Part 3: Enthalpy of Neutralization HCl(aq) NaOH(aq) a. Formula of the acid b. Formula of the base c. Mass of empty calorimeter A (g) 2.175 d. Mass of calorimeter + acid (g) e. Mass of acid solution (g) f. Mass of calorimeter B (g) 7.1495 2.2809 g. Mass of calorimeter + base (g) h. Mass of base solution (g) i. Total mass of reactants (g) 7.3360 j. Initial temperature of the acid (°C) k. Initial temperature of the base (°C) 18.5 17.9 I. Average initial temperature (°C) m. Final temperature of mixture (°C) n. Change in the temperature of the mixture, AT (°C) o. Total mass of solution (g) p. Change in heat of solution, Aqm (J) q. Change in heat of the calorimeter, Aq. (J) r. Heat produced by the reaction, Aq: = Aqm+ Aqc (J) s. Moles of water produced (mols) t. Enthalpy of neutralization, AHrxn (kJ/mol) u. Theoretical enthalpy, calculated AHrxn (kJ/mol) v. Percent error 24.0
Part 3: Enthalpy of Neutralization HCl(aq) NaOH(aq) a. Formula of the acid b. Formula of the base c. Mass of empty calorimeter A (g) 2.175 d. Mass of calorimeter + acid (g) e. Mass of acid solution (g) f. Mass of calorimeter B (g) 7.1495 2.2809 g. Mass of calorimeter + base (g) h. Mass of base solution (g) i. Total mass of reactants (g) 7.3360 j. Initial temperature of the acid (°C) k. Initial temperature of the base (°C) 18.5 17.9 I. Average initial temperature (°C) m. Final temperature of mixture (°C) n. Change in the temperature of the mixture, AT (°C) o. Total mass of solution (g) p. Change in heat of solution, Aqm (J) q. Change in heat of the calorimeter, Aq. (J) r. Heat produced by the reaction, Aq: = Aqm+ Aqc (J) s. Moles of water produced (mols) t. Enthalpy of neutralization, AHrxn (kJ/mol) u. Theoretical enthalpy, calculated AHrxn (kJ/mol) v. Percent error 24.0
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter6: Thermochemisty
Section: Chapter Questions
Problem 6.109QP: A 21.3-mL sample of 0.977 M NaOH is mixed with 29.5 mL of 0.918 M HCl in a coffee-cup calorimeter...
Related questions
Question
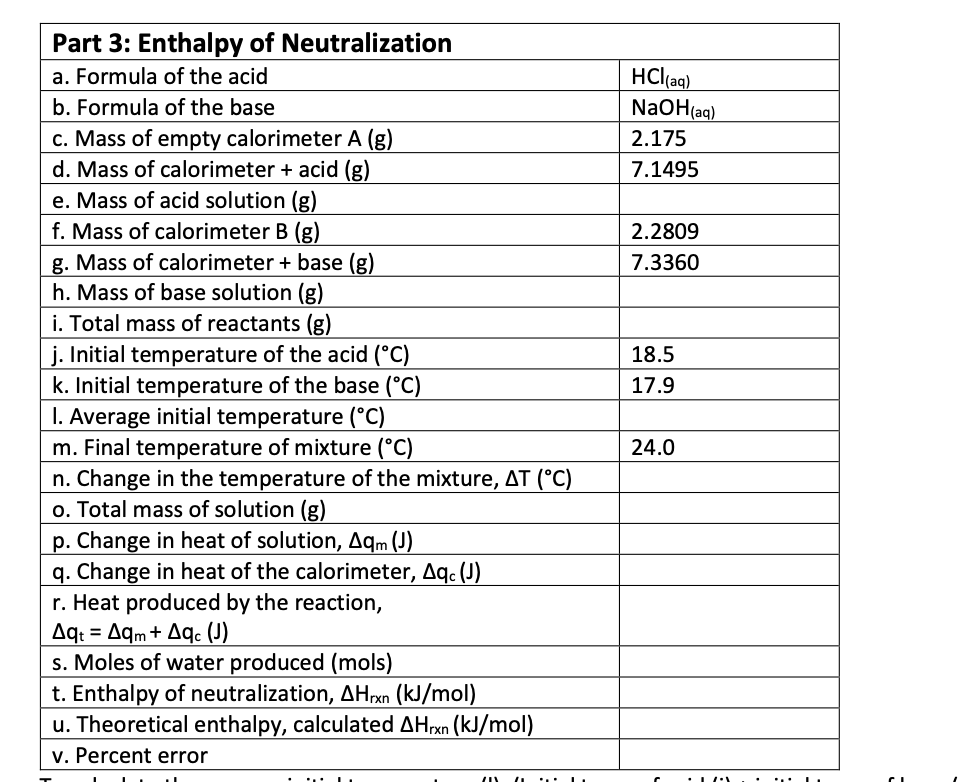
Transcribed Image Text:Part 3: Enthalpy of Neutralization
HCl(aq)
NaOH(aq)
a. Formula of the acid
b. Formula of the base
c. Mass of empty calorimeter A (g)
2.175
d. Mass of calorimeter + acid (g)
e. Mass of acid solution (g)
f. Mass of calorimeter B (g)
7.1495
2.2809
g. Mass of calorimeter + base (g)
h. Mass of base solution (g)
i. Total mass of reactants (g)
7.3360
j. Initial temperature of the acid (°C)
k. Initial temperature of the base (°C)
18.5
17.9
I. Average initial temperature (°C)
m. Final temperature of mixture (°C)
n. Change in the temperature of the mixture, AT (°C)
o. Total mass of solution (g)
p. Change in heat of solution, Aqm (J)
q. Change in heat of the calorimeter, Aq. (J)
r. Heat produced by the reaction,
Aq: = Aqm+ Aqc (J)
s. Moles of water produced (mols)
t. Enthalpy of neutralization, AHrxn (kJ/mol)
u. Theoretical enthalpy, calculated AHrxn (kJ/mol)
v. Percent error
24.0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning