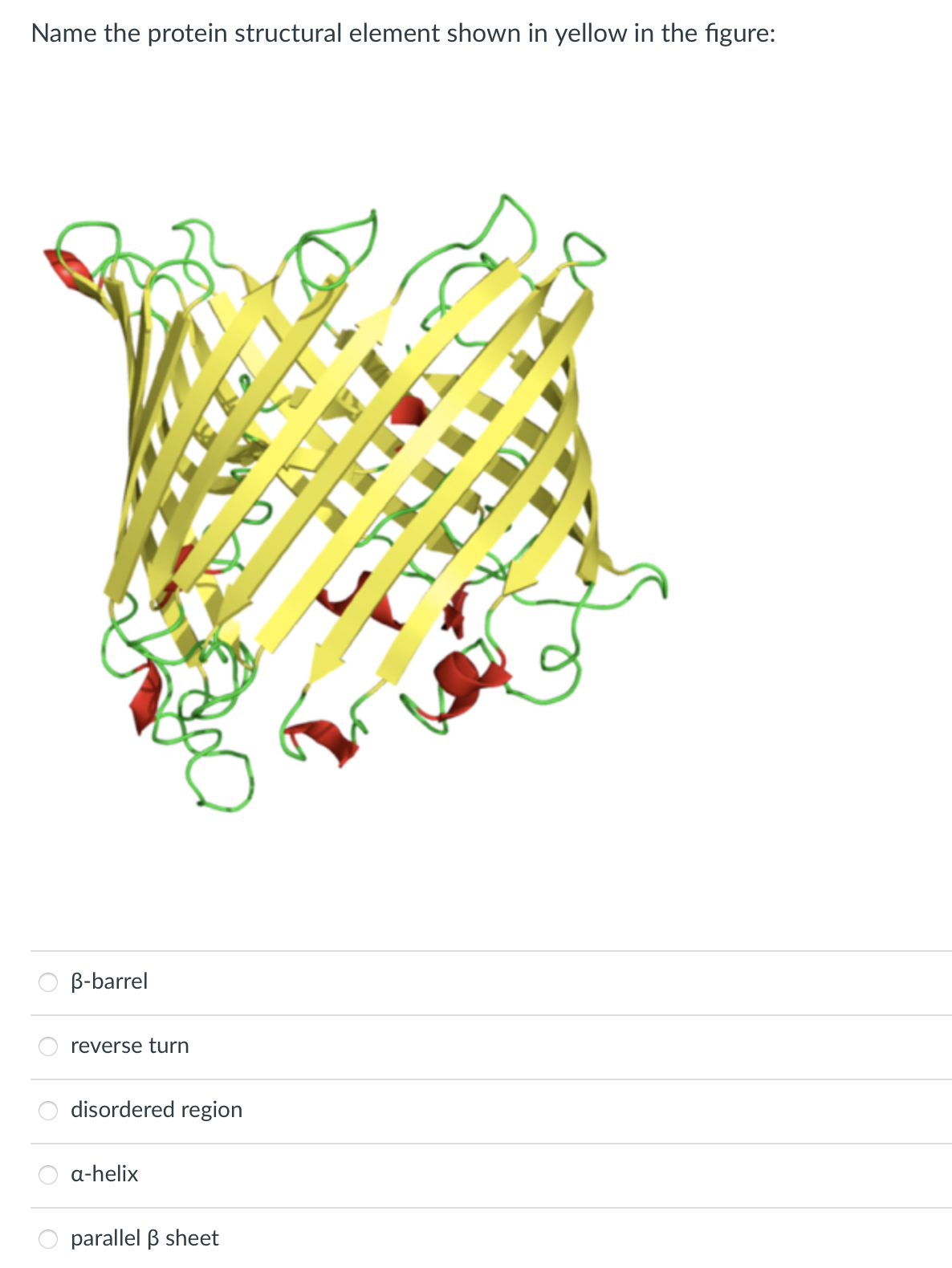
Name the protein structural element shown in yellow in the figure:
Q: GQ 7
A: The objective of the question is to determine the probability of a couple having a child with green…
Q: Draw the chemical structure of an alanine pentapeptide. Indicate the location of each peptide bond.…
A: Amino acids are biomolecules that have a carboxyl group, an amino group, a hydrogen atom and a…
Q: Which types of molecules can serve as informational macromolecules in cells? O nucleic acids O…
A: In cells, nucleic acids and proteins are the main types of informational molecules. Proteins carry…
Q: The overall, net ionic charge on this peptide at pH = 7 would be: Cys-Ala-Glu-Arg-Met-Ser +2 -1 +1 O
A: Organic substances with both amino and carboxylic acid functional groups are called amino…
Q: Amino acids- Draw out the amino acid structure for one amino acid from the list of 20. Label all…
A: Amino acids are the building blocks of proteins that are made up of an amino group and a carboxylic…
Q: The chromatogram shows fluorescent peak data from a dye-terminating nucleotide-sequencing reaction.…
A: In 1977, Frederick Sanger and his colleagues developed a method for sequencing DNA, which was later…
Q: After running a qPCR experiment, we will have graphs showing the amount of fluorescence detected by…
A: The Polymerase Chain Reaction is a qualitative procedure which is used to produce large copies of…
Q: EXAMPLE:Label the tonicity of the solution outside the cell, where Outside Inside Outside Inside are…
A: Osmosis is the movement of water across the semipermeable membrane driven by the difference in…
Q: Calculate the pH of a buffer that contains 0.75 M acetic acid and 0.35 M acetate ion in 1 L…
A: The objective of this question is to calculate the pH of a buffer solution before and after the…
Q: Draw a ribbon structure for the following polypeptide: (There is more than one right answer to this…
A: The polypeptide in question is likely to form intrastrand antiparallel beta-sheet. Beta-sheets are…
Q: Which statement best describes the protein structure shown below?
A: Proteins are a type of biomolecules that are made up of amino acids. They perform variety of…
Q: imagine that you are running a sample on the HPLC that is comprised of aspartate and isoleucine.…
A: Chromatography is a separation technique that separates a mixture of analytes based on their…
Q: The Michaelis-Menten equation models the hyperbolic relationship between [S] and the initial…
A: Those proteins or biological catalysts which help to speed up the chemical reaction are termed…
Q: Draw the chain of atoms that are part of the covalent backbone that runs the length of the DNA…
A: DNA is composed of 2 polynucleotide strands. These DNA strands have a covalent backbone which is…
Q: Genetics Question 7
A: The question is asking about the possible origins and consequences of spontaneous mutations in…
Q: Calculate the pH of a buffer that contains 0.75 M acetic acid and 0.35 M acetate ion in 1 L…
A: The objective of this question is to calculate the pH of a buffer solution before and after the…
Q: Fill in the table below Monomeric units are called: What are unique monomeric units? Linkages…
A: Nucleic acids are biomolecules that are essential for all life forms. They are polymers of…
Q: Exercise 6.3 Explain what happening (phenotype) to the following E. coli mutants when they are grown…
A: The objective of this question is to understand the phenotypic behavior of two different E. coli…
Q: 5. Explain the difference between a centriole and a centromere
A: The objective of this question is to understand the difference between two key components of a cell:…
Q: What is the pH of a buffer that contains 0.9 M ammonia and 0.3 M ammonium ion? (Ka for ammonium ion…
A: The objective of this question is to calculate the pH of a buffer solution containing ammonia and…
Q: What is the target for the following compound? O Muscarinic receptor O Acetylcholinesterase enzyme O…
A: Acetylcholine is a neurotransmitter. Many enzymes and proteins involved in the metabolism and…
Q: Experimental results describing a protein's amino acid composition are useful for estimating the…
A: Proteins are high molecular weight polymers of amino acid residues linked together via peptide…
Q: Mark any/all that apply to uncompetitive inhibition: Group of answer choices: None of these is…
A: The enzyme inhibition which is also known as anti-competitive inhibition is characterised by change…
Q: Percent saturation 100 10 0. 1 10 100 L [MM] 1000 1. Sketch a) A graded response b) c) d) e) 10000 A…
A: The Hill equation and Hill plot are powerful tools in biochemistry, used to analyze and visualize…
Q: Dihybrid Cross Problem 1: Predicting combinations of alleles in gametes of plants heterozygous for…
A: The objective of this question is to predict the distribution of alleles in the gametes of a pea…
Q: 23. Calculate the free energy of hydrolysis ATP in a rat liver cell in which the ATP, ADP, and Pi…
A: The objective of this question is to calculate the free energy of hydrolysis of ATP in a rat liver…
Q: A plot showing the % of denautration as a function of temperature for a melting point of a DNA…
A: Melting point of DNA can also be referred to as melting temperature of DNA. It is the temperature at…
Q: Assessment 2: scenario based assessment Instructions This is a ‘scenario problem’. Read the…
A: The objective of the first question is to determine the optimal speed of travel for the treasure…
Q: Mitchell presented theoretical calculations to show how a reversible ATPase system located in a…
A: In an anisotropic reversible ATPase system ATP hydrolysis is not consistent or equal in all…
Q: EF-G is a macromolecular mimic of EF-tu. It's role in translation is to To cause the large subunit…
A: Translation is the synthesis of the protein from m RNA. There are various initiation and elongation…
Q: Alpha-ketoglutarate dehydrogenase exhibits feedback inhibition by
A: Alpha-ketoglutarate dehydrogenase is an enzyme that catalyses the conversion of alpha keto glutarate…
Q: 7. Complete the scheme for isocitrate dehydrogenase. Name the reactant and product. Draw and name…
A: Kreb cycle or citric acid cycle is a sequence of reactions that take place in mitochondial matrix…
Q: A protein has a molecular mass of 400 kDa when measured by size-exclusion chromatography. When…
A: Size exclusion chromatography is a technique which separates molecules according to their size…
Q: 32) You are working with an molecule with a pl of 6.00 and the pka's of the ionizable groups on the…
A: ● If the pKa is lower than the pH the molecule will be deprotonated i.e it will carry negative…
Q: Question 1 Listen Joey has the genotype Aa; Tt where A is the gene for albinism and T is the gene…
A: The question is asking us to determine the possible types of gametes that can be produced by an…
Q: Question 4 Listen A gene known as H is epistatic to the ABO blood type genes. Based solely on this…
A: The question is asking us to determine which of the given options is possible based on the statement…
Q: Question 8 Listen In Labrador dogs, there are 2 genes that determine coat color. One gene has a…
A: The objective of this question is to determine the probability of a Labrador dog being black given…
Q: You have a separate aqueous solution of the peptide DREAMY that is at a pH of 1.0. You then proceed…
A: pH = 1/log [H+]If [H+] concentration in a solution is high, pH will be low and the solution is…
Q: Fatty Acids-Draw out one fatty acid from the list given in class. Hydrocarbon chain atoms do not…
A: Fatty acids are carboxylic acids and structural components of lipids. Fatty acids are generally…
Q: Calculate the pH of a solution containing 51 g acetic acid (K₂ = 1.75 x 10-5) and 12.3 g sodium…
A: The objective of this question is to calculate the pH of a solution containing acetic acid and…
Q: In your opinion, what is the most harmful effect of widespread opioid use? Why is opioid different…
A: A class of medications known as opioids is derived from or imitates natural chemicals present in the…
Q: Calculate the isoelectric point for histidine. (Include the chemical structures of all forms of…
A: Histidine - It is an amino acid with pyrazole ring in it. It is important to the protection of…
Q: Which of the structures above is a component of sucrose and predominantly forms a furanose ring…
A: Fructose and glucose are the two monosaccharides that make up sucrose, a disaccharide. C12H12O11 is…
Q: 2. Frank and Barbara have type B blood. Their first child has type O blood. What is the chance that…
A: The objective of this question is to determine the probability of Frank and Barbara's children…
Q: Draw the titration curve for an 800 mL 0.25 M solution of Arginine. (Graph pH vs. mole OH-.) Where…
A: An amino acid is simply an alpha-carbon bonded to 4 groups. The 4 groups are;an alpha-carboxyl…
Q: 4. Look up chymotrypsin and answer the following questions? a) Where in the body is this made? b)…
A: Chymotrypsin is a vital enzyme involved in the digestive process of animals, including humans. It is…
Q: The following question focuses on how the parameters regulating enzyme function might change, and…
A: Michaelis-Menten (MM) plot and Lineweaver Burk (LB) plot are drawn to decipher the kinetic…
Q: Which of the structures above is a component of sucrose and predominantly forms a furanose ring…
A: Sucrose is a dissacharide that can be represented as . This representation of sucrose itself tells…
Q: Amino acid analysis of the peptide gave the following residues: Asp Leu Lys Met Phe Tyr. The…
A: Edman degradation of a peptide results in the release of the N-terminal amino acid of the peptide as…
Q: Question 1: a) Below is the structure of raffinose, a main component of molasses. The trisaccharide…
A: There are four classes of biological macromolecules: nucleic acids, proteins, lipids and…
Step by step
Solved in 4 steps with 3 images

- Consider the following protein sequence as an α helix: Leu-Lys-Arg-Ile-Val-Asp-Ile-Leu-Ser-Arg-Leu-Phe-Lys-Val. how many turns does this helix make?The protein shown here is flavodoxin, which contains both alpha helices and beta sheets.If a quaternary (4°) protein structure has six N-terminus. How many total subunits does it have?
- In the following polypeptide, which amino acid would be participating in hydrogen bonding with alanine, given this sequence forms an alpha helix? Please write out the full amino acid name, not the abbreviation. Met-Ala-Leu-Glu-Lys-Thr-Leu-ValDoes right-handed alpha-helix has the least restricted ϕ and ψ angles in polypeptides, compared with left-handed alpha-helix and beta sheet?Can someone please explain why SWSTSFS has trouble becoming alpha helix and KKWWTTQ does not?
- If an Arg residue in a protein was replaced with either Lys or Glu amino acid, whichsubstitution would you expect to result in the greatest structural change and why?Consider a peptide AKCVSEAALRIKQEANSL. Which of the following secondary structures the peptide can fold into? a. colagen helix b. amphipathic alpha-helix c. alpha helix in a colied coil d. beta-strandFor the three peptides below, label whether they would form an amphiphilic beta-strand, amphiphilic helix, or nothing. Explain why. Part a) S-V-K-I-Q-M-R-A-D-L Part b) A-L-E-H-M-F-R-Y-L-A-K Part c) A-L-A-I-W-F-P-D-R-K-E
- A protein with which of the following sequences may be more prone to undergo farnesylation? (a) Trp-Ala-Ala-Cys (b) Ser-Gly-Gly-Glu (c) Ser-Ala-Ala-Trp (d) Cys-Leu-Leu-SerConvert the following sequence into protein strand using one letter code: 5'-ACCAACCAACTTTCGATCTCTTGTAGATCT-3'Consider an alpha-helix comprised of twelve amino acid residues. How many hydrogen bonds should be formed between backbone atoms in this helix?

