Q: At a particular temperature, the solubility of In₂(SO₄)₃ in water is 0.0065 M. a. At a particular te...
A: In2(SO4)3 dissociates into In3+ and SO42- as follows: In2(SO4)3 (s) ↔2In3+(aq) + 3SO42-(aq)
Q: A 0.400-g sample of Niobium(IV) oxide is reacted with oxygen gas, yielding 0.4256 g of a new compoun...
A: A reaction in which the number of atoms which are there in the reactant side and the product side is...
Q: Given the following standard enthalpies of hydration; Mg 2 + (g) + (ag) Mg2+(aq); A110 - 1920KJ C-n ...
A: The standard enthalpy of formation or standard heat of formation of a compound is the change of enth...
Q: 2. Silicon nitride is made by combining silicon and nitrogen gas at high temperature. How much in po...
A: Interpretation- To determine the amount of silicon ( in lb means in pound ) when Si is needed to re...
Q: 6. A certain mixture contains the following compounds: propionic acid, 2-hexanol, isoamyl acetate, a...
A: The components of a mixture in Normal liquid phase chromatography elute at different rates based on ...
Q: Select reagents from the table to show how you would carry out this synthesis.
A: The given substrate is 1-pentene. The given reaction is the conversion of 1-pentene to pentanal. The...
Q: Ethanol, C2H5OH, boils at 78.29°C. How much energy, in joules, is required to raise the temperature ...
A:
Q: According to the balanced reaction below, calculate the moles of NH3 that form when 4.2 mol of N2H4 ...
A: Number of moles of product formed can be obtained with the help of the balanced chemical equation.
Q: 1. Draw the Lewis Structure of CCl4 2. Determine the Electron Domain Geometry and Molecular Geometry...
A: 1) C atom has 4 e- in the outermost shell. It requires 4 e- to complete its octet. Cl has 7 e- in th...
Q: Which of the following is the best reducing agent: F2,H+, Na, Na+, or F-? Explain. Order as many of ...
A: Reducing agent is one that reduces other. They are electron donors.
Q: A mixture contains NaHCO, together with unreactive components. A 1.61 g sample of the mixture reacts...
A:
Q: If the molar mass of the hydrocarbon C3H8 was determined to be 440.97 g/mol, what is the molecular f...
A:
Q: Please help
A: Numbers and alphabets are applied to form a chemical formula which represents a chemical compound.
Q: Write a balanced equation for the complete oxidation reaction that occurs when acetylene (C,H) burns...
A: The combustion reaction of acetylene: It is a chemical change which occurs when acetylene gets oxidi...
Q: Which of the following has an effect on the magnitude of the equilibrium constant? Question 4 op...
A: Correct answer would be changing the temperature.
Q: 17 How many resonance structures are there for the NO3- polyatomic ion? Group of answer choices 1...
A: Given structure for drawing Resonance is NO3-
Q: Silver chromate is sparingly soluble in aqueous solutions. The Ksp of Ag2CrO4 is 1.12×10−12 M^3 . ...
A: (a) Solubility equilibrium: Ag2CrO4⇌2Ag+ (aq)+ CrO42- (aq)I (M) ...
Q: For each of the following reactions, predict the major product. Nao 1. Li (s) 2. `Ph Br 3. HСI, Н2О ...
A: Since you have posted a question with multiple sub-parts, we will solve first three subpartsfor you....
Q: Describe how the properties of water in relation to IMFs contribute to the upward movement of water ...
A: The upward movement of water is best explained by the cohesion-tension theory, also known as transpi...
Q: What is the absolute magnitude of the rate of change for [NH₃] if the rate of change for [H₂] is 2.0...
A: For a reaction, aA. + bB. →cC + dD rate of reaction (r) =k[A]x [B]y = -1/a( ∆[A]/∆t) =...
Q: How many milliliters of 1.0OOM sodium hydroxide are needed to neutralize 20.67 mL of 0.750M phosphor...
A:
Q: Round each number as directed: a. 429.894 – tenth's place c. 14300. – hundred's place b. 344321 – th...
A: Rounding off the given digits: a) Rounding off 429.894 to tenth's place = 429.9 Here we have rounded...
Q: 16 In carbon disulfide, how many electron pairs are shared between carbon and one sulfur? Group of...
A:
Q: Question 2 of 20 What is E at 25°C for the reaction? Mg(s) I Mg2+ (2.0 M) |I Pb2+(0.10 M) I Pb(s) V ...
A: Given, T = 25°C = 273+25 = 298 K E°cell = 2.231 V
Q: Find the concentrations of all ions in the solution at equilibrium after 0.520 L of 0.430 M aqueous ...
A: Number of moles of copper (II) nitrate = 0.52 L × 0.43 M = 0.224 mol Number of moles of potassium hy...
Q: When 2.24 mol KOH is added to 1.00 L of 0.50 M Al(NO3)3. After the aluminum nitrate has reacted with...
A: The limiting reagent in a chemical reaction is a reactant that is totally consumed when the chemical...
Q: What is the molar solubility of Pb(OH)2 in a solution that is 0.10 M NaOH? The Ksp for Pb(OH)2 is 2....
A: The reaction involved is: Pb (OH)2→Pb+2 + 2OH-Time=0 s 0 ...
Q: 11 Which of the following elements is the most electronegative? Group of answer choices Ne Rb ...
A: The given elements are Ne, Rb, P, I, Cl. Electronegativity is the tendency of an atom to attract a ...
Q: Multiple-Choice Questions 1. Determine the density of NO gas at 101 kPa and 35 °C in g dm. A 0.012 B...
A:
Q: 7. You are helping to clean out the lab at the end of the term and find a bottle of liquid whose lab...
A: Given : The bottle can have either C3H8O or C4H8O2 or both. Mass of sample burned = 0.300 g Mass of ...
Q: Menthol, a terpene, exists in a (1R,2S,5R) and(1S,2R,5S) form. Draw the two molecules in theirmost s...
A:
Q: A chemical reaction has an equilibrium constant of 2 × 106. If this reaction is at equilibrium, sele...
A: Equilibrium constant tells about the relation between reactants and products at equilibrium.
Q: A photon has a wavelength of 286 nm. Calculate its frequency and energy
A: Electrons can behave as a particle as well as wave. The dual nature of electron was explained by De ...
Q: Determine the pH change when 0.128 mol KOH is added to 1.00 L of a buffer solution that is 0.445 M i...
A: Given: Buffer solution - 0.445 M in HF and 0.255 M in F- Volume of buffer solution is 1.00L Moles o...
Q: The reaction quotient, Q, for a reaction has a value of 75 while the equilibrium constant, K, has a ...
A: Given: The reaction quotient, Q, for a reaction has a value of 75 while the equilibrium constant, K,...
Q: what is X-Ray ? what is use of it?
A: X-ray is an electromagnetic radiation having wavelength between 0.01 to 10 nm. They are produced whe...
Q: 5 Select the correct electron configuration for Te. Group of answer choices [Kr]5s25p64d8 [K...
A: Tellurium has atomic number(Z) = 52.
Q: How many grams of sodium acetate (MM=105.99) would you add to 500.0 g water to produce a freezing po...
A: Using the freezing point depression formula ∆Tf = i×Kf×mi = van't hoff factor∆Tf = freezing point de...
Q: ball & stick -- labels Name the alkene from which the 1,2-diol above was made. Submit a single name ...
A:
Q: Chemistry Question
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and s...
Q: What is the difference between dipole–dipole interactions and London dispersion forces?
A: Intermolecular force of attraction that holds the different molecules of a substance together is kno...
Q: What is Solubility Equilibrium(Ksp )
A: Solubility equilibrium is a type of dynamic equilibrium that exists when a chemical compound in a so...
Q: 15 Select the best Lewis structure for OF2: Group of answer choices D A B C E
A: Lewis dot structures are the diagrams which represent the shared pairs in a chemical bond. It shows ...
Q: 1. Draw the Lewis Structure of SeS2 2. What is the Electron Domain Geometry of SeS2 3. What is the M...
