: If 454 g of NH4NO3 decomposes, how much N₂O and H₂O (in grams) are formed? mass of mass of
A: Late of conservation states that sum of masses of reactant is equal to total mass of products.…
Q: 8. Calculate the pH and percent dissociation of 0.350 M propanoic acid, HC3H5O2. (Ka = 1.3x10-5)
A:
Q: Which of the following orbital diagrams represent the ground state configuration for an isol netural…
A: Electronic configuration: The distribution of electrons of an atom in various atomic orbitals is…
Q: If the rate constant of a reaction is 5.8 x 10-3 s1, what is the concentration after 10 seconds if…
A:
Q: When a block of alumnum loses 11,314 Joules of heat, its temperature changes from 55°C to 16°C. The…
A: The mass of aluminum block is = 326 g
Q: 33. Select the ODD one out: 000 OF OP OW OG
A: F,P,W,G are given as the codes for amino acids out of which three are similar amino acids and one is…
Q: 3. Determine Kc of the overall reaction, given: 2 O₂(g) + + Step 1: 2 CO2 (g) + H₂O (g) → Step 2:…
A:
Q: solution of carbonate (pKa = 6.3, 10.3) is at pH 10.1. Calculate the base:acid ratio at this pH.
A:
Q: Name the ff. compounds using the IUPAC system
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: Which of the following anions is the most stable? (A) ㅎ. (B) مع (C) (D) OO
A: Electron withdrawing group stabilise the anion. The presence of aromatic system stabilise the…
Q: 10 The Madrigal family grills every Sunday after their weekly soccer match with the Dorsey family.…
A:
Q: Four liquids are described in the table below. Use the second column of order of their boiling…
A: In this question, we have to match the columns with the given data of the solution.
Please help me name each
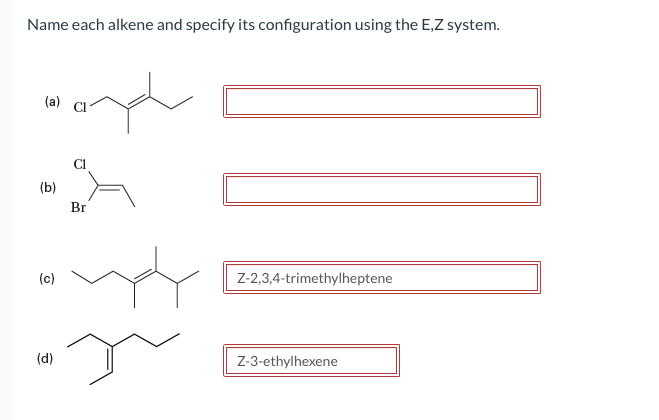
Step by step
Solved in 2 steps with 1 images

- Explain briefly and clearly the following concepts, taking as reference the molecule of n-butane and the corresponding drawings or illustrations. See pages 149-152 of the book Organic Chemistry, sixth edition (J. G. Smith). 4. What is steric hindrance in a conformation? Then draw a picture to illustrate the concept? 5. What is the torsional stress of a conformation? Then draw a picture to illustrate the concept? 6. Describe 1,3-diaxial interaction and illustrate with a specific example.Draw a three-dimensional structure for each compound, and star all asymmetric carbon atoms. Draw the mirror image for each structure, and state whether you have drawn a pair of enantiomers or just the same molecule twice. Build molecular models of any of these examples that seem difficult to you. cis-1,2-dichlorocyclobutaneDraw the skeletal (line-bond) structure of "(R)-3-ethylhex-1-ene". Useing a dash or wedge bond to indicate the stereochemistry of the substituents on the asymmetric centers, where applicable.
- For the following compound - calculate the degrees of unsaturation and enter that number into the space provided.show the completed mathematical calculation. Figures form Academia Obscura (anthonycrasto.wordpress.com)Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.)E,Z isomerism but not chiralityDraw the conformational analysis of 1-bromobutane looking at the C1-C2 bond. Provide the appropriate names for each conformation.
- Identify the stereogenic carbon in (S)- and (R)-limonene, rank the substituents around it and rationalize the assignment of their stereochemical configurations. Hint: When ranking carbons that have multiple bonds, consider the bolded carbon of C=C being connected to 2 carbons and the bolded carbon of C≡C being connected to 3 carbons.Draw a skeletal (line-bond) structure of (R)-2,3- dimethylheptane. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, where apDraw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.Chirality but not E,Z isomerism
- Calculate the degree of unsaturation for the molecular formula C6H11FS and draw a structure for the formula with a cyclohexane ring.On being heated with a solution of sodium ethoxide in ethanol, compound A (C7H15Br) yielded a mixture of two alkenes B and C, each having the molecular formula C7H14. Catalytic hydrogenation of the major isomer B or the minor isomer C gave only 3-ethylpentane. Suggest structures for compounds A, B, and C consistent with these observations.Two constitutional isomers with molecular formula C2H6O have boiling points of –23.6°C and 78.4°C. Draw both constitutional isomers of C2H6O (or provide the name). Identify the structural class of each. Match the constitutional isomers you have drawn to the boiling points provided above, and explain your reasoning.

