Q: I. 1. 4. 7. Give the nomenclature or structure of the following molecules: SOH CH3 CH3 ГГ…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Why does methyl orange have a lower pKa value (pKa~3.5) than methyl red (pKa ~5.1)?
A: We are to discuss why pKa value of methyl orange is lower than that of methyl red.
Q: Is possible to make a buffer pH 4 at 1mM using methanesulfonic acid and sodium tartrate? Please…
A: To explain: Is it possible to make a buffer pH 4 at 1mM using methanesulfonic acid and sodium…
Q: Use the References to access important values if needed for this question. A 16.4 gram sample of…
A:
Q: ome N₂ gas is mixed with some O₂ gas, and the sketch below shows a representative sample of the…
A: Total pressure (P) = 0.060 Kpa
Q: Which of the following pieces of equipment delivers the most accurate volume of solution. A Buret B…
A: We are to discuss which of the given instruments can measure volume of solution most accurately.
Q: 7 11 8 9 11 11 12 13 14 15 16 17 18 ✓ ✓ 19 20 21 ✓ -- 22 23 24 25 26 27 10 < Light was measured to…
A:
Q: All polysaccharides consist of sugar monomers joined together by O a. ester linkages O b. amide…
A: Linkage between sugars in all polysaccharides.
Q: A 10.0 L tank at 0.6 °C is filled with 3.23 g of sulfur tetrafluoride gas and 17.3 g of dinitrogen…
A: Given: Volume of tank = 10.0 L Temperature = 0.6 oC Mass of sulfur hexafluoride i.e. SF6 = 3.23 g.…
Q: given a sample mass of 0.5074 g and with a
A: Mass of sample = 0.5074 g Mass of imurity = 0.2506 g Mass of Na2CO3 = 0.5074 - 0.2506 = 0.2568 g
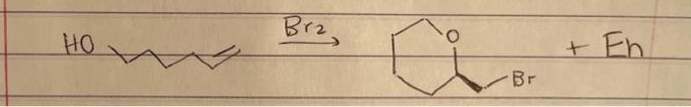
Q: 1. 0504 2. NaHSO3
A: OsO4 is used dihyroxylation of double bonds. Here methylcyclohexene reacts with OsO4 will gives…
Q: Match each molecule with a shape. H2S…
A: The shape of molecule is determined by the location of nuclei and it's electrons .the electron and…
Q: order these bond strenght ( 4 is the strongest bond, 1 is the weakest bond
A:
Q: Five liters of nitrogen gas initially at T = 27 degrees C and P = 1 atm are heated to T = 127…
A: Here 5 L of N2 gas initially at 27°C and P=1atm is heated to 127°C .We have to determine w, ∆E ,∆H…
Q: The segment -CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 - represents the polymer O a. polyhexene O b.…
A:
Q: Give the mechanism and Choose correct option. El H₂Soy heat
A: Note : tertiary alklyl halide produces tertiary carbocation. β-hydrogen is removed from carbocation…
Q: identify whether each species functions as a Bronsted - lowery and of Bronsted Lowary base in net…
A:
Q: What is the rate law for the following reaction, given the data below? 2NO + H2 → N2O…
A: The kinetics data given is,
Q: In the standardization of HCl solution, a pale pink end-point using phenolphthalein indicator was…
A:
Q: 2. (a) Give the reaction showing the dissociation of Zn3(PO4)2 in water. Write the reaction in the…
A: Given: Salt Zn3(PO4)2
Q: How many grams of Ag2CO3 will precipitate when excess (NH4)2CO3solution is added to 74.0 mL of 0.748…
A:
Q: What is the pH of a 3.1 x 10-4 M CsOH solution?
A: Given : concentration of CsOH = 3.1 × 10-4 M To calculate :- pH of solution
Q: Find the boiling point and freezing point of a solution resulting from the addition of 500 g of…
A:
Q: How many allylic hydrogen atoms are present in 1-ethylcyclohexene? 1 2 6
A: Given-> 1-ethylcyclohexene
Q: Enter your answer in the provided box. What is the emf of a cell consisting of a Pb2+ / Pb half-cell…
A:
Q: Oxidation half-reaction for 2 Cut (aq) + Mg(s) → Mg2+ (aq) + 2 Cu(s). Express your answer as a…
A:
Q: A salad dressing recipe calls for 2 parts olive oil to be combined with 1 part vinegar. a) A chef…
A:
Q: Which is the equilibrium constant expression for the following reaction? Fe₂O3(s) + 3CO(g) = 2Fe(s)…
A: Equilibrium constant is the ratio between concentration of products and the concentration of…
Q: Write the equilibrium expression for each of the following reactions. a. 203(g)=302(g)
A: Equilibrium constant is the ratio of product of concentration of products raised to their…
Q: What is the percent concentration by volume of wine made by mixing 37 mL ethyl alcohol and 120 mL…
A: All the necessary calculations have been done in the following step.
Q: Find the ΔH for the reaction below, given the following reactions and subsequent ΔH values: N2(g)…
A:
Q: What is observed with the values of Ksp?
A: The data given is,
Q: Glycolysis is the first step in the oxidation of fatty acids to produce energy. A) True B) False
A: Oxidation is a process in which there is addition of oxygen to a molecule or there is removal of…
Q: Reduction occurs at the ______ in a voltaic cell and reduction occurs at the O a. anode; anode O b.…
A: Reduction occur at cathode in both voltaic and electrolytic cell
Q: 14. Calculate and compare [H₂O"] and pH for a 0.10 M solution of HCI (a strong acid) and a 0.10 M…
A: Acid are compound which releases hydronium ion when dissolve in water We are required to compare…
Q: How much heat is needed to convert 766.g of ice at -10.0°C to steam at 126.0°C? Round your answer to…
A:
Q: An allylic free radical halogenation occurs to give the major product shown. Note NBS is a source of…
A:
Q: Is possible to make a buffer pH 4 at 1mM using methanesulfonic acid and sodium tartrate? Please…
A: To explain: Is it possible to make a buffer pH 4 at 1mM using methanesulfonic acid and sodium…
Q: synthetic route + O
A: Reaction 1 : Acetylene on reaction with NaNH2 and methyl iodide produces methyl acetylene. Methyl…
Q: 2NaOH (aq) + CuSO4 (aq) ---> Cu(OH)2(s) + Na2SO4(aq) How many grams of copper II sulfate are…
A:
Q: Backboard 00002A QUESTION 19 In the equation ke O True O False QUESTION 20 HF is a weak acid.…
A:
Q: SO₂(g) + NO₂(g) = SO3(g) + NO(g) If all four gases had initial concentrations of 0.650 M, calculate…
A: Given : Initial concentration = 0.650 M
Q: Calculate AG and the equilibrium constant for the following reaction at 298 K: Zn²+ (aq) + Ni(s) →…
A:
Q: Calculate the pH and the Ka for HC₂H3O2 if in a 1.0000 M solution of HC₂H3O2 in water there is…
A:
Q: For which of the following reactions is AS° > 0. Choose all that apply. 2H₂O(g) + 2SO₂(g) → 2H₂S(g)…
A:
Q: A Chemistry Student Trainee at a certain University Chemical Laboratory was instructed by the…
A: Given: Normality of NaOH = 0.25 N And the volume of NaOH solution should not exceed 40.0 mL i.e.…
Q: rue or False, formation of the major product has a lower activation energy in the rate determining…
A: 1) Formation of the major product has a lower activation energy in the rate determining step than…
Q: Consider the reaction. N₂(g) + 20₂(g) 2NO₂(g) Using standard thermodynamic data at 298K, calculate…
A: Given :- The balanced reaction, N2(g) + 2O2(g) → 2NO2(g) .... (A) Moles of N2 = 2.46 mol…
Q: Check the option(s) in which a chemical reaction is spontaneous (AS univ> 0) only below a certain…
A: Spontaneous processes are those processes which take place on their own without providing any energy…
Q: What is the mole percent of ethanol (C2H5OH) in the 180 proof vodka, which consists of 71.0 g of…
A:
Step by step
Solved in 2 steps with 2 images


