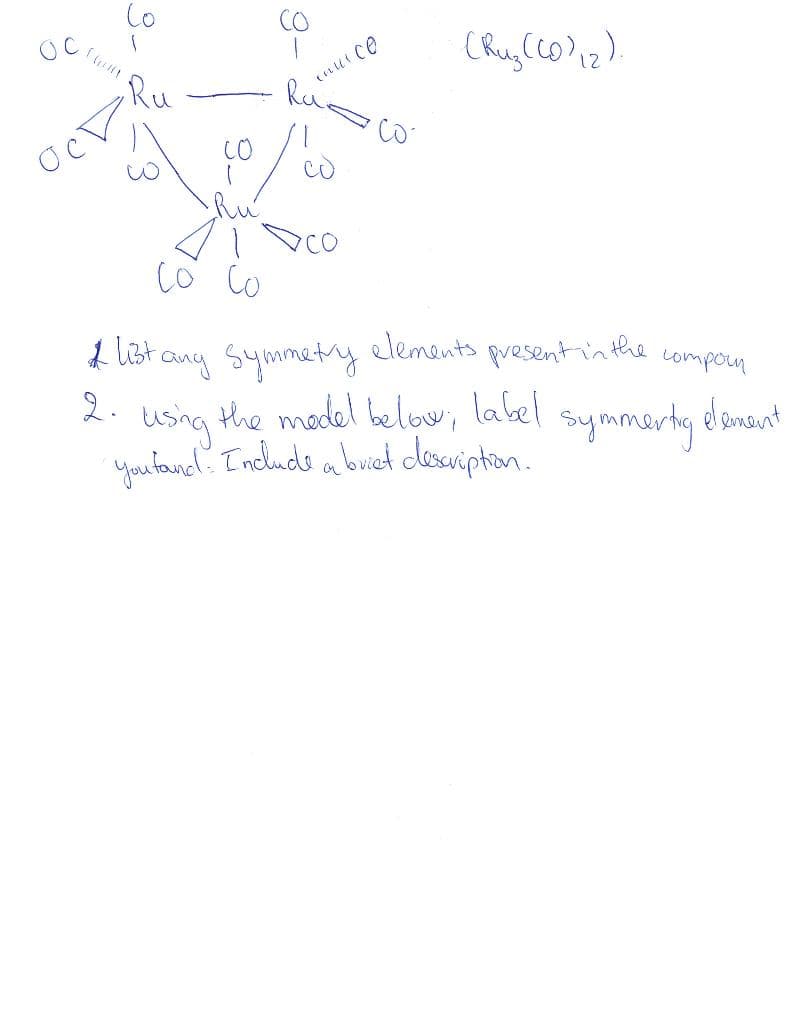
OC ост Co Ru 3 CO •Ru со inut co (Ruz ((0) ₁2). со со & list any symmetry elements present in the compoun 2. using the model below, label symmerty dement youtand. Include a buict description.
Q: the hybridization and geometry around the indicated carbon atoms. CH3 A H3C. HC. CH₂ C C CH3 C
A: Hybridization is the overlapping of the atomic orbitals to form same energy orbitals. And Geometry…
Q: A precipitant will form when Mg(NO3)2 is treated with which of the following? I. K2CO3 II. NaOH III.…
A:
Q: if 0.1 L of 0.3 M HCK is mixed with 0.1 L of 0.1 M NaOH what is the resultant pH?
A:
Q: 14. How many micro grams are in 0.15 mole of NaOH? (a) 0.15 X 106 (b) 0.15 X 10-6 (15 X 40 X 106
A: Mole can be defined as a standard unit for measuring large quantities of very small entitis such as…
Q: onsider a separation performed on a 35.0 mm long open tubular colum with a 0.500 mm diameter and a…
A: To find out the relative retention, given that the separation is carried out using 35.0 mm long…
Q: 4. Among the structures shown below, which one is not expected to give a positive test with ferric…
A:
Q: s of H-1, N-14g/mol), (R-0.0821 L.atm/K.mol), (The specific heat of water is 4.184J/g.°C) C) 6.90 L…
A: STP means standard temperature pressure. Standard temperature is 273 Kelvin and standard pressure is…
Q: For ph of 7 would the structure of arginine be the original structure with 1 NH2 group and 1 NH3+…
A: Arginine is an amino acid that contains guanidium group at the side chain. It has three ionizable…
Q: A hand warmer packet contains a mixture of powdered iron, carbon, sodium chloride, sawdust, and…
A: Answer: Given chemical reaction is: 4Fe(s)+3O2(g)→2Fe2O3(s)
Q: Adipic acid is used in the production of nylon, so it is manufactured in large quantities. The most…
A:
Q: At a certain temperature this reaction follows first-order kinetics with a rate constant of 7.88 x…
A:
Q: 2. Given the following: NaOH (c) H₂O The Lewis Base(s) is(are) (b) NaOH (c) H₂O b (f) a & c (a) NH3…
A: A compound or ionic species which can donate an electron pair to an acceptor compound is called as…
Q: At a weather station the following measurements are made: Air Pressure: 103.8kPa Air Temperature: 34…
A: Vapor pressure is the amount of pressure in millibars exerted by water vapor in the air. But for a…
Q: For a second order reaction, if the concentration of the reactant after 11 seconds is 1.00 M what…
A:
Q: 2.2. Determine the starting materials to form the following Diels-Alder products: CHO CHO CO₂CH3
A: Diels Alder reaction is electrocyclic reaction. In this reaction, diene and dienophile are involves…
Q: Consider the equilibrium for the transesterification reaction. mmmmmmmmmmmm triacylglycerol +…
A: The given equilibrium for transesterification is…
Q: calculate the ph of 0.01M NaOH
A: pH is defined as the negative logarithm of the concentration of H+ ions in a solution. pH = -…
Q: If your answer is from the internet include the references and write your own understanding.…
A:
Q: Which of the following is correct concerning the orbital energy? O4s <4p < 4d < 4f O4s <3s<2s< 1s…
A: Orbital energy:- Orbital energy is decided by (n+l) rule. •Here n=> Principal quantum number…
Q: 8 ● Steel • Platinum Titanium Ceramics Plastics 6 hank the density in order from highest to lowest.…
A: It is given to arrange the density of the given species from highest to lowest.
Q: Relationship between intermolecular forces and pressure.
A: Matter has total 5 type of states out of which three are studied mostly i.e. solid , liquid and…
Q: Please answer fast A compound with the empirical formula CH2 has a molar mass of 56 g/mol. What is…
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as…
Q: In one titration, 48.1 mL of a 0.18 M Ca(OH)2 solution was consumed to neutralize a 0.12 M HCl…
A:
Q: A student performs a separation by TLC on a silica gel platee that results in three spots. A red…
A: Given values are, Distancered spot = 23mm Distanceblue spot = 42 mm Distanceyellow spot = 49 mm…
Q: 13) A solution was made by dissolving 4.05 g of a solute in 130:9 g of acetone. The solution boiled…
A:
Q: msider the structure shown and using single letters (capitalized and without any spaces) give the…
A: Amino acids are buliding blocks of proteins. Amino acid consist of one amino group, carboxylic acid…
Q: Part 2. Macromolecules 1. Before attending Lab 3, use your text book to complete Table UI-2 by…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: how would i calculate the protons and neutrons for 131^I-
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: draw the pric equations (protonated, zwitterin, deprotonated ) of cysterine, glutamic acid and…
A:
Q: b. For electrical conductivity, is there any correlation with the periodic chart? Give the outer…
A: Elements: A pure material known as an element is made up of just one type of atom and cannot be…
Q: Which of the following does not have a associated with it at room temperature A) acetic anhydride B)…
A: Solution We know that only liquid substances have measurable density, but it is deficult to…
Q: B-Galactosidase (ß-gal) is a hydrolase enzyme that catalyzes the hydrolysis of B-galactosides into…
A:
Q: 1.1 Give the correct IUPAC names of the following compounds: 1.1.1. 1.1.2. OCH3 å OCH3
A:
Q: Describe the structure, bonding, and properties of this organic functional group. What is the…
A: A functional group is a substituent or moiety in a molecule that causes the molecule's…
Q: Part of a routine blood test is to check the level of sodium in your blood. It is optimum to have…
A: Molarity of a solution is defined as the moles of solute present in 1 liter of solution. Molarity =…
Q: 6.3x10" 19) Consider the reaction represented by the equation: Fe(aq) + SCN (aq) →→ FeSCN³+ (aq)…
A:
Q: 8) Which of the following pure substances B) H₂S A) HCI C)NH, 9) A solution was made by dissolving…
A: Given data T0 = The boiling point of solvent (pure acetone) = 55.95 °C Tb = The boiling point of…
Q: IW #2 (Chapter 2: Atoms, Molecules and Ions) 1. What is the difference between an atom and an ion?…
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other…
Q: Assume an airplane takes off from sea level at an ambient temperature of 75°F. At cruising altitude,…
A:
Q: ㅛ 9.70 9 8 9.60 7 6 d (ppm) Ⅱ 5 (w 4 3
A: spectroscopic techniques are used to determine the structure of chemical compound.
Q: B) 95 x10³ C) 143x10 D) 303 x10 20) A particular vinegar solution has a H₂O* concentration of 0.013…
A:
Q: [Review Topics] [References] Draw structural formulas for the alkoxide ion and the…
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: Proteins in our body are made of particles with the shape of long chains. The image below shows…
A: GIVEN Proteins in our body are made of particles with the shape of long chains. The image below…
Q: Select the INCORRECT statement All amino acids can exist in two forms, designated L and D. O The…
A: •Here are statement are given in which we have to choose INCORRECT statement. •So let's discuss…
Q: 4) Estimate the temperatul AS-284.5 J/K) CAUTION: Beware of units.NH3(g) A) 685 K B) 545 K 2C+2H2O…
A: 5) Here I have done question 5 as you have asked. Given, 1) CO(g) + H2(g) -----> C(s) + H2O(g)…
Q: Part A Calculate the standard enthalpy change for the reaction Use the following data: Substance ΔΗ,…
A:
Q: 2. Calculate the length of the line, in meters, that models octatetraene (m-8) L= m Submit Answer…
A: To find out: 1. Length of the line in m for octatetraene (n=8) 2. Wavelength (in nm) that is…
Q: Calculate the pH of a buffer solution in which the acetic acid concentration is 1.9 x 10¹ M and the…
A:
Q: A student who is driving home for the holidays averages 40.7 miles per hour. Set up the problem…
A: Given that, Average speed of the student = 40.7 miles per hour. We need to find hours needed to…
Q: If you were told to provide a chemical formula for a compound such as dinitrogen tetroxide, state a…
A:
Give correct answer
Step by step
Solved in 2 steps with 2 images
