Q: (↑) In the complex ion, [ML6], M* has six d electrons and L is a weak field ligand. According to…
A: The question is asking for the number of unpaired electrons in a complex ion [ML6] where M has six d…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Based on your knowledge on pKa, pH and ionisation describe a method whereby youcan separate a s…
A: The objective of this question is to understand how to separate a solution containing sodium…
Q: Draw the most likely conjugate base resulting from this acid-base reaction. Include all lone pairs.…
A: Step 1: When a proton is removed from hexanenitrile, it becomes its conjugate base (also known as…
Q: None
A: General mechanism:1) Deprotonation2) Nucleophilic attack by the carboxylate.3) Nucleophilic attack…
Q: 106.54 mg of an impure sample of the drug was diluted in 100 ml and injected in a HPLC, the AUC of…
A: The objective of this question is to determine the weight by weight percentage of purity for a drug…
Q: Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the…
A: The salt NaCN, upon dissolution in water, exhibits basic properties. This determination is based on…
Q: Answer the following questions for the molecule E2, where E is the element with atomic number 16.…
A:
Q: If 7.94 g of CuNO3 is dissolved in water to make a 0.860 M solution, what is the volume of the…
A: The objective of this question is to find the volume of the solution in milliliters when 7.94 g of…
Q: A solution is prepared at 25 °C that is initially 0.41 M in chloroacetic acid (HCH2CICO2), a weak…
A:
Q: Which substance would you expect to have the LOWEST melting point? CH3F CH3OH Ni LiCl
A: The objective of the question is to determine which among the given substances - CH3F, CH3OH, Ni,…
Q: please draw out a complete synthesis for the following molecule starting with benzene: 2 -…
A: Step 1: Step 2: Step 3: Step 4:
Q: 6. Draw the most likely major cyclic product, assuming the reaction goes to completion. 1. H3O+ HO H…
A: The major product of this reaction is a cyclic acetal. At first in presence of H+ cyclic hemiacetal…
Q: Potassium metal reacts with water to form potassium hydroxide and hydrogen gas according to the…
A: The objective of the question is to calculate the pressure of hydrogen gas in the container. The…
Q: Draw the product of the reaction shown below at physiological pH (pH = 7.4). Ignore inorganic…
A: 1) Deprotonation 2) Nucleophilic attack by the carboxylate3) Nucleophilic attack by the amine4)…
Q: Propose a reasonable mechanism for the following reaction using curved arrows to indicate the flow…
A:
Q: 1. (4) a) Predict the product(s) for each of the following reactio b) 2 -OH H r.t. Br₂, H+
A: The objective of the question is to predict the product(s) of the given chemical reaction. The…
Q: C. CO2 Lewis Structure VSEPR Sketch Name of Electronic geometry: Name of Molecular shape: d.…
A: The objective of the question is to draw the Lewis structure and VSEPR sketch for CO2, CH3NHCH3, and…
Q: Describe how the addition of an isoxazolidinone to a substrate directs the stereochemistry of a…
A: An isoxazolidinone is a cyclic compound featuring a five-membered ring composed of two nitrogen…
Q: None
A: Solving for Total Pressure at EquilibriumWe can solve this problem using the concept of equilibrium…
Q: Identify the most acidic solution. options: 0.001 M solution of HNO3 10 M solution of CH3COOH 0.001…
A: Solution: To identify the most acidic solution, we can compare the strength of the acids. The…
Q: What is the structure of the unknown? Determine it by analyzing the spectroscopic data given. thank…
A:
Q: Please make sure answer is correct a lot of controversial answer on this I will trust you and take…
A: In the above-given reaction, there are two reagents. Reagent-1: (Sia)2BH is a hydroboration reagent.…
Q: A researcher wants to determine the concentration of beta-carotene in carrots. She obtains a 0.558 M…
A: By dilution of concentrated solution, the less concentrated solution can be prepared.To figure out…
Q: Give the full IUPAC name for the following 6 molecules
A: Representations: The red numbers are for the parent carbon chain. The blue and pink 1/2/3 are for…
Q: None
A:
Q: (c) The error is in the first step. SPh SPh PhS H₂O OH (d) OH CH2 OH t-BuO- CH2
A: Step 1:The reactants is an epoxide which has stereochemistry above the plane. The nucleophile will…
Q: Please correct answer and don't use hend raiting
A: 6 carbons will be from the grignard reagent RMgBr where R is the hexyl. One carbon will be added…
Q: Which of the following is the product of the below reaction? ΘΗ HO Cat ΟΗ Ο Ι
A: Here, given reactants are benzyl alcohol (phenylmethanol) and acetic acidThe reaction between benzyl…
Q: Find the electronegativity difference between Mg and Br.
A: The objective of this question is to find the difference in electronegativity between Magnesium (Mg)…
Q: he titration of 20.0-mL sample of 0.105 M Succinic Acid (Ka= 6.16 X10^-5) with 0.125 M NaOH. The…
A:
Q: Find the electronegativity difference between Br and Br
A: The objective of this question is to find the electronegativity difference between two bromine (Br)…
Q: 42. Assume the reaction: A-2B goes to equilibrium and further assume that the data below represent…
A: To determine the equilibrium constant Kc for the reaction A→2B, we can use the equation:Kc =…
Q: Predict the major product: OH 1. SOCI₂, Et3N 2. CH3S Na HS CI CI ?
A: Step 1: ReactionSN2 reaction follow second-order kinetics.SN2 reaction is Single-step reaction.SN2…
Q: Which two of the following compound are tautomers? I O III and IV O II and IV OI and IV OI and II…
A:
Q: None
A: This molecule contains a carbon chain that contains seven carbons (heptan).There is a phenyl group…
Q: What is the molar solubility of AgCl (Ksp = 1.80 x 10-10) in 0.440 M NH3? (Kf of Ag(NH3)2 is 1.7 ×…
A: Given: [NH3]=0.440M;Ksp=1.80x10−10;Kf=1.7x107Step 1: Write the dissociation of the…
Q: Use the following atomic weights and quantities to calculate the overall % yield of dibenzyl ketone.…
A:
Q: 2. Which indicator could be used for the titration of 25.0 mL of 0.100 M propanoic acid (HC3H5O3, K₂…
A: Propanoic Acid is a weak acidSodium Hydroxide is a strong baseSo, the resulting pH will be greater…
Q: Determine the number of electron groups around the central atom for each of the following molecules.…
A: The objective of the question is to determine the number of electron groups around the central atom…
Q: None
A:
Q: None
A: Let's delve deeper into the mechanism and explanation:1. **Formation of Carbocation (E1…
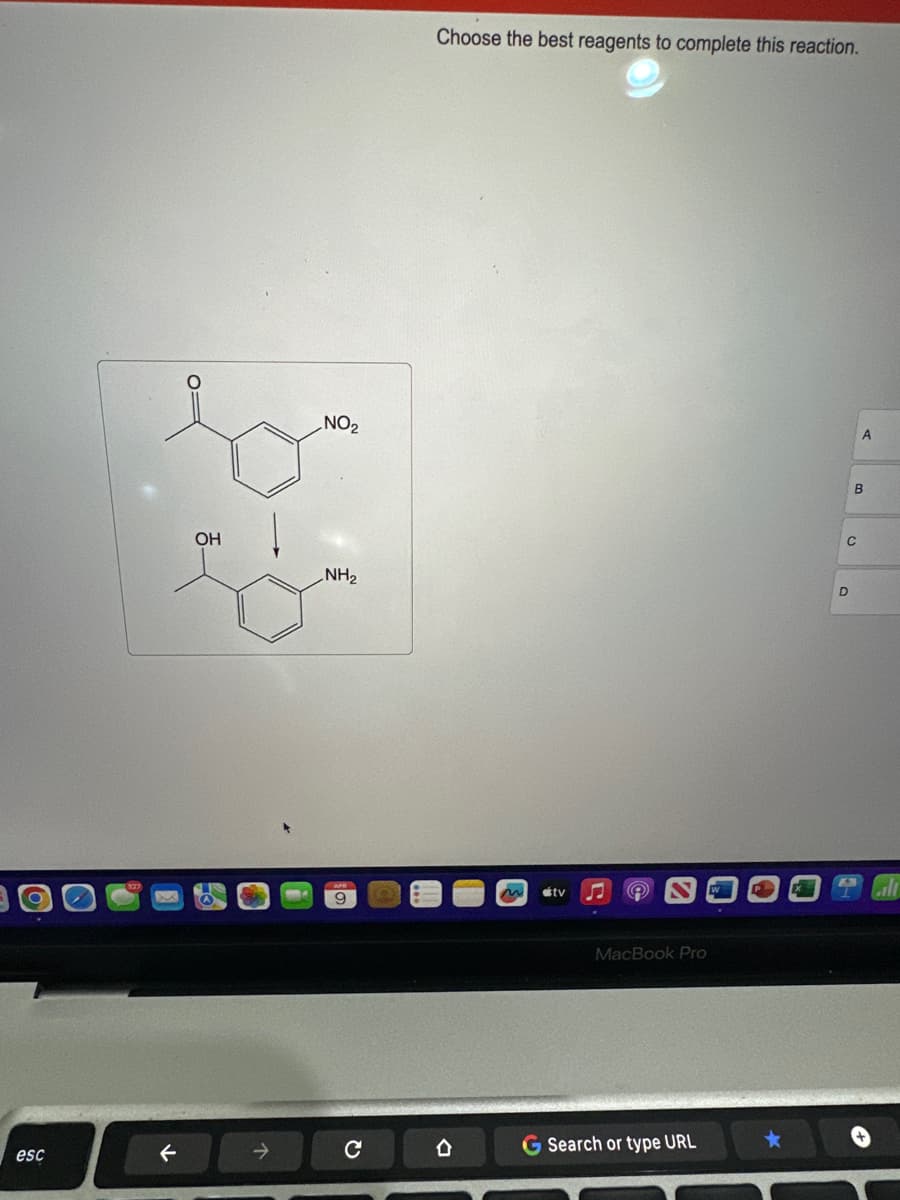
Q: GIVE PROPER EXPLANATION AND PROPER FORMATTING AND DO NOT GIVE SOLUTION IN IMAGE
A: Step 1:• In this step, reaction follows the friedel craft acylation reaction. • Friedel Craft's…
Q: Task: For the reactions below, decide whether the nucleophilic aromatic substitution shown proceeds…
A:
Q: None
A: While directly synthesizing 2-methylbutanoic acid (b) from benzene is a complex process with several…
Q: Select the graph that demonstrates the relationship described by Charles's law. О n P d Λ T
A:
Q: The saturated (or equilibrium) solubility of fumaric acid in n-propanol at L'¹. However, it was…
A: The objective of this question is to predict and explain the phenomenon of supercooling in a…
Q: The synthesis of ethanol from ethylene and water proceeds according to the reaction C2H4(g) + H2O(1)…
A: The objective of the question is to calculate the Gibbs free energy of the reaction under…
Q: The Solubility Product Constant for barium phosphate is 1.3 x 10^-29 . The maximum amount of barium…
A: Given:Ksp of Ba3(PO4)2 = 1.3 × 10⁻29Find:Solubility of Ba3(PO4)2 in presence of common…
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A: AgCN: Silver cyanide (AgCN) is sparingly soluble in water. However, its solubility increases with…
Step by step
Solved in 2 steps with 1 images

- Propose an appropriate intermediate for the following reactionWhy are SN1 reactions favored by polar protic solvents? Group of answer choices Polar protic solvents stabilize reaction intermediates Polar protic solvents cannot form hydrogen bonds. Polar protic solvents contribute leaving groups. Polar protic solvents contribute leaving groups.Fill in the missing reagents a-e in the following scheme:
- What kind of reagent is KmnO4? Draw the possible reactions of this reagent with the specific compounds tested in the following reaction?What is the product of the reaction P4O6 + H2O? A) H3PO4 B) H2PO4 C) H3PO3 D) H2PO3 E) H2P4O7(please answer all questions) 1) Predict the products from reaction of 1-hexyne with thesereagents: a) One equivalent HBr b) One equivalent Cl2 c) H2 , Lindar Catalyst d) NaNH2 in NH3 , then CH3Br

