Q: B) 1. Provide TWO Grignard reactions, A and B, that can be used to make the provided target…
A: Answer below this question Explanation:
Q: Draw the structure of A, the minor E1 product of the reaction. Br CH₂OH, D A
A:
Q: Draw the missing organic structures or select the missing reagents in the following multistep…
A: Given,The reaction is:
Q: :0: K 10: Select to Add Arrows CH3CH2ONA ,CH3CH2OH > CH3CH₂ONA, CH3CH2OH JOH CH3CH₂ONA CH3CH2OH…
A: The objective of the question is to draw the curved electron-pushing arrows to represent the…
Q: 4 Part A Which of the two acid solutions is more concentrated? Acid (b) is more concentrated. Acid…
A: The titration curve for two weak acids is given in the question.Both are titrated with 0.100 NaOH
Q: 6. What is the relationship between the following two compounds? A) Constitutional isomers B)…
A: If carbon atom is surrounded by four different groups or atoms then that carbon known as chiral…
Q: Et20 H2O-H2SO4 BH3-H2O2 EtOH B(OH)3-H2O NaOEt Which reagent would you use to accomplish each of the…
A: Answer:In this problem we have to convert the given compound into an alkene and then have to perform…
Q: b. enamine: H3C NH2 N' H O c. imine: NH3 Θ NH3 OH 70
A: When secondary amine react with ketones they form enamines, catalysed by adding a weak acid.If…
Q: what product would result from the following reaction? ΟΙ O II O III O IV Br Br2 light Br Br Br III…
A: When ethylbenzene reacts with bromine (Br2) in the presence of light, a substitution reaction known…
Q: C. Reaction with phenylmagnesium bromide in THF, followed by H+, H₂O. Select Draw Templates More…
A: To perform the listed reactions with succinic anhydride.
Q: States of Matter = Using the combined gas law For many purposes we can treat butane (C4H10) as an…
A: The objective of the question is to determine if it is possible to change the temperature of butane…
Q: Q37. What will be the rate equation for the reaction 2X + Y → Z, if the order of the reaction is…
A:
Q: 2H2S (g) + O2(g) 2S (s) + 2H₂O(1) redox reaction? O yes reducing agent: O no ☐ oxidizing ☐ agent:…
A: Answer:Oxidizing agent is the chemical specie that gets reduced during the redox reaction and…
Q: Im sorry, what happens in step three to step four?
A: Resonance structure III is not related to resonance structure IV.In the given compound (I), the…
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A: In Micheal's addition reaction, a nucleophilic addition reaction takes place at the carbon-carbon…
Q: When 25.0 mL of a 7.04×104 M sodium carbonate solution is combined with 25.0 mL of a 3.84×104 M…
A: We know if the Ionic product is more than solubility product then precipitate forms.If ionic product…
Q: is dication cyclobutadiene aromatic? Based on what I know it is but im unsure about the conjugation.
A: The question is asking whether the dication form of cyclobutadiene is aromatic or not. Aromaticity…
Q: Using the periodic table as a guide, which of the following choices orders the elements correctly…
A: The objective of the question is to verify the order of the elements based on the trends stated in…
Q: Draw the major organic product(s) of the following reactions including stereochemistry when it is…
A: Hydration of alkyne will lead to ketone via keto-enol tautomersim. Intially hydrogen and hydroxy…
Q: Three gases (8.00 g of methane, CH4, 18.0 g of ethane, C2H6, and an unknown amount of propane, C…
A: Given,mass of methane gas ( CH4 ) = 8.00 gmass of ethane gas ( C2H6 ) = 18.0 gtotal pressure = 5.30…
Q: The rate of a reaction is how quickly the reaction goes to completion. If two reactions have the…
A: The objective of this question is to understand how the concentration of reactants affects the rate…
Q: Robinson Annulation 5 Try the mechanism again on your own. H3CO Base
A: The Robinson annulation is an organic chemical reaction in which a ketone having -hydrogen is…
Q: Can the molecule on the right-hand side of this organic reaction be made in good yield from no more…
A: Given is organic compound.Given compound is fused ring compound. As we can see one six member ring…
Q: A chemist prepares a solution of mercury(II) iodide (HgI2) by measuring out 0.00087 µmol of…
A: Molality, molarity, normality, mass percent, and volume percent are all quantitative concentration…
Q: Balance this equation Mn2+(aq) + O2(g) → MnO2(s) in basic solution.
A: A redox reaction is balanced by balancing the mass and charge. Hence the number of atoms of all the…
Q: Which of the following ions has a higher concentration in Sea water on average, according to your…
A: Thus, the correct answer is C. Na+ (Sodium).Explanation:Step 1: The correct answer is C. Na+…
Q: The rate of a reaction is how quickly the reaction goes to completion. If two reactions have the…
A: The rate of a reaction is how quickly the reaction goes to completion. If two reactions have the…
Q: raw the major organic product(s) of the ollowing reactions including stereochemistry when it is…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: AIC3 AICI 3 KMnO4 NaOH
A: In Friedel-Crafts alkylation, an alkyl group is introduced onto an aromatic ring using a Lewis acid…
Q: Predict the major organic product of this reaction: Select one: CI S S CI _SH CI ?
