Part A. Titration of Pure Saturated Ca(OH)2 Solutionsnip Trial 1 Trial 2 Volume of Ca(OH)2 solution used, mL Final HCl buret reading, mL Initial HCl buret reading, mL Volume of HCl solution used, mL Concentration of HCl solution used, M 25.0 25.0 21.3 40.5 0.0 21.3 21.3 19.2 0.05 0.05 Moles of HCl used 1.065 0.96 Moles of Ca(OH)2 solution Concentration of Ca(OH)2 solution, M Concentration of Ca2+ ions, M Concentration of OH- ions, M Ksp value for Ca(OH)2 Average Ksp value for Ca(OH)2 Sample Calculations: Moles = molarity X volume Moles = (21.3)(0.05) = 1.065
Part A. Titration of Pure Saturated Ca(OH)2 Solutionsnip Trial 1 Trial 2 Volume of Ca(OH)2 solution used, mL Final HCl buret reading, mL Initial HCl buret reading, mL Volume of HCl solution used, mL Concentration of HCl solution used, M 25.0 25.0 21.3 40.5 0.0 21.3 21.3 19.2 0.05 0.05 Moles of HCl used 1.065 0.96 Moles of Ca(OH)2 solution Concentration of Ca(OH)2 solution, M Concentration of Ca2+ ions, M Concentration of OH- ions, M Ksp value for Ca(OH)2 Average Ksp value for Ca(OH)2 Sample Calculations: Moles = molarity X volume Moles = (21.3)(0.05) = 1.065
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter14: Equilibria In Acid-base Solutions
Section: Chapter Questions
Problem 74QAP: Fifty cm3 of 1.000 M nitrous acid is titrated with 0.850 M NaOH. What is the pH of the solution (a)...
Related questions
Question
I completed a lab, and have some questions on the write up.
I have about half of the question done, but I'm not sure how to find the moles of Ca(OH)2, and I assume I need that to move on, as the rest doesnt make sense to me either.
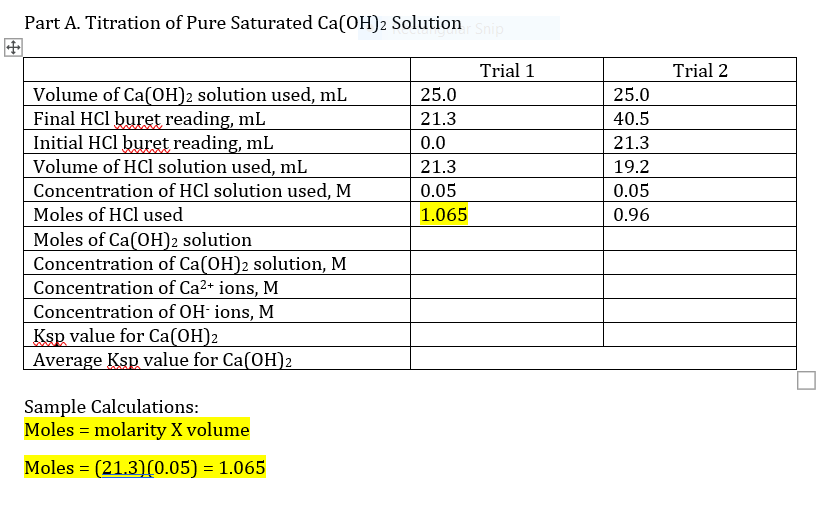
Transcribed Image Text:Part A. Titration of Pure Saturated Ca(OH)2 Solutionsnip
Trial 1
Trial 2
Volume of Ca(OH)2 solution used, mL
Final HCl buret reading, mL
Initial HCl buret reading, mL
Volume of HCl solution used, mL
Concentration of HCl solution used, M
25.0
25.0
21.3
40.5
0.0
21.3
21.3
19.2
0.05
0.05
Moles of HCl used
1.065
0.96
Moles of Ca(OH)2 solution
Concentration of Ca(OH)2 solution, M
Concentration of Ca2+ ions, M
Concentration of OH- ions, M
Ksp value for Ca(OH)2
Average Ksp value for Ca(OH)2
Sample Calculations:
Moles = molarity X volume
Moles = (21.3)(0.05) = 1.065
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning
