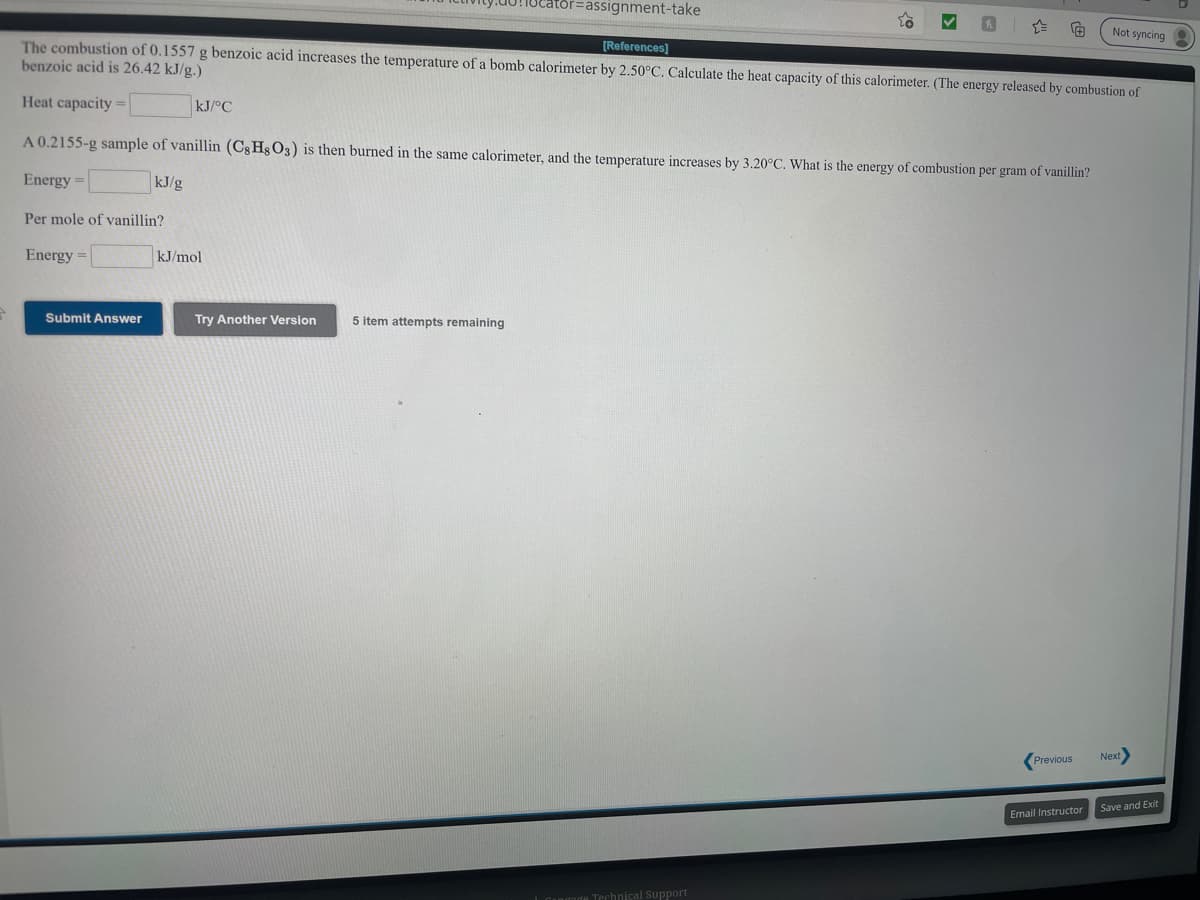
Not syncing [References) The combustion of 0.1557 g benzoic acid increases the temperature of a bomb calorimeter by 2.50°C. Calculate the heat capacity of this calorimeter. (The energy released by combustion of benzoic acid is 26.42 kJ/g.) Heat capacity = kJ/°C A 0.2155-g sample of vanillin (CgHs O3) is then burned in the same calorimeter, and the temperature increases by 3.20°C. What is the energy of combustion per gram of vanillin? Energy = kJ/g Per mole of vanillin? Energy = kJ/mol
Not syncing [References) The combustion of 0.1557 g benzoic acid increases the temperature of a bomb calorimeter by 2.50°C. Calculate the heat capacity of this calorimeter. (The energy released by combustion of benzoic acid is 26.42 kJ/g.) Heat capacity = kJ/°C A 0.2155-g sample of vanillin (CgHs O3) is then burned in the same calorimeter, and the temperature increases by 3.20°C. What is the energy of combustion per gram of vanillin? Energy = kJ/g Per mole of vanillin? Energy = kJ/mol
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter5: Thermochemistry
Section: Chapter Questions
Problem 51E: A sample of 0.562 g of carbon is burned in oxygen in a bomb calorimeter, producing carbon dioxide....
Related questions
Question
Transcribed Image Text:ator%=Dassignment-take
Not syncing
The combustion of 0.1557 g benzoic acid increases the temperature of a bomb calorimeter by 2.50°C. Calculate the heat capacity of this calorimeter. (The energy released by combustion of
benzoic acid is 26.42 kJ/g.)
[References)
Heat capacity =
kJ/°C
A 0.2155-g sample of vanillin (Cg Hg O3) is then burned in the same calorimeter, and the temperature increases by 3.20°C. What is the energy of combustion per gram of vanillin?
Energy =
kJ/g
Per mole of vanillin?
Energy =
kJ/mol
Submit Answer
Try Another Version
5 item attempts remaining
(Previous
Next
Save and Exit
Email Instructor
Technical Support
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning