Q: Draw in all the lo
A: Lone pair electrons are those which are not participates in the bonding therefore, these are also…
Q: PREDICT THE PRODUCT 1. BH3 2. H₂O₂, NaOH 3. PCC 4. PhMgBr 5. H3O+ workup 1. H₂O, H₂SO4 2. PCC 3.…
A: Reactions are involves anti Merkonikove addition of H2O, oxidation of alcohol to ketone or aldehyde,…
Q: Partial pressure is exhibited by one gas in a mixture. O True O False
A: True
Q: 47. The Daniel cell is (a) Pt₁ (s)| Zn(s)| Zn²+ (aq) || Cu²+ (aq) | Cu(s)|Pt₁ (s) (b) Pt₁ (s)| Zn…
A: Daniel cell consists of two half cells. The half cell on the left contains a zinc metal electrode…
Q: (a) addition polymerization (b) alternating copolymer (c) block copolymer (d) branched polymer (e)…
A: A polymer is a large molecule composed of repeating units, called monomers, connected by covalent…
Q: Draw an equivalent resonance structure that minimizes charge. Include all lone pairs in your…
A:
Q: c) NH3 Cl thermal initiator A polymerization
A: This is addition polymerization reaction. In this reaction addition of C=C bond occurs by free…
Q: How many grams are in 8.37x10^10 particles of Li2S04?
A: Given Number of particles of Li2SO4 = 8.37×1010
Q: An 18 carat white gold is made by dissolving 12.5188 g of Copper, 14.1431 g of Silver, and 63.5172 g…
A: The percent composition by mass of silver in metal alloy is = 3.86 %
Q: Identification of Hydrocarbons Pre-lab Questions 1. What changes in color occur when bromine or…
A: This question is based on practical test of hydrocarbon.
Q: What is the molar mass of an unknown gas with a density of 2.10 g/L at 1.00 atm and 25.0 °C?
A:
Q: Determine the pH of an aqueous solution of sulfuric acid at a concentration of 0.5 mol.L-¹. What is…
A: Concentration of sulfuric acid is 0.5 mol.L-1
Q: Stock hydrochloric acid (HCI) has a concentration of 12.1 M. What volume of concentrated HCI, in mL,…
A: Dilution : Addition of solvent to the solution is called dilution In dilution process number of…
Q: 10) A physician desires a dose of 10 mcg/kg of digoxin for an 8 lb newborn child. How many…
A: Weight of new born = 8lb Dose required = 10 mcg/kg
Q: Here are two possible methods for measuring the volume of an irregular shaped object: a) Add it to a…
A: For irregularly shaped body - it cannot be measured through a ruler.
Q: H3C CH2 - CH CH 3 CH3 CH. CH3
A: Rule of IUPAC- 1)Choose the longest carbon chain as parent chain. 2) Numbering start from those side…
Q: Amines and Amides: "Describe the chemical properties and use of Kevlar
A: Amines and amides are two organic compounds with similar properties. Amines contain nitrogen atoms…
Q: 3. For the molecule below, CIRCLE the major contributing resonance structure. کل ا
A: Resonance is the phenomenon of describing the delocalized electrons within molecules or ions where…
Q: 2-12. a. Use the ideal gas law (Problem 1-17) to calculate the density (g/mL) of helium at 20°C and…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: A dense plastic-like shape has a mass of 42.985 g and has a volume of 41.96 mL. What is the density…
A: The mass per unit volume of a substance is known as the density of the substance. The density of a…
Q: A canister containing 8g of helium gas (atomic mass 4.0 u) was used to fill up party balloons. When…
A: Answer: This question is based on ideal gas equation and we have to use it to find out the mass of…
Q: Which of the following statements is false? Hd 12 10 8 6 4 2 ON 0 0 10 20 30 Volume of titrant (mL)…
A: Titration is a method to determine the concentration of an unknown solution by using a solution of a…
Q: Calculate the energy needed to heat the cube of silver, with a volume of 10.0^3cm, from 12 degrees…
A:
Q: A popular blue colored medicine usually used to trea symptoms of flu has been analyzed and found to…
A: We have to first calculate the molar mass of each component and then we will calculate the…
Q: Polymers are often thought to be manufactured in immense chemical plants. Although many are, there…
A: A polymer is a large molecule composed of repeating structural units, called monomers, connected by…
Q: How many moles of 2-methyl-2-propanol are used in the synthesis of 2-chloro-2-methylpropane, if 11…
A: The given alcohol is 2-methyl-2-propanol. Density of 2-methyl-2-propanol is 0.781 g/mL
Q: 1. Find the gaseous oxygen solubility in H20 at 20°C when oxygen partial pressure is 1 bar. And take…
A:
Q: Practice Questions for Percent Solutions then fill in your answers in the table. a. To make a 10mL…
A: According to the question, The mass of the NaCl is to be calculated for the given percentage of the…
Q: ОНС CH3 OCH3 -CH3 Н. O Р-оме OMe N₂ KOtBu, THF Н CH3 OCH3 92% CH3
A: This is an example of Seyferth-Gilbert homologation reaction
Q: Determine the formula weight of NH4CI. Provide an answer to two decimal places.
