Q: a. How many non-polar and polar bonds are in the reactants? b. How many non-polar and polar bonds…
A: a. In reactants all bonds are non-Polar. As there is no difference in electronegativity. b. In…
Q: The following transformation can be accomplished by using to make the product. 1) Br2, FeBr3 2) HBr,…
A:
Q: H₂C H₂C CH3 CH₂ + NH3 + Br 4. .... + NH₂ Br Br H3C CH3 H3C Br
A:
Q: Consider the equilibrium system: 2 NOBr (g) = 2 NO (g) + Br₂ (g) At a 298K, analysis of the…
A:
Q: When the Following ronction Using 19 starting material 500g Propanate, A-C-C-C-0-C-4 fi Methyl…
A:
Q: For the following reaction, 21.5 grams of sulfur dioxide are allowed to react with 9.75 grams of…
A: A question based on kinetics of chemical reaction that is to be accomplished.
Q: OH 1. PBr 2. Mg 1) Bra, light 2) KOC(CH₂) 1) SOCT, 2) LEAID OH
A: #(c): Alcohol when reacts with PBr3 forms alkyl bromide which on reaction with Mg forms Grignard…
Q: 33. Provide the major product of the following hydroboration-oxidation. 1) BH, THF 2) H₂O₂. OH
A: Hydrocarbon oxidation reaction is a reaction of an alkene in which water added according to Anti…
Q: Iminodiacetic acid (IDA) is often used in ion exchange resins as a way to remove heavy metals from…
A:
Q: Which of the following is not a possible starting material for this reaction: CH₂OH но- -H но- -Н HO…
A: A carbohydrate is a biomolecule consisting of carbon, hydrogen and oxygen atoms. Glucose, fructose ,…
Q: Curved arrows are used to illustrate the flow of electrons. Follow the arrows and draw the missing…
A: Here we are required to find the intermediate and the product of the reaction.
Q: 375.m2 Solution of Nace was electrolyzed for 3.10 min. if the PH the pH at the find Solution was…
A:
Q: Phosphoryl chloride, POCI3(g), is used in the manufacturing of flame retardants. It is manufactured…
A: Given: At 298 K, the equilibrium concentration of POCl3 was 0.68 mol/L, the concentration of NO was…
Q: An analytical chemist is titrating 139.2 mL of a 0.2600M solution of ammonia (NH3) with a 0.5900M…
A: we have to calculate the pH of the solution
Q: Please briefly discuss your opinion on greening or/and growing and propose three actions that you…
A: A question based on environmental pollution that is to be accomplished.
Q: An unknown 10 gram sample in a lab required 44.9 Joules of energy to change the temperature 10…
A: The formula used to calculate heat energy is : q = m × C × dT
Q: Draw the products of the following reaction sequence. Ignore any inorganic byproducts formed. Br
A: Organic reactions are those in which organic reactant react to form organic products.
Q: Benzene Colto, ) was found to be totally unroactive toward addition Reactions. Based upon Resonance…
A: Here we have to explain why addition reaction of benzene is completely unreactive according to…
Q: 11. PF3 - use a blue ball for phosphorus when constructing this model Total # of Valence Electrons:…
A: Dear student, we are authorised to solve only one question at a time. Please post rest of the…
Q: Draw the four possible organic products for the reaction.
A: An acid anhydride is more reactive than amide, hence it reacts with ammine to form an amide. The…
Q: Question 27 Reaction below gave 3 isomers (o, p and m) of di substituted products in a ratio shown.…
A: Given reaction is called as a electrophilic aromatic substitution reaction (EAS). Reagents are…
Q: rate of 6 mcg/kg/hr in a patient who weighs 100 kg. at what flow rate in ml/hr will you set
A: Given Drug is available as = 12.5 mg in 250 ml Rate of infusion= 6 mcg/kg/hr Mass of patient =…
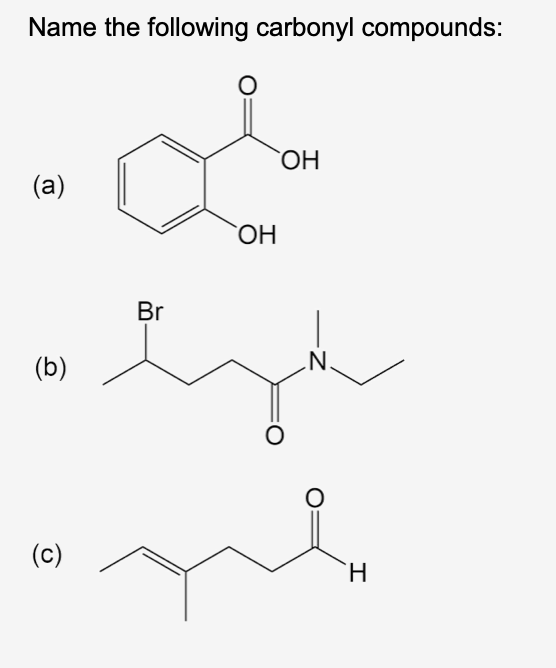
Q: Draw the chemical structure for each of the following
A: Given both compounds are same, there is only one difference between them and that is trans and cis.…
Q: b. Z: A Z D 0 B :ZII 2
A: Rules for determining major contributor towards resonance : A. Greater is the no. of covalent…
Q: 5) Draw the p-orbitals that are involved in the pi-system(s) in the molecule shown below. Make sure…
A: The pyridine ring of the above compound is aromatic due to the conjugation and followed Huckel rule…
Q: Identify the correct Fischer, Haworth, and chair structure of the a-anomer of this compound: CHO H-…
A:
Q: CH3 F CH3 B 0 10 ОН
A:
Q: What is the empirical formula of a compound composed of 35.1 g of potassium (K) and 7.18 g of oxygen…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: 6. 7. 8. Br OH NH₂ N NH₂
A: The above conversions can be carried out by using the reagents and reaction conditions in a single…
Q: Please define the term distribution coefficient, Kd? 2) How the value of Kd indicates the…
A:
Q: What are the different ways of separating mixtures? A beaker contains a mixture of iron fillings,…
A: Step 1 : Magnetic separation : Iron is magnetic in nature and the others are not so using a magnet…
Q: 2. Consider the disaccharide SUG a. The glycosidic linkage is named as b. Is it a reducing sugar?…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Draw the major addition product for this reaction. Do not include any byproducts formed.