A: The Lewis structure or molecular skeleton is also known as electron dot structure. It is the complet...
Q: Part A. If 5.00 mol of hydrogen gas and 1.00 mol of oxygen gas react, what is the limiting reactant?...
A: Part A: The balanced reaction is:' 2H2(g)+O2(g)→2H2O(l) According to the balanced reaction, 2 mol of...
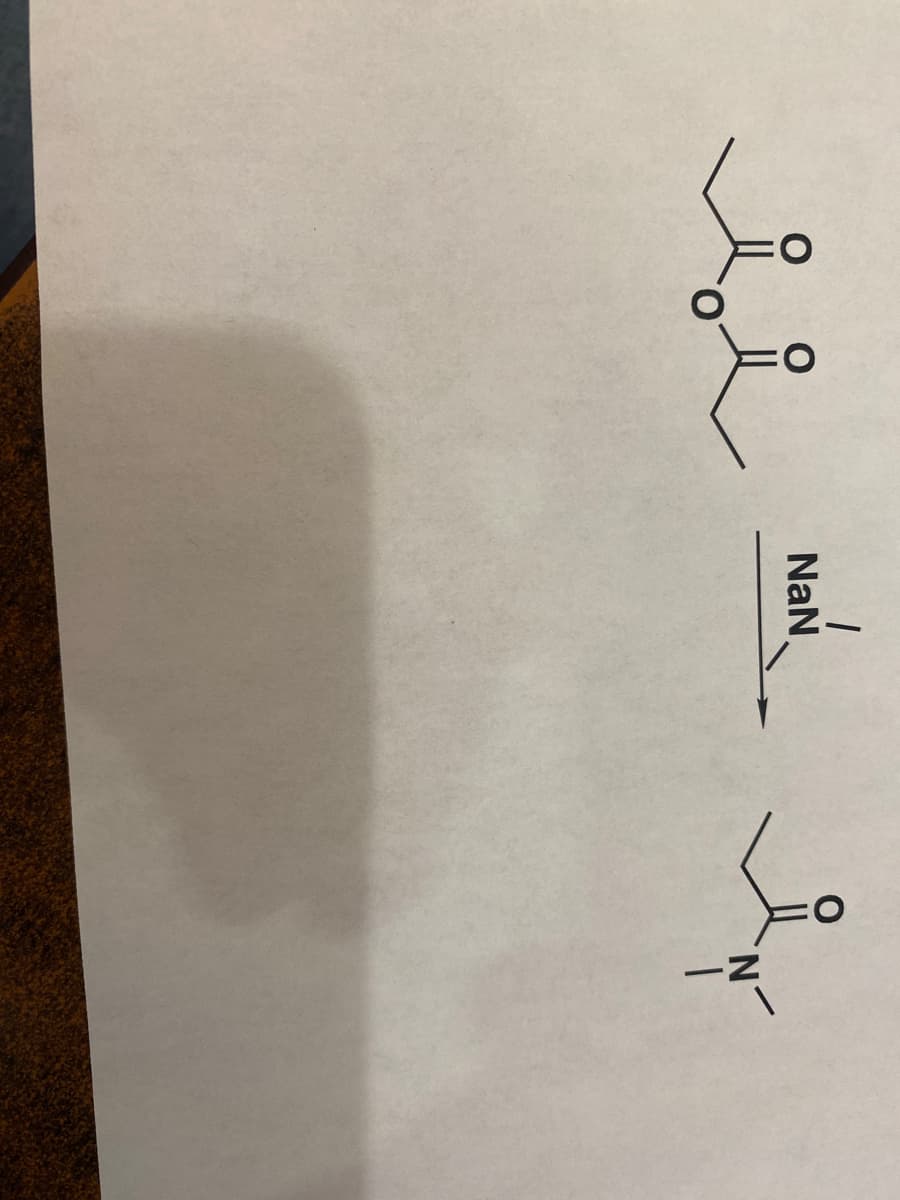
Nucleophilic addition- elimination reaction is a reaction in which the substitution of any group takes place by another group.
Step by step
Solved in 2 steps with 1 images


