Q: When calibrating a pH meter, 3 (three) standard color-coded buffer solutions are used, which of thes...
A: The correct answer about PH calibration is given below
Q: A certain reaction is second order in N, and first order in H,. Use this information to complete the...
A: Given Order of reaction with respect to N2 = 2 Order of reaction with respect to H2 = 1 [ N2 ...
Q: Write the balanced NET IONIC equation for the reaction that occurs when hydroiodic acid and potassiu...
A:
Q: What is the volume of the following sample of gas at STP? 1.05 mol C2N2
A:
Q: Which Newman projection is generally more stable? A Staggered B Eclipsed
A: Stereochemistry is branch of chemistry which deals with the spatial arrangement of atoms in molecule...
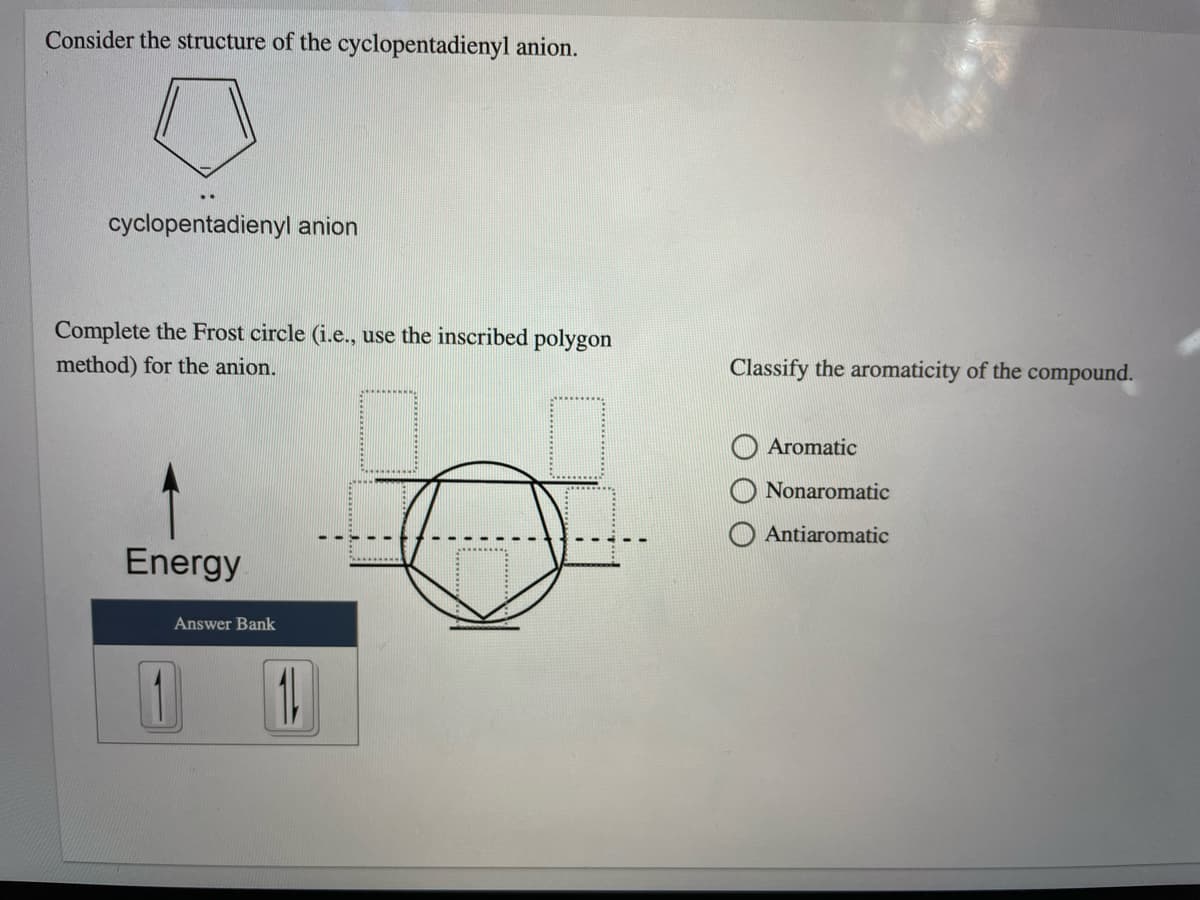
Q: Indicate whether each structure is aromatic, nonaromatic, or antiaromatic. Assume planarity. C BH Co...
A:
Q: Some measurements of the initial rate of a certain reaction are given in the table below. [N:] H2|in...
A:
Q: Under certain conditions the rate of this reaction is zero order in dinitrogen monoxide with a rate ...
A:
Q: The freezing point of pure benzene was measured as 5.49 °C. The freezing point of a solution prepare...
A:
Q: Some measurements of the initial rate of a certain reaction are given in the table below. [H.] 12 in...
A: Rate constant is proportionality constant of rate law expression, rate law is experimental analysis ...
Q: Balance the following equation with the smallest whole number coefficients. What is the coefficient ...
A: The balanced equation and coefficient of Oxygen in balanced equation is given below
Q: Draw the structure of the following compounds. Use line-angle structural formula. 1-bromo-4-ethyl-5-...
A:
Q: 19. Provide a complete curved arrow mechanisms that accounts for the transformation below. Be sure t...
A: At first double bond take H+,possible formation of two carbocation one is 2 degree and another is 1 ...
Q: Q8: Below is a unit cell for a hypothetical metal. (a) To which crystal system does this unit cell b...
A: The crystal systems are defined based on the length of the cell, and bond angle between cell lengths...
Q: volume
A:
Q: At a certain temperature this reaction follows first-order kinetics with a rate constant of 8.78 × 1...
A:
Q: Hl ОН The structure shows which configuration? AR B) s
A:
Q: Explain using the ideas of bases, acids, nucleophiles and electrophiles why this reaction wouldnt wo...
A: This reaction does not takes place without catalyst. Catalyst catalyses the reaction.
Q: y MOLECULES of molocules
A:
Q: How many grams of Xe are there in a sample of Xe that contains 8.97×1023 atoms?
A: First we have to determine the moles of xenon.
Q: describe the elements that can have more than an octet. Briefly describe how you can tell how many c...
A:
Q: Type of delocalization that occurs in a conjugated system? A pi electron delocalization B lone pair ...
A:
Q: 3. Draw a Fischer projection of: a) (S) 3-iodo-1-butene
A: for determine the R/S configuration arrange the atoms by using CIP rule that is the numbering the at...
Q: t a certain temperature this reaction follows second-order kinetics with a rate constant of 1.47M CI...
A: Given Reaction followed second order kinetics
Q: 4. Justify the following statement: (i) The symmetric H-bonds are stronger than asymmetric ones
A: Symmetric hydrogen bonds are stronger than assymetric hydrogen bonds. The reason is as given below.
Q: isomers.
A:
Q: 3. 4. 500 mL of 0.75 N of K2Cr207 from a pure solid reagent if it follows the following half reactio...
A: In this type of question first thing should be balance the half reaction. Cr2O72- ...........> Cr...
Q: Give the conjugate base of H2O.
A: Acid is substance which release hydrogen ions and base is substance which release hydroxyl ions when...
Q: HO, HO Refer to the given structure, the -OH substitutions are in, position of each other. A trans B...
A:
Q: 21. Based on your knowledge concerning halogenation of alkenes, provide a step-wise curved arrow mec...
A:
Q: Read carefully the given partial names in IUPAC alkane nomenclature and check the columns where they...
A:
Q: Some measurements of the initial rate of a certain reaction are given in the table below. H2 2 initi...
A: The sum of the exponent term in the rate law for a chemical reaction is known as the order of the re...
Q: Why cannot oils and water-soluble products be mixed and heated together? Why should heating be at a...
A: Oils and fats are non-polar compound (hydrocarbon) and water is non-polar compound and water molecul...
Q: Directions: Complete the table below about the Kinetic Molecular Theory of Liquid and Solid. Descrip...
A: Please find your solution below : According to Kinetic Molecular Theory, the matter consist of parti...
Q: Calculate the concentration equilibrium constant, K’sp for La(IO3)3 in a saturated solution of La(IO...
A: For a sparingly soluble salt, there is a dynamic equilibrium between the dissociated ions and undiss...
Q: 3. Propose an efficient synthesis to get the final product using malonic ester Hint: 6 steps to get ...
A: Solution If effcient synthesis to get the final product using malonic Ester..
Q: A certain element consists of two stable isotopes. The first has a mass of 68.9 amu and a percent na...
A:
Q: 11:36 O ... 66 X How many grams of Au are in ../1 0.053 mol of Au? 1 X How many Ga atoms are in ./1 ...
A: We know that, Number of moles = mass/molar mass
Q: 5. Explain the trend in binding constants (in CH,Cl2) for the complexation of phenol derivatives by ...
A: Solution -
Q: Using a curved fish hook arrow, propose a mechanism for the reaction: a) CH3 H3C C=CH2 H3C HBr CH-C-...
A:
Q: 1. How many MOLES of dinitrogen tetroxide are present in 4.67 grams of this compound ? moles. 2. How...
A: Mole of substance is defined as given mass divided by its molar mass.
Q: 3 4) il a Pump is used to gas at Volume of 1.50L Rapidly expand 25°C from at 500 bar to a 9, 9. Volu...
A:
Q: CHO Ethanol `NH; OH OH но Salicylaldehyde 16lesanediamine NN-Disalicylaldehydehy de-1,6-hexanediimin...
A: The theoretical yield and percent yield can be calculated as
Q: Under certain conditions the rate of this reaction is zero order in dinitrogen monoxide with a rate ...
A: The integrated rate equation for a zero order reaction is, [ C ]t = -kt + [ C ]0 [ C ]t = Co...
Q: Read carefully the given partial names in IUPAC alkane nomenclature and check the columns where they...
A:
Q: A certain reaction is second order in N, and first order in H,. Use this information to complete the...
A:
Q: A student heats a 5.0 g sample of an unknown metal to a temperature of 207∘C, and then drops the sam...
A:
Q: sa dipole moment?
A:
Q: A student carried out an experiment and used 20.00 mL of 0.0100 M NaOH to titrate the excess HCl in ...
A: We are given an amount of NaOH being used to titrate the acid in an antacid reaction. We have to fin...
Q: 4
A: Given :- Cu-Ni alloy contains 64 wt% Cu and 36 wt% Ni. wt% of Cu in liquid is 54% and in solid is 3...
Step by step
Solved in 2 steps with 2 images

- Which of the following are consistent with the requirements for aromaticity?I. A system with delocalized p electrons in a ring.II. 4n p electrons in the ring.III. All the ring atoms must be carbons.IV. (4n + 2) p electrons in the ring. Give the answer why.what aromatic annulene has the same number of bond molecular orbital as the hypothetical all cis (20) annuleneAromatic compounds also known as arenes or aromatic are chemical compounds that contain conjugated planar ring system with delocalized pi electron clouds instead of discrete alternating single and double bond. One of the precursor to produce aromatic compounds is arenediazonium.Critize this statement
- Aromatic heterocyclic chemistry: State whether the following drugs are aromatic heterocycles and if they follow the 4 aromaticity rules (unsaturation, (4n + 2)pi electrons, cyclic and flat/planar), NAME ANY HETEROCYCLES. Draw the structure of the core of any aromatic heterocycles in the drug. Riociguat, Imatinib, Clopidogrel, Glatiramer acetate, AxitinibWhich of the following statements is FALSE? Select one: a. Aromatics are compound that are conjugated. b. The Cs and Hs in an aromatic compound are equivalent. c. The pi electrons are delocalized around an aromatic ring. d. None of the statements is false.Use the inscribed polygon method to show the pattern of molecularorbitals in cyclooctatetraene. Indicate the arrangement of electrons in these orbitals forcyclooctatetraene, and explain why cyclooctatetraene is not aromatic.
- 5Thiamin, or vitamin B1, contains a positively charged five-memberednitrogen–sulfur heterocycle called a thiazolium ring. Explain why the thiazoliumring is aromatic.Use the inscribed polygon method to show the pattern of molecular orbitals in cyclonona-1,3,5,7-tetraene, and use it to label its cation, radical, and anion as aromatic, antiaromatic, or not aromatic.Is this molecule aromatic? Why doesn't the pi bond in the benzene ring on the left side not count torward aromaticity?
- Use the inscribed polygon method to show the pattern of molecularorbitals in cyclonona-1,3,5,7-tetraene, and use it to label its cation,radical, and anion as aromatic, antiaromatic, or not aromatic.Biphenyl has the following structure.(a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon?(b) How many pi electrons are there in the two aromatic rings of biphenyl? How does this number compare with that for naphthalene?(c) The heat of hydrogenation for biphenyl is about 418 kJ>mol (100 kcal>mol). Calculate theresonance energy of biphenyl.(d) Compare the resonance energy of biphenyl with that of naphthalene and with that of two benzene rings. Explain thedifference in the resonance energies of naphthalene and biphenyl.For heterocycles containing nitrogen atoms, determine whether nitrogen’s lone pairs are used in the aromatic system, and predict whether the nitrogen atom is strongly or weakly basic.