A: Butadiene reacts with HBr to give 1,2 and 1,4 - addition products in which 1,2 - adduct is kinetic…
Q: In a titration procedure, 25.00 mL, 0.0181 M of sulfuric acid is titrated with 0.0357 M potassium…
A:
Q: Draw the structures of compounds A to D: A (C6H12) Br2, B 1) Excess NaNH, 2) H₂O с NaNH, D +
A: The chemical structure of A, B, C, D has been solved.
Q: CHỊCH SNa LII,HO 1. LMANHy CH H. Pd
A:
Q: OH 1.PBO 2. Mg OH
A:
Q: Use the following two titration curves to answer each of the following statements. Select the choice…
A: A solution with pH greater than 7 is a basic. A solution with pH leass than 7 is a acidic. A…
Q: Complete the Reaction below by providing the major product and write what the type of reaction is…
A:
Q: C2H5OH(1) has a higher entropy value than C2H5OH(g) A true b false
A: Entropy is a measure of the randomness of the system. The entropy change in a chemical reaction can…
Q: Given the following reaction: XBO3(s) XO(s) + BO2(g) If the concentration of BO2 at equilibrium was…
A:
Q: 1. Briefly describe or define and give an example of a. a weak electrolyte b. an amphiprotic solvent…
A: Define and give exmaple of the following :
Q: A 1.00-L flask initially contains 2.0 mol of A, 1.0 mol of B and 2.0 mol of C. What must occur to…
A:
Q: The air in an automobile tyre with a volume of 0.015 m³ is at 30°C and 140 kPa. Determine the amount…
A:
Q: Determine the pH of a buffer solution by constructing an ICE table, writing the equilibrium constant…
A: Buffer solution consists of weak acid and its salt or weak base and its salt. In this given problem…
Q: Calculate the pH of a 0.50 M aqueous solution of HF. (K₂ 7.2 x 104) A. 3.6 x 10-4 B. 0.019 C. 1.7 D.…
A: Given: The Ka for HF = 7.2 × 10-4 We have to calculate the pH of a 0.50 M aqueous solution of HF.
Q: Find the amount of heat energy needed to convert 25 grams of steam from 115°C to 160°C.
A: Given, mass of steam = 25 g Initial temperature (Ti) = 115 °C Final temperature (Tf) = 160 °C Note:…
Q: A student was titrating a solution of hydrazine, H₂NNH₂, with a strong acid solution. Determine the…
A: In the given question we have to fill the BCA table of titration reaction between hydrazine and…
Q: The definition for a Arrhenius acid is a compound that donates a proton (H+) when dissolved in…
A:
Q: What is one of the products of the following reaction? 8 e N=NC1 ОН OCH3 ?
A:
6
Step by step
Solved in 2 steps with 2 images

- Benzene can be hydroxylated by treating it with hydrogen peroxide and a strong acid such as trifluoromethanesulfonic acid (TfOH). Propose amechanism for this reactionIbufenac, a para-disubstituted arene with the structureHO2CCH2C6H4CH2CH(CH3)2 , is a much more potent analgesic thanaspirin, but it was never sold commercially because it caused livertoxicity in some clinical trials. Devise a synthesis of ibufenac frombenzene and organic halides having fewer than five carbons.Draw the organic products formed when cyclopentene is treated withfollowing reagent. [1] OsO4 + NMO; [2] NaHSO3, H2O
- An alkene is treated with OsO4 followed by H2O2. When the resulting diol is treated with HIO4, the only product obtained is an unsubstituted cyclic ketone with molecular formula C6H10O. What is the structure of the alkene?What is the basicity of H2SO4Draw the structure of a compound fitting each description: a.an aldehyde with molecular formula C4H8O b. ketone with molecular formulab C4H8O c. a carboxylic acid with molecular formula C4H8O2 d. an ester with molecular formula C4H8O2
- Draw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. NaNO2, HClDraw two enol forms of the following diketonewhat ate the structures for the following cinnamate reacts with methyl cinnamate reacts with ethyl cinnamate reacts with 1-propyl cinnamate reacts with 2-propyl
- An unknown compound A of molecular formula C10H18O reacts with H2SO4 to form two compounds (B and C)of molecular formula C10H16. B and C both react with H2 in the presence of Pd-C to form decalin. Ozonolysis of B forms D, and ozonolysis of C forms a diketone E of molecular formula C10H16O2. Identify the structures of compounds A, B, C, and E.Draw the organic products formed when cyclopentene is treated withfollowing reagent. [1] CH3CO3H; [2] H2O, HO−Draw the principal organic product for the reaction of 2-bromobutane with magnesium in diethyl ether, followed with benzaldehyde in diethyl ether, and then followed by dilute acid...

