Q: Balance this redox reaction using change in oxidation number. H3PO2 + Cr2O7^2- + H+ —> H3PO4 + Cr3+…
A: …
Q: From each of the following pairs of substances, Choose the one that is the stronger…
A: SOLUTION: Step 1: An oxidizing agent is a chemical species that tend to oxidize other substances.…
Q: 5.What
A: 1) Oxidation number of Rb = +1 2) Oxidation number of O = -2 3) Oxidation number of Tc = Findout…
Q: Determine whether eachof the following substances is likely to serve as an oxidantor a reductant:…
A: (a)
Q: Define the term nonzero oxidation states?
A: Oxidation state or oxidation number depicts the charge carried on a chemical atom in order to make…
Q: Assign oxidation numbers to each element and balance the equation in a basic solution. H2O2 + ClO2…
A: The balanced equation in basic medium is given below
Q: Which ion can be most easily reduced? O a. Zn2+ O b. Ca2+ O c. Cu2+ O d. Fe2+
A:
Q: Please draw the structure of the possible product for the following reaction, and how does the…
A:
Q: Assign an oxidation state to each element in each compound. a.) Cu(NO3)2 b.) Sr(OH)2
A: The oxidation state of each ion in the given molecules is as follows:
Q: solution containing Fe2+ requires 26 mL of 0.0265 M K2Cr2O7 in acidic solution. Balance the…
A: When potassium dichromate oxidise iron in presence of acid, then Iron (II) is changed to iron (III)…
Q: In the smelting of iron from iron oxide according to the equation what is the change in oxidation…
A: Since in Fe2O3 we have O in 2- oxidation state Hence total -ve charge due to O = 3 X (-2) = -6 And…
Q: In which of the following unbalanced reactions does chromium undergo oxidation ? CrO 2 Cr20,2 Cr3+…
A: The process in which a substance loses an electron duringa chemical reaction is known as…
Q: What is the change in oxidation state for the chromium atom in the following unbalanced reduction…
A: Assigning the oxidation number involves a few simple steps. The sum of each element's oxidation…
Q: what is the oxidation number of each atom in ammonium sulfite (NH4)2SO3?
A: Oxidation number is the charge on an element in its bonded state. The given compound is ammonium…
Q: Given the reaction: Fe +2+ Cr₂07-2Fe +3 + Cr*3 Which is the reducing agent? O Cr+3 O Cr₂07 7-2 O Fe…
A: The substance which oxidises others but itself reduced is called oxidising agent. The substance…
Q: In which compound does carbon have an oxidation number of -3? a) CO b) CO2 d) CH. c) CH,
A: Oxidation Number of O = -2 Oxidation number of H = +1
Q: Determine the oxidation number of the transition element in the following: (a) MnO2 2+ (b) Mn² (c)…
A: Oxidation Number: oxidation number, also called oxidation state, the total number of electrons that…
Q: d) FeCl3 + SO2 + H2O → FeCl2 + HCI + H2SO4
A:
Q: Détermine the oxidation number for the following compound Mg in MgCl2 2+ 1+ 2- 1- 0
A:
Q: rite a balanced equation for the reaction m and zinc metal to yield zinc(I) and chromium(III) in…
A: Balance chemical equation is defined as which has equal number of atoms or ions in both sides…
Q: Find the element with the highest oxidation number in each of the following formulas: 1.Na2Cr2O7…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Be sure to answer all parts. Assign oxidation numbers to all elements in the following compound and…
A: Oxidation number: During the formation of chemical bonds with other atoms of a different element,…
Q: What is the equilibrium constant for the following redox reaction? Cu2+ + 2Li Cu + 2Lit Litte Cu²+ +…
A: Given, Cu2+ + 2 Li ------> Cu + 2Li+ Standard reduction potential are given, Li+ + e- ---> Li…
Q: Assign the oxidation numbers (ON) Zn S8 NaCl Cu2O CuO H2O2 H2O CaH2
A:
Q: Show the complete step on how to derive the balanced net ionic equation of Limestone in the presence…
A: Calcium carbonate decomposes into calcium ions and hydrogen carbonate ions when it is dissolved in…
Q: Given that sodium oxalate is dissolved in a flask and is titrated to its equivalence point with 30…
A:
Q: Assign oxidation numbers to each element and balance the equation in an acidic solution. MnO4- +…
A: The sum of the oxidation numbers of any species should be equal to the net charge on that species.
Q: Which is more reducing, a solution with 20 mM sufide (HS-; assume S0 as product of sulfide…
A: A reducing agent is the species defined as the substance which oxidizes itself while reduces others.…
Q: Is C18H32O2+2H2 =C18H36O2 an oxidation or reduction reaction?
A: Oxidation : Oxidation involes loss of electrons and in this process oxidation number of reactant…
Q: Assign oxidation numbers to each atom in potassium dichromate, K2Cr207 I
A:
Q: III. Assign an oxidation state to each atom in each of the following: 1. Zn 2. Cu+2 3. CaCl₂ 4. CF4…
A: Oxidation state is defined as the real or imaginary charge present on an atom in a species on the…
Q: Which is the strongest oxidizing agent in 1M solution from the reagents below? Ca+2 Ag+ Li+ Cu+2…
A: A chemical reaction in which loss of electron and gain of electron takes place simultaneously is…
Q: Which of the following can act as an oxidizing reagent but not a reducing reagent? Zn Cl- Sn2+…
A: The oxidizing agent is a substance that causes oxidation by accepting electrons; therefore, its…
Q: The following skeletal oxidation-reduction reaction occurs under basic conditions. Write the…
A:
Q: Assign an oxidation state to each atom in each compound. (c) SO2 (d) H2S
A: The oxidation state of each atom in the compound has to be provided.
Q: From the equations below, identify the following: a. Oxidized element b. Reduced element c.…
A: (iii) a. Oxidized element = Chlorine.b. Reduced element = Chromium.c. Oxidizing agent = K2Cr2O7d.…
Q: The Haber process is the principal industrial route for converting nitrogen into ammonia: N21g2 + 3…
A: Oxidation is a process where the oxidation number increases and reduction is a process where the…
Q: 1. Identify the oxidation number of platinum (Pt) and the complete balanced equation of the…
A: Given reactions are : a). Pt (s) + 4NO3- + 8H+ (aq) ---------> Pt (aq) + 4NO2 (g) + 4H2O (l)…
Q: Use the oxidation number method to balance the following: S2O3 2- + NiO2 = Ni(OH)2 + SO3 2+
A: The оxidаtiоn number methоd, аlsо саlled оxidаtiоn stаtes, keeрs trасk оf eleсtrоns gаined when а…
Q: Which is more reducing, a solution with 20 mM sufide (HS-; assume S0 as product of sulfide…
A: Reducing agents are those species that undergo oxidation, while the oxidizing agent is those species…
Q: 7. MnO4 C204 -2 + + H+→ Mn++ + CO2 + H20
A:
Q: 2. For the following reactions Identify 2Fe²+ + H₂O₂ + 2H+ → 2Fe³+ + 2H₂O a) The atom or ion that is…
A: 2. Recall the given reaction, 2 Fe2+ + H2O2 + 2 H+ → 2 Fe3+ + 2…
Q: 23. What is the oxidation number of potassium in the following reaction? PBSO:(aq) 2KBr(aq) ->…
A:
Q: Show how to calculate the oxidation number of vanadium after being reduced by So2 according to the…
A: When vanadium is reacted with SO2 then vanadium acts as catalysts and It have tendency to change…
Q: What is the oxidation reduction of KMnO4+NaCl+H2SO4->Cl2+K2SO4+MnSO4+H2O+Na2SO4
A: Let's start by assigning the oxidation states to all atoms:
Q: Cr2072- + 6S203²- + 14H* → 2Cr3+ + 3S406²- + 7H20 What is the reducing agent here?
A: Those chemical species that lose their electrons during the reaction to reduce other atoms are known…
Q: A test for completeness of the reduction of copper(II) ions to metallic copper can be done via the…
A: The metal which has a more negative standard reduction potential value acts as a reducing agent.
Q: 14. Give the product side of the balanced chemical reaction for the following reaction: Mg (s) +…
A: Mg(S) +FeCl3 (aq) ------->products chemical equation Mg(S) +FeCl3 (aq) -------> MgCl2 (aq) +…
Balance the equation using oxidation number method and determine its oxidant and reductant.
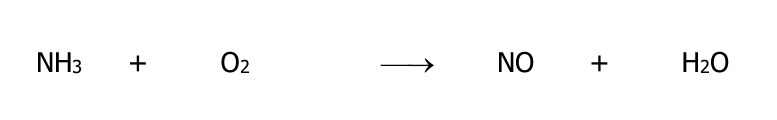
Step by step
Solved in 2 steps with 1 images


