Q: onsider the following equi;ibrium: 3 HClO3 (aq) ⇌ HClO (aq) + 2 HClO4 (aq) If 0.15 M HClO and…
A:
Q: Write the cell notation for an electrochemical cell consisting of an anode where Al (s) is oxidized…
A: Here we are required to find the cell notation for an electrochemical cell consisting of anode where…
Q: A buffer solution is prepared by adding NH4Cl to a solution of NH3 (ammonia). NH3(aq) + H₂O(1) =…
A: Given : NH3 (aq) + H2O(l) ----------->NH4+ (aq) + OH-(aq) To find : find the effect of addition…
Q: aromatic anti-aromatic not aromatic aromatic anti-aromatic not aromatic aromatic anti-aromatic not…
A: Here we need to predict whether the compound given is aromatic, anti aromatic or non aromatic. Four…
Q: The equilibrium constant for the following reaction is 260 at -1 °C. 2 NO(g) + Br₂(g) 2 NOBr(g) K…
A:
Q: 1. What is the wavelength of the radiation absorbed when an electron undergoes a transition from n =…
A:
Q: Show by calculations whether these are good data. Compute the minimum work to separate (at 20°C) a…
A: Mass percent - It is defined as the ratio of the mass of each element to the total mass of the…
Q: 4 C following Friedel-Crafts ia B OMe A What is the product of the reaction? ( H AICI, OMe to OMe…
A:
Q: According to the Aufbau Principle, which of the following energy sublevels will be filled with…
A: Since, Electronic configuration represent the electron present in orbital in which way. Arrangement…
Q: CH3 1. HNO3/H₂SO4 2. Sn, HCI 3. H₂SO4 요. H3C O 4. Br2 CH3
A: This problem is based on basic organic reaction mechanism. The reaction products are given below…
Q: 27. Complete the following table, including all math work relating to the development of the Lewis…
A: Intermolecular Forces of attraction are the forces held between two molecules.
Q: Draw the spectrum of 4-bromophenyl ethyl ether respecting the multiplicity, the chemical shifts, the…
A: There will be 4 signal Multiplicity will be (n+1) Delta value will be like 3H= 0.9-14 ppm triplet…
Q: Question 9 of 24 > Identify the detectors in gas chromatography that can be used for the detection…
A: A gas chromatograph (GC) is a piece of analytical equipment that counts the number of different…
Q: Select the major organic product of the following organic reaction! N-H Z-H HCI
A:
Q: 200.0 mL of a 3.50 mol/L solution of potassium chloride is added to a concentrated lead (II) acetate…
A: Given, The concentration ( molarity) of the potassium chloride = KCl = 3.50 mol/L The volume of the…
Q: Which of the following statements about the hydrogen atom is FALSE. (A) The hydrogen atom is…
A: The given statements are, A) Hydrogen atom is neutral B) Hydrogen atom has uniform charge…
Q: :CI_O_N_O: -N- :Ö: :0:
A: Chlorine nitrate is polar in nature. BrCl3 is also polar in nature and T-shape geometry.
Q: When the Pb2+ concentration is 5.46×10-4 M, the observed cell potential at 298K for an…
A:
Q: 5. Consider a copper concentration cell. One half of the cell has 1.00 M CUNO3 and the other…
A: According to the question, The concentration of the CuNO3 half cell = 1.00 M The cell potential =…
Q: How many grams of NaOH are needed to prepare 45.0 ml of a 6.00 Solution?
A:
Q: Be sure to answer all parts. Write the formula for the conjugate base of each of the following…
A: According to Bronsted and Lowry concept an Acid is a substance that has the tendency to donate a…
Q: Which of the following is (are) the likely major product(s) of the reaction shown? Br A) I B) II C)…
A: Step by step mechanism.
Q: Question 7 Name the following: A B Include E/Z A = Blank 1 B = Blank 2 C = Blank 3 D = Blank 4 Blank…
A: Name : A). 3,5-diiodo hep-2-en-5-yne B). 3,7-dimethyl non-1,3, 6-ene. C). (Trans) 4,4-dimethyl…
Q: 1) A sealed flask contains 2.00 moles of helium and 3.00 moles of nitrogen. If the total pressure of…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 1) 03 2) Zn H₂O COOEt heat NaOEt EtOH Ph. Br₂ H₂O HCI, 1 eq -60°C NaN 3 DMF
A: Bromination on alkene is the anti addition reaction. Cis alkene give the recemic mixture whereas…
Q: What is the product of the following reaction sequence: KO CH₂CH3 1. NBS, (PhCO2)2 2. Mg, Et₂O…
A: N-bromosuccinamide is a bromonating agent used to allylic bromination of alkane. The reaction…
Q: Explain the working principle of following types of distillation, i. Simple Distillation; ii. Steam…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: 1.a) Name the following: (a) CH, — NH (b) C(CH3)3CH2-N-CH₂CH3 H (c) CH3-CH2-CH-CH3 (d) NH₂ CH3 N-CH3
A: 1) Since you have posted a question with multiple sub parts, we will provide the solution only to…
Q: Н3С. Н3С CH3 OH ~² `CI H3C OH Н3С XS (CHÍNH о о HBr CH₂
A:
Q: Match one of the given waves with each of the categories shown below. (Waves may be used more than…
A: Given waveforms -
Q: Measurements show that the energy of a mixture of gaseous reactants decreases by 153. kJ during a…
A: According to the first law of thermodynamics, the total heat change of a system at constant pressure…
Q: [www. The following equilibrium was studied by analyzing the equilibrium mixture for the amount of…
A:
Q: KOH(aq) + Fe(NO3)3(aq)
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: The following fictitious unbalanced REDOX reaction is made of some teachers initials and some oxygen…
A:
Q: 1. Write the main products of the synthesis reaction according to the specified raw materials,…
A: As per the rules, only three sub-parts can be answered. So the products for the reactions in…
Q: STEP 4: Balance the equation. Mission accomplished. Ex: Practice: 3 Li₂CO3(aq) + 2 Al(NO3)3(aq) →6…
A: Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: Select the true statements about protein secondary structure. In a B-pleated sheet, the side chains…
A:
Q: 1. The disintegration energy of the decay30 P15 by position emission is 4.30 Mev. The daughter…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Identify if the numbered compound functions as a bythen bes or neither. R R 1 O ? H-O-R R-O-H? 3 R R…
A: Hydrogen-bond is formed when H-atom is bonded to a highly electronegative atom like F,O,N. H-bond…
Q: A 100.0 mL sample of spring water was treated to convert any iron Fe2+. Addition of 25.00 mL of…
A:
Q: N₂(g) + 3H₂(g) → 2NH₂(g) - AH-92.6 kJ/mol IXB If 2.0 moles of N₂ react with 6.0 moles of H₂ to form…
A:
Q: 2Fe3+(aq) + Mn(s) 2Fe2+(aq) + Mn2+(aq) The anode reaction is: + + The…
A:
Q: 7. Calculate the pH of 0.10M Ammonia (NH3) Repeat the calculation after addition of 25 mL of 0.1M…
A: •Q.7)=> •Here initially only solution of ammonia is given.So firstly we solve pH for only ammonia…
Q: A saturated solution of iron(II) sulfide contains 2.50 x 108 M Fe2+ and 2.50 x 10-8 M S². Calculate…
A:
Q: Measurements show that the energy of a mixture of gaseous reactants decreases by 179. kJ during a…
A: We know that the thermodynamic equation H= U + PV ∆H. =∆(U + PV). ∆H. = ∆U +P∆V +V∆P But in the…
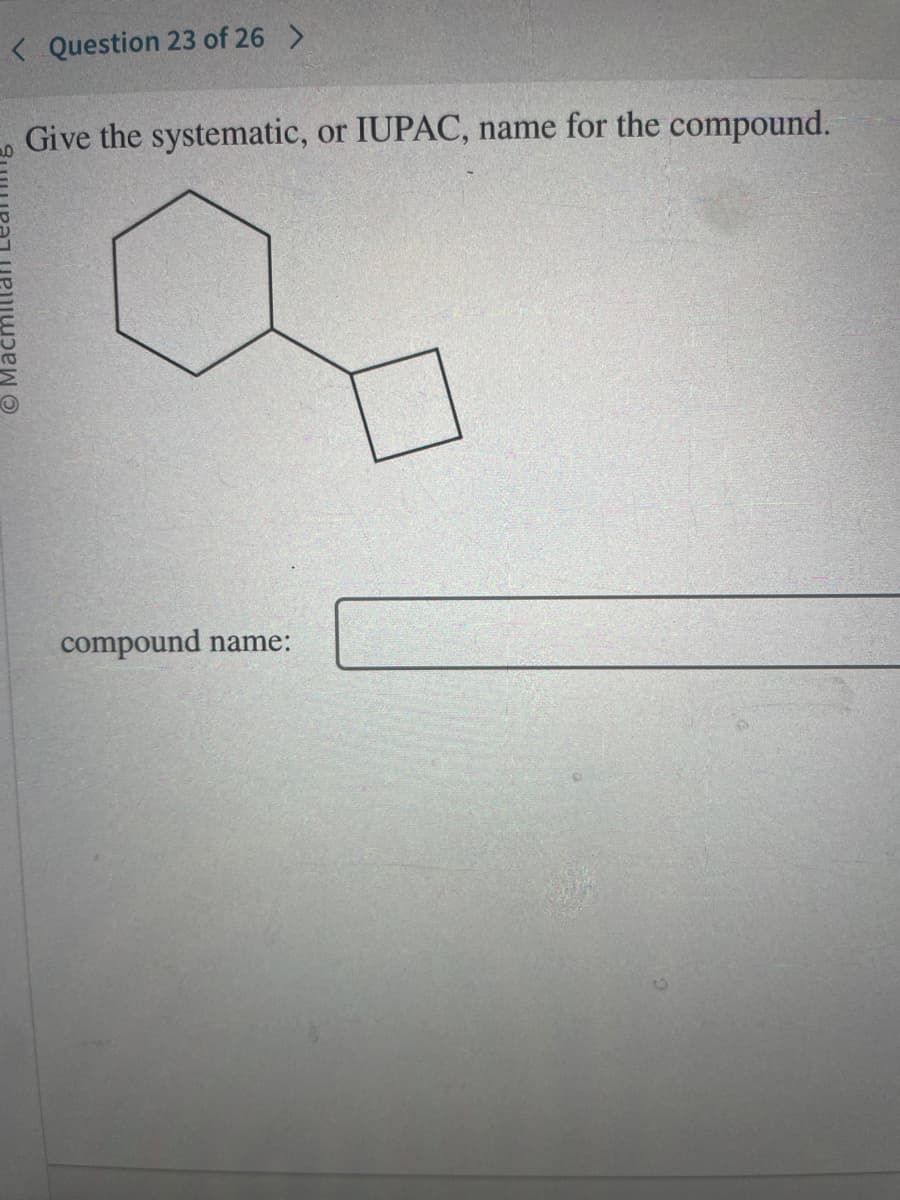
Q: Pls help ASAP. Pls provide all IUPAC names.
A: Ans :- 1) 2 Butanone 2) N octane 3) propyl hexanoate 4) propan-1-ene 5) Chlorobenzene 6) 3 methyl…
Q: Identify the amino acids make up the following peptide. Select all that apply. H. SH NH O Mijo NH SH…
A: The carboxylic acid group of one amino acid and the amine group of another amino acid undergo…
Q: Calculate the quantities of lime and soda required to soften 25000 liter of water having following…
A: According to the question, Ca(HCO3)2 = 4.86 ppm Mg(HCO3)2 = 7.3 ppmCaSO4 = 6.8 ppmMgSO4…
Q: How to identify bronsted Lowry acid from bronsted Lowry base? As well the conjugate Give an example.…
A: Acid base defined as Bronsted acid base pair based on they are able to accept and donate proton.
Q: Part II: i) Provide the structure for the product(s) that is(are) produced when these two alkenes…
A: Step by step each mechanism.
Step by step
Solved in 2 steps with 1 images


