One mole of acetyl chloride was mixed with one mole of dimethylamine. After the reaction is complete, what species can be found in the mixture? Draw only the organic structures (i.e., omit inorganic ions). Show charges where necessary. -NH ↑
One mole of acetyl chloride was mixed with one mole of dimethylamine. After the reaction is complete, what species can be found in the mixture? Draw only the organic structures (i.e., omit inorganic ions). Show charges where necessary. -NH ↑
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter21: Benzene And The Concept Of Aromaticity
Section: Chapter Questions
Problem 21.63P
Related questions
Question
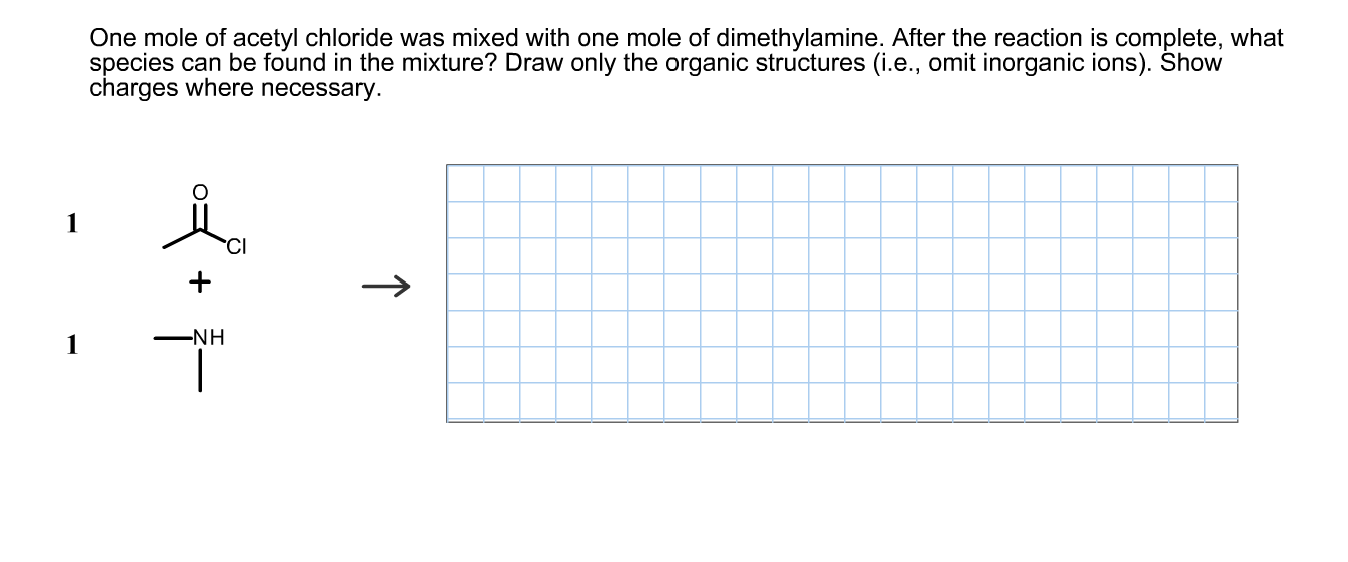
Transcribed Image Text:One mole of acetyl chloride was mixed with one mole of dimethylamine. After the reaction is complete, what
species can be found in the mixture? Draw only the organic structures (i.e., omit inorganic ions). Show
charges where necessary.
-NH
↑
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning