Q: Qno4 A Chiral B Achiral, but not meso Achiral, and meso НО OH **** OH 1 OH
A: If a molecule has a chiral carbon, then it may or may not be a chiral molecule. The best way to…
Q: Calculate the energy required to heat 180.0g of iron from 1.3°C to 11.2°C . Assume the…
A: The mass of iron sample, M = 180.0gThe specific heat capacity of iron under these conditions, C =…
Q: 4 Propose a synthesis of compound B from compound A. ??? OH A Note: More than one reaction is…
A: Given,The reaction : A…
Q: Calculate the pH at the equivalence point in titrating 0.088 M solutions of each of the following…
A: Answer:Salt of weak acid and strong base gets hydrolyzed in water to produce OH- ion, That's why its…
Q: 1. For the coordination compound: [Fe(CO) (H₂O) CL a. Can the complex ion demonstrate cis-trans…
A: Coordination number: total number of donor atoms linked to central metal atom.Complex ion of form…
Q: 5.
A: Given is organic synthesis reaction.Wey have to prepare this compound.
Q: Hello, I do not understand these questions and I am stuck. May I get help please please??…
A: The question is based on the concept of organic synthesis. We need to synthesize the product using…
Q: Phosphorus pentachloride decomposes to phosphorus trichloride at high temperatures According to the…
A: The given equilibrium is ..........(A) Temperature = T = 350 0C = 623 K Concentration of PCl5 added…
Q: Which of the following compounds or ions has the strongest conjugate acid? K₁ (HCN) = 4.0 × 10−¹⁰,…
A: Higher the value of Ka, stronger is the acid.Those acid-base pairs which differ by a proton are…
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: Given,The solutions are:2.3 mol of HNO3 is added to 1.0 L of a 1.4 M NH3…
Q: What is the missing reactant in this organic reaction? NH₂ + R + H₂O Specifically, in the drawing…
A: Given,The reaction:
Q: 2. Br + + H₂O HBr a Proton transfer b = Lewis acid/base c = Electrophilic addition Aqueous acetone…
A: Given are organic reactions.
Q: 1. Draw the product(s) of the reaction shown below and circle the major product. CH ON Br CH,OH,…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Consider the following silver-cadmium cell: Cd(s)/Cd2+(0.50 M)//Ag+(0.0025 M)/Ag(s). Compute the…
A: To calculate the cell potential for the given silver-cadmium cell at 25oC, we can use the…
Q: What elements of Democritus' theory did Dalton incorporate into his model of the atom? (Select all…
A: The transition from ancient philosophical ideas about the nature of matter to the development of…
Q: (f) NaOH, H₂O, A
A:
Q: Which major IR absorption(s) is/are present above 1500 cm for the following compound? Check all that…
A:
Q: The following compound undergoes an intramolecular Diels-Alder reaction to give a bicyclic product.…
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: When 1.10E-1 g of Zn(s) combines with 5.50E1 mL of HCl(aq) in a coffee cup calorimeter, all of the…
A: Answer:According to law of conservation of energy, energy can't be created nor destroyed. It can…
Q: 1. For the coordination compound: [Fe(CO), (H₂O)₂]Cl₂ a. Can the complex ion demonstrate cis-trans…
A: Coordination number: Total number of donar atoms linked to central metal atom.Comple ion of form…
Q: What is the IUPAC name for the compound shown? H H- I IUPAC name: -H
A: IUPAC name of the compound =?
Q: When heated, KCIO, decomposes into KCI and O₂. 2KCIO, - 2 KCI +30₂ If this reaction produced 81.4 g…
A: The mass of KCl produced = 81.4 gThe mass of O2 produced = ?
Q: Calculate the reaction enthalpy for the reaction below: C(g) 2D (g) - A(1) + 3B (g) given the…
A: The given thermochemical reactions are C(g) X(s) + B(g)…
Q: c) For the following compound identify how many chiral carbons present and how many isomers are…
A: Chiral carbon is carbon that has four different groups attached.It is denoted by *.Isomers…
Q: 4. What is the sign of the entropy change for the following reactions? Explain you a. 2Ca(s) + 02(g)…
A: The objective of the question is to predict the sign of the entropy change for the given reaction.
Q: Calcium hydroxide is "sparingly soluble" with a Ksp value of 6.5 x 10-6 . Which substance can be…
A: Answer:Le Chatalier's principle states that on changing any parameter of the system that is in…
Q: A chemistry graduate student is given 300. mL of a 0.40M methylamine (CH₂NH₂) solution. Methylamine…
A:
Q: ОН H2SO4 ОН
A: Aldehyde on treatment with alcohol in the presence of acid produces acetal.
Q: C 12 НО. В Br A х
A: Addition reactions are one of the important classes of reactions that an alkene molecule undergoes.…
Q: Need full explanation Classify each element as a representative element, transition metal, or…
A: The element De, Ge, Rh, Ta, Am, Ra, PrRepresentative element = ? Transition metal = Inner transition…
Q: 7B Compound A shown below is given. OH A |||
A:
Q: For a He⁺, calculate the wavelength of light (in m) that would be emitted for the orbital transition…
A: Given that :Initial orbit = 4Final orbit = 1The Rydberg constant = 1.09678 x 10⁷ m⁻¹
Q: Give the name for this molecule: CH3CH₂CH₂ N-H H
A: The name for the given compound.
Q: Draw the line structure of triglyceride formed from the esterification of glycerol and three…
A:
Q: 3. Draw all products for the following reactions. For your products, write whether the nucleophile…
A: To fine out Re/Si face of a carbonyl center - first prioritize the three substituents attached to…
Q: Question 4: Identify whether the following molecule is chiral or achiral. If it is achiral, is it…
A: If a molecule has a chiral carbon, then it may or may not be a chiral molecule. The best way to…
Q: 2A dentify and justify which of the structure A or B are coordination structure of compound I. | HO…
A: Coordination structure is the structure in which molecular formula is same but structural formula is…
Q: When measuring the volume of a liquid, which of these instruments should not be used due to its lack…
A: Given:a) A pipette b) A graduated cylinder c) A burette d) A beakerWhen measuring the…
Q: X N2 (g) + 02 (g) - 2NO (g) Is 2N20 (g) - 02 (g) +2N2 (g) the enthalpy of reaction for 2N20 (g) -…
A:
Q: The equilibrium constant, Kc, for the following reaction is 9.52x10-2 at 350 K. CH4(g) + CCl4(g)…
A:
Q: Which side of the following equilibrium is favored? Gl O The right side. O The left side.…
A: Answer:Any specie that loses proton in its solution is called as Bronsted-Lowry acid and the specie…
Q: How many milliliters of 0.807 M HBrO4 are needed to titrate each of the following solutions to the…
A: Answer:Mole of a specie in a solution is always equal to the product of molar concentration of the…
Q: [References] Use the References to access important values if needed for this question. The volume…
A:
Q: A reaction between liquid reactants takes place at 17.0°C in a sealed, evacuated vessel with a…
A:
Q: For each pair of substrates below, choose the one that will react faster in a substitution reaction,…
A: There are two pathways known through which substitution reactions occur ---------1. SN1 :In this…
Q: Alkene choices A₁ (5)-3-etayl cyclobantene B: 1-ettel a clobatene C: (R) - 3- ctuyl cyclobratene D:…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 6. Choose the chiral compounds F LOH OH OH CI NH₂ CI CI NH₂
A: Given are organic compounds.Chiral compounds are compounds that have chiral centers and no…
Q: Find the pH of a solution 0.20 M in NH4CI. For NH3 Kb-1.8×10-5. Report your answer to two decimal…
A: NH4Cl is a strong electrolyte and dissociates as:0.20 M 0.20 M 0.20 MSo, the…
Q: 3. Draw what you would expect to see in the 1H NMR of this compound. ur drawing clearly show the…
A: 1H-NMR spectroscopy is mainly used for the identification of the complete structure of the unknown…
Q: 4. Using the tabulated values, calculate AS for the following reactions (Table on the next page): a.…
A: Answer:Here: ai and bi are the stoichiometric coefficients
Only typed explanation
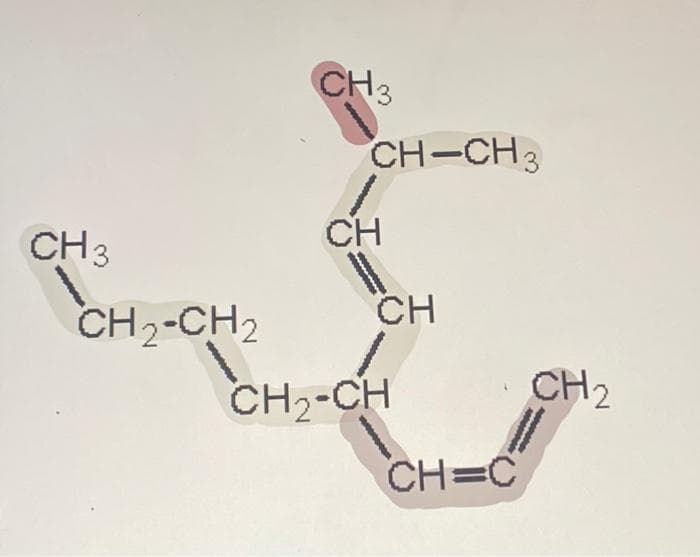
Name this compound
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 5 images

- Draw all stereoisomers formed when each alkene is treated with CHCl3 and KOC(CH3)3.Rank the following groups in order of decreasing priority. a. – COOH, – H, – NH2, – OH b. – H, – CH3, – Cl, – CH2CI c. -CH2CH3, -CH3, -H, -CH(CH3)2 d. – CH = CH2, – CH3, – C ≡ CH, – HIndicate what to do to obtain the compund (cyclohexylidenemethyl)cyclohexane from (C6H5)3P=CHCH2CH3
- What is the major stereoisomer formed when attached alkyl halide is treatedwith KOC(CH3)3?Draw the products formed when CH3CH2C ≡ C−Na+reacts with each compound. a. CH3CH2CH2Brb. (CH3)2CHCH2CH2Clc. (CH3CH2)3CCld. BrCH2CH2CH2CH2OHe. ethylene oxide followed by H2Of. propene oxide followed by H2ODraw the product obtained when cis-2-butene is treated first with Br2 in CH2Cl2, second with NaNH2 in NH3, and then finally with Li in NH3.
- The reaction CH3-CH=CH-CH3 CH3CH(I)CH(I)CH3 can be achieved withBicyclo[2.2.1]heptan-7-one + PCC (in CH₂Cl₂) => A.) Bicyclo[2.2.1]heptan-7-ol B.) Bicyclo[2.2.1]heptanoic acid C.) Cyclohexanecarbaldehyde D.) All the given choices are possible products E.) No reactionAs a rule, axial alcohols oxidize somewhat faster than equatorial alcohols. Which would you expect to oxidize faster, cis-4-tert-butylcyclo-hexanol or trans-4-tert-butylcyclohexanol? Draw the more stable chair conformation of each molecule.
- What is the major stereoisomer formed when attached alkyl halide is treated with KOC(CH3)3?What is the major stereoisomer formed when each alkyl halide is treated with KOC(CH3)3?Draw the products formed when CH3CH2C=C Na+ reacts with each compound. a. CH3CH2CH2Br b.(CH3)2CHCH2CH2Cl c.(CH3CH2)3CCl d.BrCH2CH2CH2CH2OH e. ethylene oxide followed by H2O f.propene oxide followed by H2O

