Part 2. Predict the Organic Products. Predict the major organic product(s) for each of the following reactions. Be sure to show stereochemistry when appropriate. 1) BH3/THF 2) H₂O₂/NaOH/H₂O
Q: The half life for the radioactive decay of carbon-14 to nitrogen-14 is 5.73 × 10³ years. Suppose…
A: Given: Half-life = 5.73×103 Initial number of atoms of a daughter product = 0.218 mmol Number of…
Q: 4) Identify the element with least electronegativity 1. Fluorine. II. Lithium. III. Oxygen. IV.…
A:
Q: Calculate (1) 4S for the system and its surroundings involved in the reversible isothermal expansion…
A:
Q: Consider the titration of 25.00 mL of 0.100 M nitric acid HNO3 with the 0.100 M KOH. Compute the…
A:
Q: wen the reactions, X(s) + O₂(g) →→→ XO(s) XCO3(s) XO(s) + CO₂(g) at is AH for this reaction? X(s) +…
A:
Q: Iodine-131 is radioactive and has a half life of 8.04 days. Calculate the activity of a 6.6 mg…
A: We need to find the activity of a 6.6 mg sample of I-131. Here we are provided the half life of…
Q: The balanced combustion reaction for CH is 2 C H₂(1) + 15 O₂(g) - 12 CO₂(g) + 6H₂O(1) + 6542 kJ If…
A:
Q: Pentane (C5H12) undergoes combustion according to the following thermochemical equation: C5H12(l) +…
A: Given reaction: C5H12(l) + 8 O2(g) → 5 CO2(g) + 6 H2O(l) ΔH°rxn = -3509.0 kJ ΔH°f [CO2(g)] =…
Q: Complete the following sequence of reactions, giving structural details of all key intermediates
A: The first step of the reaction is the Diels Alder reaction and the second step of the reaction is a…
Q: Work out the Lewis structures for each of the following molecules or ions. Determine formal charges…
A:
Q: What is the right chemical equation when FeCl3 (aq) is added to aqueous phenol? What is the name of…
A:
Q: Predict the major product of the following reactions: H-CH3 OH CHO НО. CH3 CHO CHO CHO 1. H3O 2.…
A:
Q: Chemical Formula XeF4 NH4+ C₂H6 C₂H4 C₂H₂ Lewis Structure (Do not exceed the octet just to minimize…
A: The following molecules given and we have to tell the details of the question like their Lewis…
Q: Calculate the heat of reaction AH for the following reaction: CH₂(g) + 2O₂(g) →CO₂(g) + 2 H₂O(g) You…
A:
Q: Describe the reason why the pH of the reaction medium affects the rate of coupling of the…
A: In this question we are required to answer why the pH of the reaction medium affects the rate of…
Q: Where does LDF, hydrogen bond, and dipole dipole happen in the molecule of ethylamine?
A: LDF - london dispersion forces is a type of interaction where electrons are equally distributed with…
Q: A 25.0 ml sample solution of MG(OH)2 requires 37.3ml of a 0.15M solution of CH3COOH for complete…
A: The reaction between Magnesium hydroxide react with acetic acid to give Magnesium acetate and water.…
Q: answer. Two unique starting materials HBr H3O* ? Br ? *OCH3 大 ? OH 水 OH
A: 2. We have to give the required starting material, reagents, and the products of the given reaction.…
Q: Exercise: Humans maintain an internal temperature of about 37 °C. At this temperature, Kw = 3.55 ×…
A:
Q: Consider the following cell reaction: Mn(s) + 2 H+(? M) Mn2+(1.00 M) + H2(g)(1.00 atm) If the cell…
A: According to the question, The cell reaction is as follows, Mn(s) + 2 H+(? M) Mn2+(1.00 M) +…
Q: Consider the reaction. 2 A(g) = B(g) Kp = 8.24 x 10-5 If a sample of A(g) at 3.20 atm is heated to…
A: According to the question, The reaction at equilibrium is as follows, 2Ag→Bg The temperature of the…
Q: What is the name of the following? CI NO₂
A:
Q: The 1995 Nobel Prize in Chemistry was shared by Paul Crutzen, F. Sherwood Rowland, and Mario Molina…
A:
Q: Strontium-90 is radioactive and has a half life of 28.8 years. Calculate the activity of a 2.1 mg…
A:
Q: In a galvanic cell, Cr(s) oxidizes to form Cr3+(aq) while Cu2+(aq) reduces to form Cu(s). Write its…
A: Electrochemistry can be defined as the branch of chemistry in which we deal with oxidation and…
Q: A voltaic cell is constructed from a standard Fe²+ Fe half cell (E° red = -0.440V) and a standard…
A: Given : Fe2+/ Fe half cell (E° red= -0.440V) Ni2+/Ni half cell (E° red = -0.250V To find :…
Q: The following thermochemical equation is for the reaction of C(s,graphite) with O2(g) to form…
A:
Q: What is the total number of oxygen atoms on the left-hand side of this chemical equation? 2 Ca₂…
A:
Q: (b) a OH OH Hemiacetal: Acetal: + CH3CCH3 Draw Your Solution
A:
Q: Draw the mechanism for the SN2 step. Explain why the products of this step are favored over the…
A: We have find out the mechanism of SN2 reaction.
Q: Quantum numbers arise naturally from the mathematics used to describe the possible states of an…
A: Quantum number are used to define the position and energy of an electron in an atom. Value of 'l' =…
Q: Calculate the value of the equilibrium constant, Kc, for the reaction Q(g) + X(g) 2 M(g) + N(g)…
A: From the question, the reaction is as follows, M(g)⇌Z(g)…
Q: How many milliliters of oxygen gas at STP are released from heating 4.65 gg of calcium chlorate?…
A:
Q: The K sp of Cu 3(AsO4)2 at 25 °C is 7.6 x 10-36. What is the molar solubility (s) of Cu3(AsO4 )2 in…
A: Molar solubility is the maximum amount of salt that can dissolve in the solvent to form Saturated…
Q: 1) Rank the alkaline earth cations (Mg2+, Ca²+, Sr²+, Ba²+) from the one forming the most…
A: 1. Precipitation: It is a chemical reaction in which you mix two solutions of two ionic substances…
Q: Check the box next to each molecule on the right that has the shape of the model molecule on the…
A: We have provided with a bent or V shape structure in the column and we need to identify the suitable…
Q: Concerning the rate law, Rate = k[A], what are appropriate units for the rate constant k? M-¹5-1 M/s…
A:
Q: Use the References to access important values if needed for this question. A standard galvanic cell…
A: Here the cathode is Pb2+||Pb is given. Wne need to find the correct statements from the given…
Q: What is the IUPAC name for the following compound? CH₂=CH-CH=CH-CH=CH-CH₂-CH3 Submit Answer Retry…
A: Organic compound are compound which have different functional group present and they are named using…
Q: Which of the following is the strongest reducing agent? Group of answer choices F-(aq) Na+(aq)…
A: Greater the value of standard reduction potential of a redox couple , stronger be the oxidizing…
Q: What is the molar solubility of PbI2 in 0.10 M Pb(NO3)2 (aq)? [Hint: to which ion concentration…
A: The solution to the given numerical is as follows:
Q: ,4.580g of acetaldehyde C2H4O are put into the "bomb" and similarly completely burned in an excess…
A: The heat gained or absorbed by a substance in a calorimeter is calculated using the following…
Q: Examine the compounds and then select all the following statements that are correct. oi pr 2 00000 3…
A:
Q: One mole of a perfect gas in 22.8 L is allowed to expand irreversibly into an evacuated vessel such…
A:
Q: Draw the skeletal (line-bond) structure of (1R,2S)-1-bromo-2-methylcyclohexane
A: The given compound is (1R,2S)-1-bromo-2methylcyclohexane.
Q: QUESTION 2 6.2 liters of an ideal gas is contained at 3.0 atm and 37 °C. How many moles of this gas…
A:
Q: Which ion shown has empty 4d orbitals? O Nb OY+ O Mo²+ OZ³+ O Mo
A: Given five 4d transition metal ions : Nb5+ , Y+ , Mo2+ , Zr3+ , Mo3+ . We have to select the ion…
Q: 10: :Q: ต่าง * H H H
A:
Q: What happens if you have interchanged the assigned anode as cathode and the cathode as anode in…
A:
Q: % Transmittance 100 50 a) 0 4000 inH₂ NH₂ b) 3000 g 2000 NH₂ d) hemistry Steps 1500 Wavenumber…
A: With the help of IR we can identify presence of functional group in the molecule.
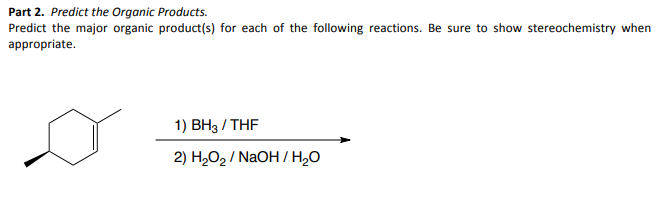
Step by step
Solved in 2 steps with 2 images

- 1) Redraw the structure and assign E or Z configuration to each double bond in the molecule. 2) Draw the organic products obtained from the ozonolysis of the compound, using a reductive work-up with zinc/HAc.Show stereo chemistry and possible outcomes and the steps for the reaction.The addition of an alcohol to an acid chloride is an example of alcoholysis (alcohol addition with bond breakage). Consider the alcoholysis reaction below and answer the questions that follow. 1. Show the tetrahedral intermediate that is formed after the nucleophilic addition of the alcohol to the acid chloride. Be sure to include all lone pair electrons and formal charges on your intermediate structure. 2. Show the final product of this alcoholysis reaction that forms after the intermediate you made in Part 1. Do not include inorganic or charged products in your answer. Be sure to include all lone pair electrons and formal charges.
- Using your reaction roadmap as a guide, show reagents and conditions to bring about these conversions, which may require more than one step.CH₂ + Using the reagents below, list in order (by letter, no period) those necessary to prepare 2-butyne from acetylene. Note: Not all spaces provided may be needed. Type "na" in any space where you have no reagent. a. Br₂, heat, light b. Na, (-H₂) c. Nal(SN2) d. NH4NO3 e. 1) NaNH2, liq. NH3, 2) CH3Br f. (CH3)3COK, (CH3)3COH, heat g. NaOEt, EtOH, heat Step #1 Step #2 Step #31. In the reactions involving the three isomeric alcohols with the formula C4H9OH, describewhat each of the following tests showed about reactivity of the -OH group and reactions of 1°,2°, and 3° alcohols.• the test with neutral KMnO4• the test with concentrated HCl2. Predict how the fourth alcohol with the formula C4H10O would react if tested with:• 0.01 M KMnO4• concentrated HCl at room temperatureExtend FurtherUse your observations of the solutions formed in the previous experiments and yourunderstanding of alcohols to complete a table like the one shown below. Research the meltingand boiling points to verify your answers.
- Illustrated below is the hydrolysis of a C-C bond (in the first molecule) hydrolyzed by water (2nd molecule). Which pair of open boxes best identifies the location of the (-H) and (-OH) components of water on the products. Choose one from the following: (a) A (b) B (c) C (d) DUsing the wittig reaction, synthesise aldehyde 4 to form cis alkene 2, and trans alkene 3. In your answer, with your choice of R group, describe synthetic routes to each alkene and include reagents and conditionsWrite the structure of the principal organic product formed in the reaction of 1-propanol with each of the following reagents:(a) Sulfuric acid (catalytic amount), heat at 140°C(b) Sulfuric acid (catalytic amount), heat at 200°C(c) Dimethyl sulfoxide (DMSO), oxalyl chloride [(COCl)2], triethylamine [N(CH2CH3)3](d) Pyridinium chlorochromate (PCC) in dichloromethane(e) Potassium dichromate (K2Cr2O7) in aqueous sulfuric acid, heat(f) Sodium amide (NaNH2)
- Phineas and Ferbs, two brothers who enjoy vacations, doing fun things every summer. This summer the brothers and their friends carry out an organic synthesis with an unknown compound (L1) that contains 52% Carbon, 6% Hydrogen and 42% bromine, this compound (L1) is treated with magnesium in ether to obtain L2 , which reacts violently with D2O for 1-methyl cyclohexene with a deuterium atom in the methyl group (L3). The L2 reaction is treated with acetone followed by hydrolysis to give L4. Heating L4 with concentrated sulfuric acid gives L5, which decolors the bromine, obtaining L6. L5 undergoes hydrogenation with excess hydrogen and platinum as a catalyst giving rise to isobutyl cyclohexane. Determine the structures of compounds L1 through L6.NH4Cl is sometimes preferred instead of HCl or H2SO4 for “acid” work-up after Grignard reactions, particularly when the expected and desired product is a tertiary alcohol: why?Chhloropropane + sodium ethoxide in the presence of ethanol will yield what products (SN2 reaction). Show the configuration of the product as well as the structural formula

