P. 'B 2Vo Vo 0.
Q: please do part d
A:
Q: On the way from a planet to a moon, astro- nauts reach a point where that moon's grav- itational pul...
A:
Q: It's a Statistical Mechanics question.
A: Basic Details The iso-thermal compression is a process in which the gas is compressed at constant te...
Q: please redo question for part 1
A:
Q: For this problem, I know the answer is C but I do not know how to get there. Seeing the steps and eq...
A: The expression for the ratio of the speeds is,
Q: For this problem, I know the answer is E but I do not know how to get there. Seeing the steps and eq...
A: Basic Details The position of the object is determined of the convex lens by the lens formula and th...
Q: A 75-kg block of ice at -13°C is placed in an oven set to a temperature of 105°C. The ice eventually...
A: Basic Details The system consists of ice that is placed in oven so the ice transforms by emitting by...
Q: A large room contains a volume of air of Va = 15 m3 at a temperature of Ta = 53° F. It also contains...
A:
Q: Problem 1: Fermi temperature of the sun At the center of the sun, the temperature is approximately 1...
A:
Q: A barefoot field-goal kicker imparts a speed of 34 m/s to a football initially at rest. If the footb...
A:
Q: A 0.55 kg football is thrown with a velocity of 17 m/s to the right. A stationary receiver catches t...
A:
Q: Fit to page b Page view Current, Resistance, Resistors Circuits, Lightbulbs, Power, Magnetic field, ...
A:
Q: A 0.4 kg mass is attached to a spring that can compress as well as stretch (spring constant 50 N/m)....
A:
Q: When a 24.0-W resistor is connected across a 12.0-V battery, a current of 460 mA flows. What is the ...
A:
Q: A black hole of mass mB = 1.0 x 10^32 kg and a star of mass mS = 2.0 x 10^30 kg rotate about their c...
A:
Q: A 2140 kg car traveling to the west at 17.1 m/s slows down uniformly. How long would it take the car...
A:
Q: I already know the answer to this practice problem, and it is shown is the image. I do not know how ...
A:
Q: Two objects constitute an isolated system. In an elastic collision between the two objects, which on...
A: For elastic collision, both momentum and kinetic energy will be conserved.
Q: An imaginary cubical surface of side LL has its edges parallel to the xx-, yy- and zz-axes, one corn...
A:
Q: A glass optical fiber having an index of reflection nglass = 1.45 is submerged in a liquid with inde...
A:
Q: High-speed stroboscopic photographs show that the head of a 260-g golf club is traveling at 51 m/s j...
A: Consider the scenario in which head of the golf club of mass M hits the golf ball of mass m with spe...
Q: A typical person begins to lose consciousness if subjected to accelerations greater than about 5g (4...
A:
Q: 37 see photo
A: Basic Details Magnetic force is the force of attraction or repulsion that acts between the charged p...
Q: A 2600 kg car traveling to the north is slowed down uniformly from an initial velocity of 30.5 m/s b...
A:
Q: For this problem, I know the answer is E but I do not know how to get there. Seeing the steps and eq...
A:
Q: A submarine is stranded on the bottom of the ocean with its hatch 25 m below the surface. In this pr...
A: The expression for the force is,
Q: There is a minimum energy of (.5[hbar][omega]) in any vibrating system; this energy is sometimes kno...
A: In case if E = 0, then the change in momentum is also zero, this violates the uncertainty principle ...
Q: For this problem, I know the answer is B but I do not know how to get there. Seeing the steps and eq...
A:
Q: (b) The density of iron is 7.85 g/cm3, and its atomic mass is 55.8. Assuming each iron atom contribu...
A: Hey, since there is multiple subpart question posted, we will answer first three question. If you wa...
Q: A rectangular coil of wire, 22.0 cm by 35.0cm and carrying a current of 1.95A, is oriented with the ...
A: Hey there, since you have asked a question with multiple sub-parts, we will solve the first three su...
Q: 51 see photo
A:
Q: A 75-kg block of ice at -13°C is placed in an oven set to a temperature of 105°C. The ice eventually...
A:
Q: On the way from a planet to a moon, astro- nauts reach a point where that moon's grav- itational pul...
A:
Q: Problem 15: A point charge, Q, of –75nC is affixed to the y-axis at yo electron is placed at some po...
A:
Q: = 12.0 V battery, and the C2 = 9.41 capacitor is uncharged. With the switch in position A, the C1 = ...
A:
Q: List three things you could do to reduce your dependence on oil and gasoline
A:
Q: 2) In figure, a solid dielectric sphere of radius a = 2 cm, whose center is at the origin of the coo...
A:
Q: please help in both parts asap
A: Part 1: Given: Charges on the spheres are q1 and q2 ,which are separated by the distance r.
Q: joel is performing a leg curl. To raise the weight, he exerts a vertical force of 1500 N of force to...
A:
Q: Shown in the figure is a closed wire loop. The loop consists of a straight section and a semicircula...
A: Area normal from equation of the plane
Q: How is the net magnetic force is downward for Wire 2?
A:
Q: 10. A coil of 50 turns and area 0.5 m^2 is in magnetic field of induction 2 x 10 -3 Wb/m^2. If the c...
A: The induced e.m.f given as the rate of change of magnetic linkage is, expressed as, Here N denotes...
Q: Yang can focus on objects 150 cm away with a relaxed eye. With full accommodation, she can focus on ...
A:
Q: Two initially uncharged capacitors, of capacitance C and 2C, are connected in series across a batter...
A:
Q: Nitrogen molecules are made of two nitrogen atoms with atomic weight of 14 each. Water molecules are...
A: Given: Water molecule and nitrogen molecule in the vapor form at high temperature show vibrational a...
Q: Q1) A point source with a source strength 0.15 m³ /s radiates sound in air at a frequency of 2400 Hz...
A: Sound pressure level is a logarithmic measure of the effective pressure of a sound with respect to a...
Q: A hydrogen atom consist of a positively charged nucleus (+e)) and a negatively charged electron (-e)...
A: Part i) Basic Details The electron cloud is the continuous charge density whose equation is provide...
Q: Problem 2: Refrigerator Suppose that heat leaks into your kitchen refrigerator at an average rate of...
A:
Q: Problem 6: If a double-slit is illuminated at normal incidence by coherent (laser) light it produces...
A:
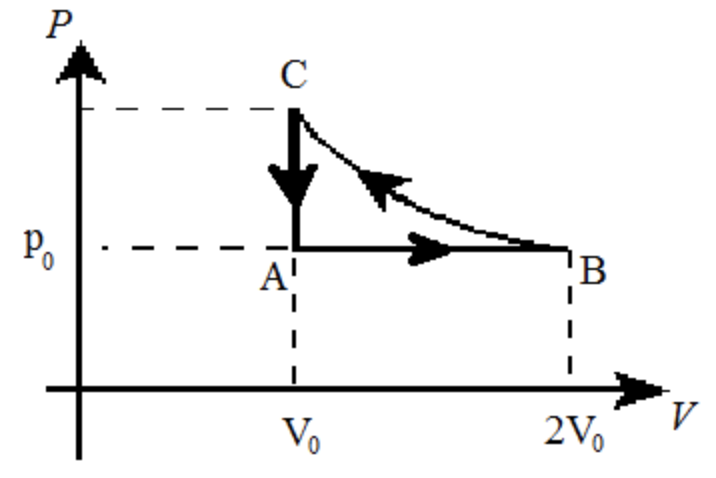
A monoatomic ideal gas is taken through the cycle A → B → C → A shown in the figure.
|
If we know that the internal energy of the monoatomic ideal gas remains constant during the process B → C, what must be the pressure pC at point C in terms of the original pressure p0? Express the work WBC done by the gas during the process B → C in terms of p0 and V0. |
| Express the heat QBC flowing into the gas during the process B → C in terms of p0 and V0. |
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images
