Q: A flask has a total pressure of 6.8 atm. The partial pressure of Ne is 450 torr, the partial…
A:
Q: Calcium hydroxide will precipitate from solution by the following equilibrium: 2+ Ca²+ (aq) +…
A:
Q: What is the major product of this reaction? O H HCN ???
A:
Q: Which of the two setups below is more appropriate for the separation of two liquids in the…
A: Separating funnel is used to separate immiscible liquids , when these tere separatory immiscible…
Q: For the reaction Fe(s) + 2HCl(aq) → FeCl2 (s) + H₂ (9) AH° -7.40 kJ and AS = 108 J/K The equilibrium…
A:
Q: Calculate the change in energy (in Joules) when an electron transition from n=4 to n=2 in a hydrogen…
A: The electronic energy levels in an atom are quantized and when an electron undergoes a transition…
Q: Air is approximately 80 mol% Nitrogen (M = 28kg/kmol) and 20 mol% Oxygen (M = 32kg/kmol). Consider a…
A: It is given that before the addition of hydrogen, pressure in container = 2 atm After addition of…
Q: In a steady-state approximation, it is assumed that O the concentration of the reaction…
A: •Steady state of approximation is related to derivation of rate equation for a reaction. • In steady…
Q: The following equilibrium is established in a closed container: C(s) + O2(g) CO₂(g) AH = -393 kJ =…
A: Given reaction is : C (s) + O2 (g) <-----------> CO2 (g) Standard change in enthalpy of the…
Q: 15) The positively polarized carbon atom of a carbonyl group acts as: A) an electrophile and a Lewis…
A: we have to identify the correct statement for the carbon atom of carbonyl group
Q: 2) Provide the structure of the major organic product of the following reaction. O HN C₂H5COCI AIC13
A:
Q: Variation of the rate constant with temperature for the first-order reaction is given in the…
A:
Q: Consider the following system at equilibrium where AH = -108 kJ and Kc = 77.5, at 600. K. CO(g) +…
A: As per Le Chatelier's principle, any change during a chemical reaction causes the reaction to flow…
Q: List the molecules in decrease strength of intermolecular forces. (So the strongest intermolecular…
A: we have to put the given molecules in order of decreasing intermolecular force strength
Q: Calculate the solubility at 25 °C of BaCrO4 in pure water and in a 0.0130M BaCl₂ solution. You'll…
A: Given, Concentration of BaCl2 = 0.0130 M (a). Calculate the solubility of BaCrO4 in pure water in…
Q: 1) Describe a simple chemical test to differentiate the following pairs of compounds. Write the…
A: #1: (i): 2-propanone and propanoic acid: 2-propanone(CH3COCH3) has terminal -COCH3 group that gives…
Q: What is one quantitative fact about the element of Sulfur with proper significant digits and units?
A: Please find your solution below : An element in chemistry is defined as a substance which cannot be…
Q: The coefficient of fugacity of percent gas at 290 K and 2.1 MPa is 0.68. Calculate the molar Gibbs…
A:
Q: The enthalpy change for a certain reaction is -209.4 kJ and the entropy change is 585.7 J/K. What…
A:
Q: 4) Rank the following sigma complexes in order of increasing stability. NO₂ A + H H3CO. Br B H -Br H…
A: Answer of this question :- Sigma complex :- Sigma complex refers to an arenium ion. The suffix…
Q: 14) The intensity of a signal in a ¹H NMR spectrum is determined by A. The number of neighboring C.…
A: In the given question we have to select the correct option from the following. the intensity of…
Q: At what temperature is a reaction spontaneous if it has; ΔH = +71 kJ/mol and ΔS = +167.2 J/Kmol.…
A: We have to calculate the temperature for which the reaction will be spontaneous
Q: How many acidic hydrogen atoms and how many nonacidic hydrogen atoms are present in each of the…
A: Acidic Hydrogens are those which can get ionised but non acidic hydrogens don't get ionised.
Q: pH=pKa if the pH of the buffer solution contains equal concentrations of a weak acid and its…
A: A buffer solution contains a weak acid and its conjugate base. The pH of a buffer solution can be…
Q: The 0-16 nucleus has a mass of 15.9905 amu. A proton has a mass of 1.00728 amu, a neutron has a mass…
A:
Q: Suggest starting materials for the formation of the following aldol products: (a) (b) O I `H H
A: Aldol condensation: The alpha hydrogen of carbonyl compounds is acidic in nature and it is removed…
Q: (9) Energy and electron transfer processes in a supramolecular system A-L-B, where A is the light…
A: when molecule A absorbs the light it got excited and give electron to B , B emits the light and act…
Q: What is the general class of reaction of potassium cyanide? How much heat must be added or removed…
A:
Q: 2) How many nodes, other than the node coincident with the molecular plane, are present in the 3 of…
A: Annulene: Annulenes are monocyclic hydrocarbons that contain the maximum number conjugated double…
Q: CO₂(g) + 2H₂O(g) → CH₂(g) +20₂ (g) C6H₁₂O6 (s)→ 6C(s) + 6H₂(g) + 30₂ (g) An AS = 2766. AG = KJ Which…
A:
Q: 2. Write the neutralization reaction between Ca(OH)₂ and HCI. Include both the molecular reaction,…
A: Neutralisation between CA(OH)2 & HCl.
Q: The balanced chemical equation for the combustion of propane gas is: C3H8 (g) + 5 O2 (g) --> 3 CO2…
A:
Q: The very high melting point and boiling point of ionic compounds such as NaCl is due to the very…
A: • We need to determine which intermolecular force of attraction exist in ionic compounds.
Q: The major product that would form during the alkylation reaction shown below is? Br. A. B. OC. D.…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: Which statement best describes the method you would to isolate compound 2 from the mixture of…
A: Compound 1 is an ester and is soluble in ether only. Compound 2 is a carboxylic acid and is soluble…
Q: Chlorineoxide (ClO), which plays an important role in the depletion of ozone decays rapidly at room…
A: We can find the order of the reaction by the graphical method of order determination. If ln[A] Vs t…
Q: Calculate the entropy change (∆SRXN) in J/K that occurs when one mole of solid iron (III) oxide is…
A: Given, The unbalanced chemical equation for the process is: Fe (s) + H2O (g) --> Fe2O3 (s) + H2…
Q: c. Ba3(PO4)2, barium phosphate, 3.9 x 107 g/L Ksp d. Ag₂ SO4, silver sulfate, 5.1 mg/mL Ksp
A:
Q: Question: Use the solubility chart. What is the precipitate when 4.9 grams of solute are used to…
A: In the given question we have to select one which get precipitated after addition. When NaF solute…
Q: Q3/ Answer with the word true or false for each of the following: () -1 1- The appearance of the…
A: In the given problem we have to address whether the given statements are correct and or not and…
Q: 31. Draw the curved arrow mechanism for the formation of the enolate formed when a ketone reacts…
A: In a ketone, the H attached to the α-carbon atoms are the most acidic. NaOH or OH- being a strong…
Q: Which of the following would you expect to have the highest boiling point? Xe Kr He Ne O Ar Kr O Ne…
A: Please find the attachment.
Q: In the following molecule, which bond(s) is (are) a peptide bond? Mark all that apply. DA ОВ BC O D…
A: The peptid bond is an amide type bond which is formed by the condensation of two amino acids through…
Q: how will you detect the presence of citrate ion using carboxyflorocein ?
A:
Q: What is the molar solubility of barium bromate at 25°C? The formula for this ionic salt is…
A:
Q: . What sequence of reagents will accomplish the following transformation? O 1. 2. 3. HCI H₂O H₂O NH₂…
A: Introduction :- In organic chemistry we can convert one compound into another by using suitable…
Q: Ammonium sulfate fertilizer is manufactured by reacting sulfuric acid with ammonia. In the…
A:
Q: Consider the following reaction and the following conditions: Fe₂O3(s) + 3 CO(g) → 2 Fe(s) + 3…
A: Given -> Fe2O3(s) + 3CO(g) <-----> 2Fe(s) + 3CO2(g) ∆G°= -2.8 × 104 J P(CO) = 1.4 atm ,…
Q: How many moles of iron metal are required to react completely with with 597 mL of 1.49 mol/L…
A: Given - Volume of copper(II) sulfate = 597 mL Molarity of copper(II) sulfate = 1.49…
Q: pH 14 12 10 ∞ 6 4 2. 0 0 25 50 Volume of NaOH added (mL)
A: The given titration curve has 1 point (at V = 25 mL) where the pH change is maximum. Hence the…
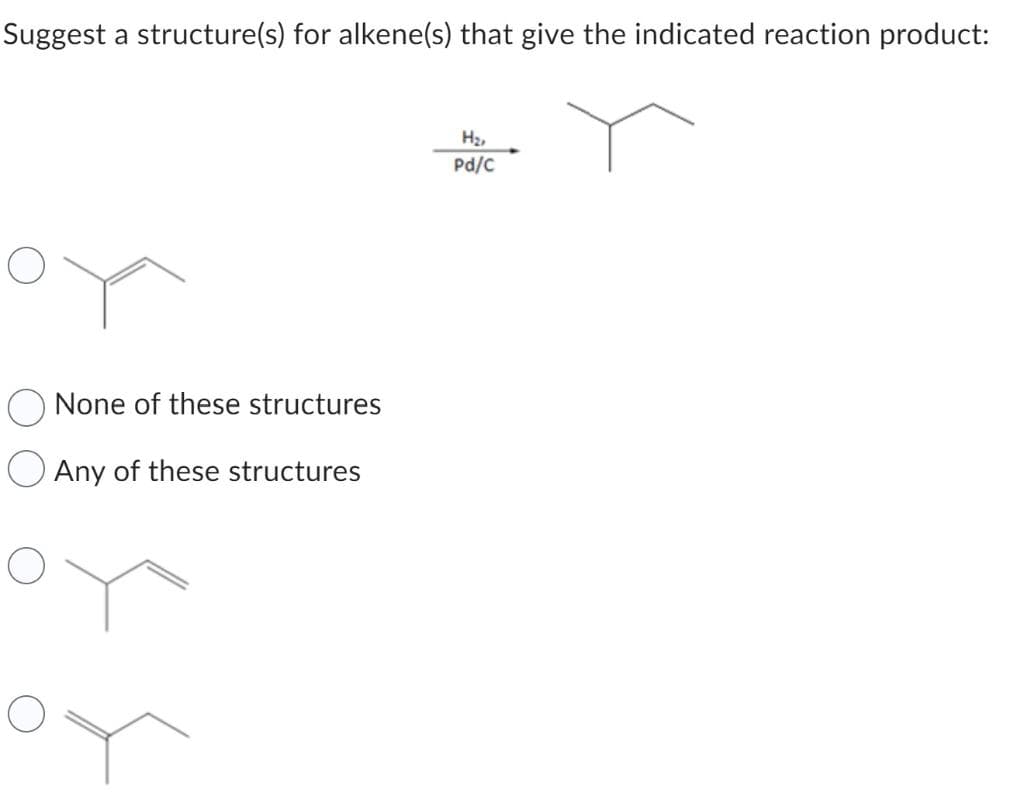
Step by step
Solved in 2 steps with 1 images

- Suggest structures for alkenes that give the following reaction products. There may be more than one answer for some cases.Name the following alkene and what will be the reaction below with KMnO4 in aqueous acid.Treatment of propylbenzene with NBS + hv affords a single constitutional isomer. Suggest a structure for the product and a reason for its formation.
- Reaction of -pinene with borane followed by treatment of the resulting trialkylborane with alkaline hydrogen peroxide gives the following alcohol. Of the four possible cis,trans isomers, one is formed in over 85% yield. (a) Draw structural formulas for the four possible cis,trans isomers of the bicyclic alcohol. (b) Which is the structure of the isomer formed in 85% yield? How do you account for its formation? Create a model to help you make this prediction.Suggest a strategy for converting 1-methylcyclohexene into 1-cyclohexylmethanol. Provide the structures for the major organic products frormed in each step.Introduce the reaction and structure of the expected products (indicate the main product, when possible) for a reaction of 3-ethyl-5-methyl-hex-2-ene with the following reagents: (a) Br2, CH2Cl2 (b) H2O , Diluted H2SO4.
- Birch reduction of 2-methoxynaphthalene gave a mixture of two isomeric compounds, each having the molecular formula C11H14O. Suggest reasonable structures for these compounds.Using benzene and any necessary organic or inorganic reagents, suggest efficient syntheses of (a) Isobutylbenzene, C6H5CH2CH(CH3)2 (b) (2,2-Dimethylpropyl)benzene, C6H5CH2C(CH3)3Provide the E2 mechanism for β-elimination reactionemploying 2-chloro-2-methylbutane to prepare 2-methyl-2-butene and 2-methyl-1-butene reaction. Use the actual structures of thereactants and products. Explain which of the alkenes is the major product ofthis reaction ?
- Help with the following ochem reaction scheme... Consider 3,4-dimethylpiperidine being subjected to the below reaction scheme: step 1) CH3I (excess) step 2) NaOH, heat step 3) CH3I (excess) step 4) NaOH, heat Provide the bond line structures for the major organic product obtained in each step and then discuss the regiochemistry for step 2.Predict the organic product(s) of the reaction of 2-butene with each reagent. Q.)Br2 in H2OAn unknown hydrocarbon decolorizes bromine in CCl4CCl4, and it undergoes catalytic reduction to give cis-decalin. When treated with warm, concentrated potassium permanganate, this compound gives cis-1,2-cyclohexanedicarboxylic acid. Propose a structure for the unknown compound.

