Paracresol (CHROH) has a Ka- 5.5 x 1011. if 100.0 mL of a 0.5000 M aqueous paracresol solution is mixed with 100.0 ml of 0.5000 M aqueous sodium hydroxide, the resulting solution will have a pH OA-7 OB. <7 OC.>7 O D. Cannot be predicted from the information given
Paracresol (CHROH) has a Ka- 5.5 x 1011. if 100.0 mL of a 0.5000 M aqueous paracresol solution is mixed with 100.0 ml of 0.5000 M aqueous sodium hydroxide, the resulting solution will have a pH OA-7 OB. <7 OC.>7 O D. Cannot be predicted from the information given
Chapter14: Principles Of Neutralization Titrations
Section: Chapter Questions
Problem 14.32QAP
Related questions
Question
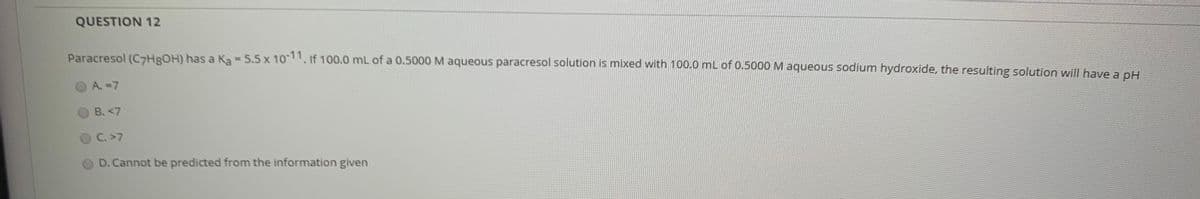
Transcribed Image Text:QUESTION 12
Paracresol (C7HROH) has a Ka - 5.5 x 10-11, if 100.0 mL of a 0.5000M aqueous paracresol solution is mixed with 100.0 mL of 0.5000 M aqueous sodium hydroxide, the resulting solution will have a pH
%3D
A.-7
B. <7
C. >7
D. Cannot be predicted from the information given
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