A: Molar mass of N = 14 g/mol Molar mass of H = 1.01 g/mol Molar mass of Cl = 35.45 g/mol
Q: A Fischer projection of a monosaccharide is shown below: CH2OH C=O CH₂OH a. Classify this…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: What quantity of heat, in kJ, is required to convert 50.0 g of ethanol (C₂H5OH) at 23.0 °C to a…
A:
Q: 18. Starting from alcohols of four carbons or fewer, and making use of any necessary solvents or…
A: Note: Since you have posted a question with multiple sub-parts, we will provide the solution only to…
Q: Part E What is unusual about the bonding in the compound boron trifluoride, BF3? The compound is…
A: Part E --> The BF3 (Boron Trifluoride) compound in which the Boron is the central atom and it…
Q: OH PCO a) Compare the two structures. What new functional group is present in the product, but not…
A: On comparing the two structures, we found that carbonyl (C=O) is the new functional group, present…
Q: Balance the following equations using the smallest possible whole number stoichiometric…
A:
Q: Draw an alkyl bromide with proper stereochemistry that can be used to synthesize the given alkene as…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Zinc Phosphate with chemical formula Zn3(PO4)2 is an inorganic dry chemical compound. The Zinc…
A: Given that, The mass of the zinc phosphate, Zn3(PO4)2 is 252.0785 g. The volume of the solution is…
Q: c. The table below shows characteristics of two dopants added in Czochralski growth processes of…
A: Solutions- We have to find the doping concentration of second process (growth 02).
Q: (c) HOCH2 + CH₂OH H H (large excess)
A: Alkene metathesis is a chemical reaction in which two carbon-carbon double bonds come together and…
Q: A sample of iron(II) sulfate was heated in an evacuated container to 920 K, where the following…
A: This question is based on calculation of equilibrium constant in terms of pressure.
Q: Draw the Lewis structure that corresponds to each of the following Newman projections. CH3 CH3 H H…
A: Lewis structure Lewis structures are diagrams that illustrate how atoms in a molecule are bonded to…
Q: Calculate the amount of heat needed to melt 186. g of solid octane (C₂Hg) and bring it to a…
A: •Given:-
Q: Question 14 Aluminum sulfate (Al2(SO4)3) is a chemical compound that is soluble in water. The…
A:
Q: Question 18 of 20 The normal boiling point of a liquid is 282.0 °C. What is the vapor pressure of…
A:
Q: Aluminum metal is a solid at room temperature. When heated, it can turn into liquid, then gas at the…
A: According to the question, The mass of the aluminum is given by = 42.0 gram The initial temperature…
Q: a. 5. 1. Below are transforms taken from retrosynthetic analyses. Show how each would be written as…
A:
Q: 50) It takes 208.4kJ of energy to remove 1 mole of electrons from an atom on the surface of rubidium…
A: As you have asked only for the Q.50, Hence we are solving for the same. According to the question,…
Q: For the following organic reactions, draw the structures of all reactants and products. a)…
A: Organic reactions are chemical reactions involving organic compounds. These reactions typically…
Q: 1. Predict the major product(s) for each of the following reactions aos CH3 NH₂ [H+] [H+] & B (-H₂O)…
A:
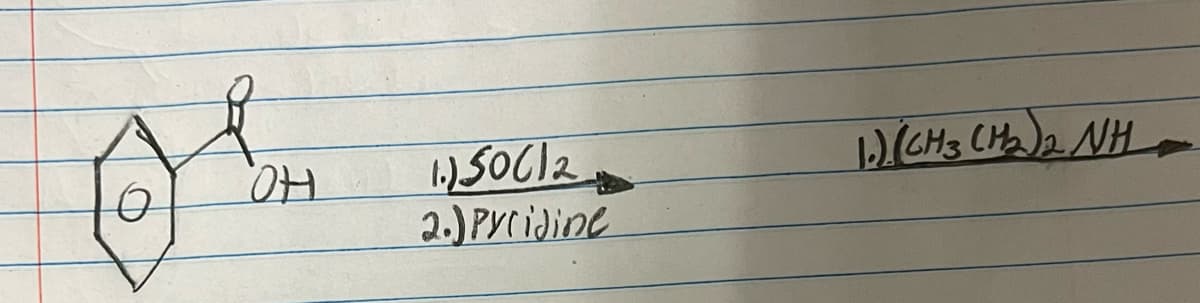
Step by step
Solved in 2 steps with 1 images