A: Nucleophilic substitution reaction is a class of organic reactions where one nucleophile replaces…
Q: Table 1. Standardization of HCI Solutions (40 pts) Trial Volume of HCI (To the nearest 0.01 mL)…
A: The objective of the question is to calculate the uncertainty in the molarity of the HCI solution…
Q: Eolie ? & EtO OEt
A: The given synthesis is from diethyl malonate to 5-methylcyclohexane-1,3-dione. This synthesis can be…
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A: Sum of oxidation state of each atom present in the compound is equal to the charge on that compound…
Q: Predict the major products of the following organic reaction:
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: Draw the Lewis structure for NO2, including any valid resonance structures. Which of the following…
A: Ions are the species that carry a charge. Positively charged ions are known as cations and…
Q: Be sure to answer all parts. How many inner, outer, and valence electrons are present in an atom of…
A: Outer Electrons : Electrons located in the highest energy level (shell) of an atom.Inner electrons…
Q: Draw the major organic product(s) of the following reaction. NaNH2 / NH3(1) CH3CH2-CEC-H CH3…
A: Terminal alkyne undergo alkylation when react with a weak base or conjugate base followed by the…
Q: Please predict the products for each of the following reactions. Clearly show the regiochemistry…
A: In presence of O3 , alkene will generate ozonide.In presence of Me2S, cleavage occurs to generate…
Q: Draw a structural formula for N - phenylpentanamide.
A: Answer:Structural formula is the representation of the molecule in which all the atoms present in…
Q: Copper(I) ions in aqueous solution react with NH3 (aq) according to Cu+ (aq) + 2 NH3 (aq). →…
A: Two equations can be represented as; CuBr ⇌ Cu+ + Br- ...KspCu+ + 2NH3 ----> [Cu(NH3)2]+…
Q: Draw the major product(s) of the following reaction. CH3 HBr •You do not have to consider…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Report the mean concentration of salicylate in acne face wash (in units of weight percent) with…
A: The objective of this question is to calculate the mean concentration of salicylate in the acne face…
Q: Temperature, T (°C) Volume in syringe, Vsyringe (mL) Total volume, V (mL) 21.5 45.50 227.50 30.0…
A: The slope of a line is the ratio of the amount of y increases as the value of x increases. It tells…
Q: None
A:
Q: Splitting of a signal in a proton NMR spectrum tells us the number of chemically non-equivalent…
A: In proton NMR, the splitting pattern gives an insight into the structure of a molecule. The number…
Q: Provide the following information for a photon in the Ultraviolet region of the Electromagnetic…
A: The objective of this question is to calculate the energy and frequency of a photon in the…
Q: Add ONE remaining curved arrow to draw the next step of the mechanism. Modify the given drawing of…
A: The product which is formed in this reaction is given below-
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A: Solubility of a compound changes with PH, if any constituent ion of the compound reacts with Proton…
Q: MCPBA-H3O+ OsO4-H2O2 CH3CO3H H2O-H2SO4 03-Me25 Which reagent would you use to accomplish each of the…
A:
Q: Part A One kind of polyester is a condensation copolymer formed between terephthalic acid and…
A: The monomers given are,
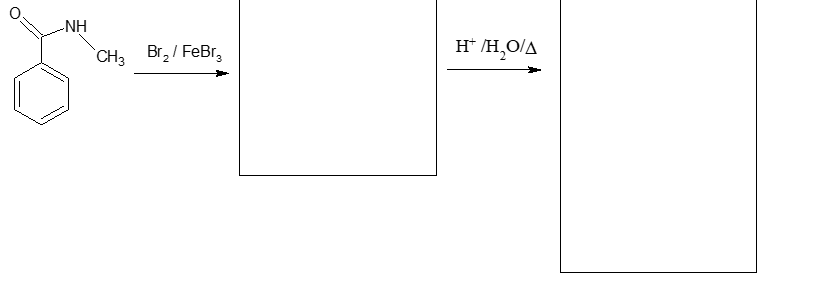
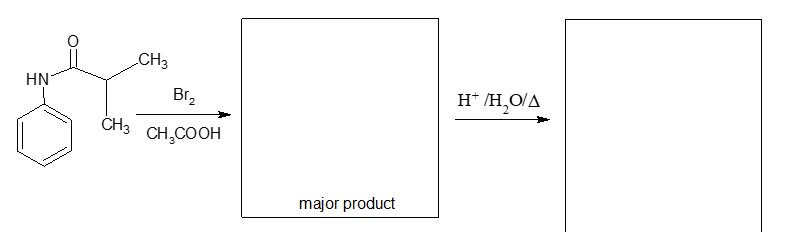
Fill in the boxes below with the appropriate reagents or structures. Each part includes a hydrolysis reaction, thus the boxes should contain two products (assume that the pH has been adjusted so that the products exist as neutrally charged species).
Step by step
Solved in 3 steps with 2 images

- Isoleucine, another of the twenty amino acids found in proteins, is metabolized by a pathway that includes the following step. Propose a mechanism.What reagents are needed to convert (CH3CH2)3CC=CH to each compound?Draw the products formed (including stereoisomers) when each compound is reduced with NaBH4 in CH3OH.
- Which of these reagent(s) will not react with HOCH2CH2CH2COOH? A) NaCN in ethanol B) C2H5OH in the presence of an acid catalyst. C) (CH3CO)2O D) Concentrated H2SO4Draw the products formed when CH3COCH2CH2CH = CH2 is treated with each reagent:(a) LiAlH4, then H2O;(b) NaBH4 in CH3OH;(c) H2 (1 equiv), Pd-C;(d) H2 (excess), Pd-C;(e) NaBH4 (excess) in CH3OH;(f) NaBD4 in CH3OH.Which substance in each pair should have the greater ∆Hvap? CH3CH2CH2CH2CH3 or CH3CH2CH2CH2CH2OH CH3–O–CH3 or CH3CH2OH

