Part A A person who has been a smoker and has a low oxygen blood saturation uses an oxygen tank for supplemental oxygen. The following equation depicts oxygen binding with hemoglobin: What is the stress on the equilibrium of the reaction between oxygen and hemoglobin? Hb(aq) + O2(g) HbO2(aq) O The oxygen concentration is decreased. Answer the following questions to describe what happens when oxygen is first supplied. O The oxygen concentration is increased. O The HbO2 concentration is decreased. O The hemoglobin concentration is increased. In what direction does the hemoglobin equilibrium shift? O The equilibrium shifts in the direction of the reactants. O The equilibrium shifts in the direction of the products. O The equilibrium does not change.
Part A A person who has been a smoker and has a low oxygen blood saturation uses an oxygen tank for supplemental oxygen. The following equation depicts oxygen binding with hemoglobin: What is the stress on the equilibrium of the reaction between oxygen and hemoglobin? Hb(aq) + O2(g) HbO2(aq) O The oxygen concentration is decreased. Answer the following questions to describe what happens when oxygen is first supplied. O The oxygen concentration is increased. O The HbO2 concentration is decreased. O The hemoglobin concentration is increased. In what direction does the hemoglobin equilibrium shift? O The equilibrium shifts in the direction of the reactants. O The equilibrium shifts in the direction of the products. O The equilibrium does not change.
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter14: Chemical Equilibrium
Section: Chapter Questions
Problem 14.95QE: Nitrogen, hydrogen, and ammonia are in equilibrium in a 1000-L reactor at 550 K. The concentration...
Related questions
Question
100%
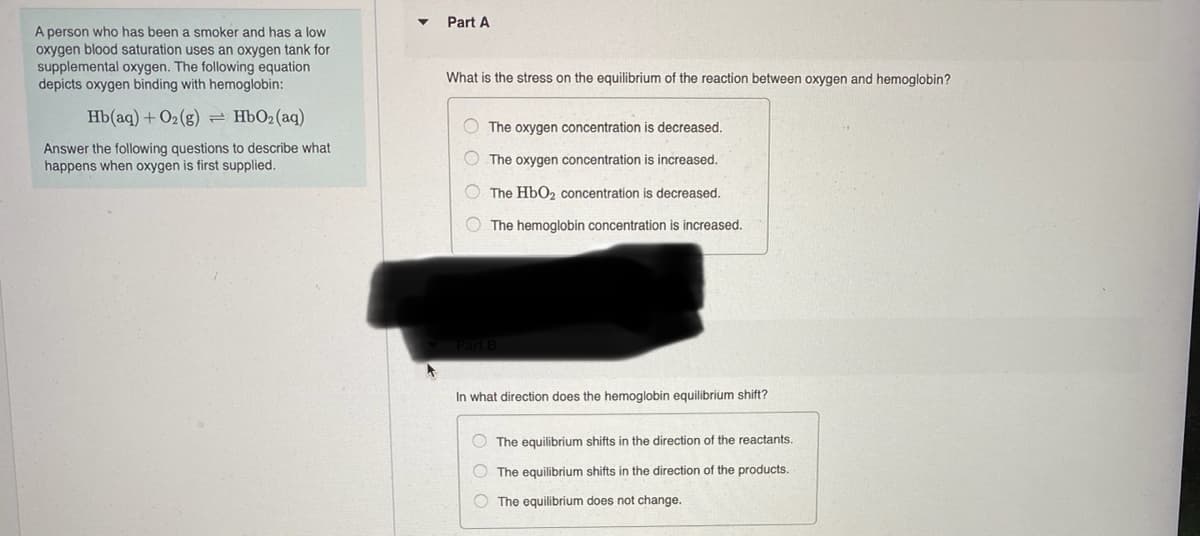
Transcribed Image Text:Part A
A person who has been a smoker and has a low
oxygen blood saturation uses an oxygen tank for
supplemental oxygen. The following equation
depicts oxygen binding with hemoglobin:
What is the stress on the equilibrium of the reaction between oxygen and hemoglobin?
Hb(aq) + O2(g)= HbO2(aq)
O The oxygen concentration is decreased.
Answer the following questions to describe what
happens when oxygen is first supplied.
O The oxygen concentration is increased.
O The HbO2 concentration is decreased.
The hemoglobin concentration is increased.
In what direction does the hemoglobin equilibrium shift?
O The equilibrium shifts in the direction of the reactants.
O The equilibrium shifts in the direction of the products.
O The equilibrium does not change.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax