Chapter7: Solutions And Colloids
Section: Chapter Questions
Problem 7.86E
Related questions
Question
Please solve the normality of the sample in eq/L and the (DO) content of the sample in mg/L using the following equation.

Transcribed Image Text:Trial (Based on the Hypothetical Data 1
youtube link)
20 mL
Parameters
Hypothetical
Data 2
Volume of Sample
10 mL
15 mL
Volume of 0.025 N
1.4 mL
0.60 mL
1.0 mL
Na:S.Osused
Normality of
Sample
Dissolved Oxygen
Content of the
Sample
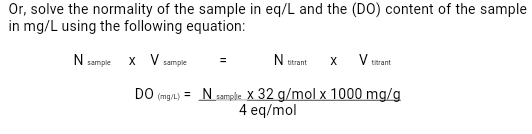
Transcribed Image Text:Or, solve the normality of the sample in eq/L and the (DO) content of the sample
in mg/L using the following equation:
N utrant
V.
X
x V sample
titrant
N sample
DO (mg/) = Nsamphe. X 32 g/mol x 1000 mg/g
4 eq/mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



