Number of Inotope Name Owimass number) Total Electric Number of Neutrons Atomic Number of Number Protons Charge Electrons Zine66 +2 343 92 Plhie 9 21
Number of Inotope Name Owimass number) Total Electric Number of Neutrons Atomic Number of Number Protons Charge Electrons Zine66 +2 343 92 Plhie 9 21
Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter10: The Mole
Section10.5: Formulas Of Hydrates
Problem 78SSC
Related questions
Question
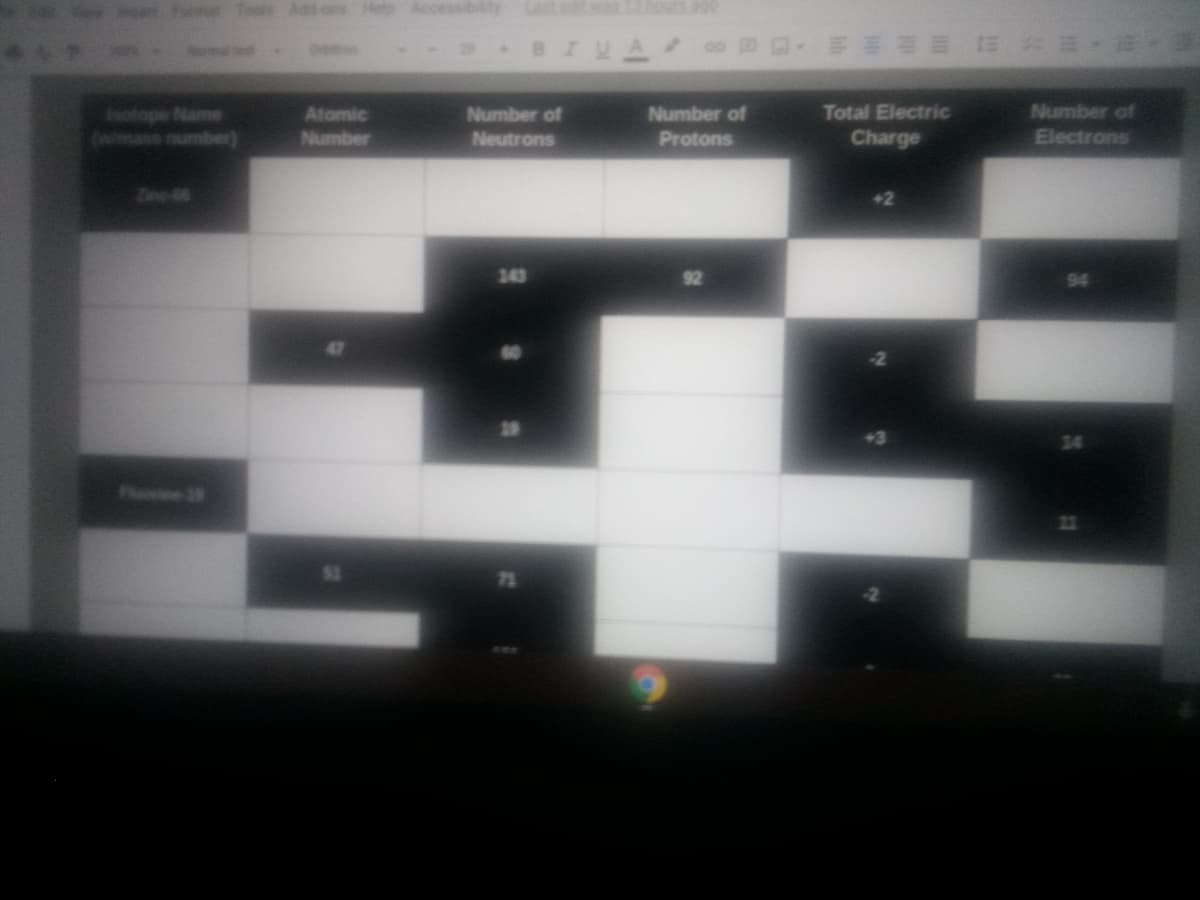
Transcribed Image Text:Tools Ad-ons Hetp
ccessibility
BIUA
Dロ- F mE
Total Electric
Number of
inotope Name
(qun s )
Atomic
Number of
Neutrons
Number of
Number
Protons
Charge
Electrons
Zine 6
+2
143
92
Plosine 2
21
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning