Q: If the normal boiling point of a liquid is 67 °C, and thestandard molar entropy change for the boili...
A: The boiling point of a given liquid can be defined as the temperature at which the vapor pressure of...
Q: 2 N2O5 -> 4NO2 + O2 Dinitrogen pentoxide N2O5, undergoes second-order decomposition in chloroform...
A: The given reaction is an example of first order reaction since the unit of the rate constant is min-...
Q: which of the following reactions is not a redox reaction? a. C2H4 + 3 O2 -> 2 CO2 +2 H20 b. 2LiBr...
A: Since in a redox reaction, there will always be change in oxidation state of species This is because...
Q: 13.16 What is the correct answer to this? Please see two photos attached.
A: SOLUTION: Step 1: The base-catalyzed ring-opening of epoxide is an example of SN2 reaction. The nucl...
Q: Why do living systems fight equilibrium?
A: A living cell’s primary task is transforming the foods taken in and using that to do the useful work...
Q: Show the mechanism for the generation of the acylium ion if an acid anhydride is used instead of an ...
A: The mechanism for the generation of the acylium ion if an acid anhydride is used instead of an acyl ...
Q: One compound that contributes to the “seashore smell” at beaches in Hawai‘i is dictyopterene D’, a c...
A: The compound dictyopterene D reacts with excess H2 in presence of Pd catalyst to form butylcyclohept...
Q: What is the correct name for the polyatomic anion for K3PO4?
A: Given, K3PO4
Q: Oxygen masks for producing O2 in emergency situation contains potassium superoxide (KO2). The chemic...
A: A) Given : mass of CO2 exhaled = 0.65 g/min hence in 15 min, mass of CO2 exhaled = 0.65 X 15 = 9.75 ...
Q: Under what circumstance can the entropy of a system decrease for a spontaneous process?
A:
Q: Sulfur dioxide gas reacts with sodium hydroxide to form sodium sulfite and water. The unbalanced che...
A:
Q: Classify the carbocations as 1°, 2°, or 3°, and rank the carbocations in each group in order of incr...
A: Given carbocations,
Q: Orsellinic acid, a common constituent of lichens, is synthesized from the condensation of acetyl thi...
A: Concept introduction: The orsellinic acid is most commonly found in lichens. It can be synthesized f...
Q: g7
A: Given,
Q: Give an IUPAC or common name for each compound.
A: (a) The functional group present in the given compound is acid chloride. The parent chain consists ...
Q: Briefly describe the contributions of Walter Nernst, T. W. Richards, Max Planck and G. N. Lewis in t...
A: Walther Nernst (1864 – 1941) made a significant breakthrough with his statement of the Third Law of ...
Q: Why is chemical potential analogous to temperature and pressure?
A: Chemical potential is given as, From the above equations, chemical potenti...
Q: Need help on the highlighted one thank you
A: (C) In the given compound the molecular structure and position of -OH are same. In molecule (1) -O...
Q: The pKa of p-methylthiophenol (CH3SC6H4OH) is 9.53. Is pmethylthiophenol more or less reactive in el...
A: The electrophilic aromatic substitution reaction can be described as the type of chemical reaction i...
Q: The compound PCl3 can be formed by combining P4 and Cl,. This can be represented at the atomic level...
A: Given: Mass of P4= 28.5 g Mass of Cl2= 97.9 g
Q: In which model is it easiest to follow thepolypeptide backbone?
A: Polypeptide backbone is that when covalent linkage occurs from one amino acid to the other which has...
Q: A solution is 2.25% by weight NaHCO3. How many grams of NaHCO3 are in 150.0 g of solution? A) 1.50...
A: SOLUTION: Step 1: The weight percentage of a solution can be calculated as follows.
Q: Draw all possible constitutional isomers formed by dehydrohalogenation of each alkyl halide.
A: a.
Q: An aqueous solution of a strong monoprotic acid (HA) contains 0.550 g of acid and is completely neut...
A: Titration The molar mass of a compound can calculate using its given mass and the number of moles....
Q: A student is provided with a 0.1 M stock solution of NaOH. Student was then asked to mix 50.0 mL of ...
A: (A) The molarity of the diluted solution is to be calculated. (B) The hydronium ion concentration, [...
Q: 2.94 g of hydrated calcium chloride CaCl2•xH2O is treated with excess K2SO4 in a double replacement ...
A: Balanced double replacement reaction is CaCl2. xH2O + K2SO4 → CaSO4 + 2KCl + xH2O
Q: Bupropion, sold under the trade name of Zyban, is an antidepressant that was approved to aid smoking...
A: In the first step, benzene undergoes Friedel-Craft acylation to form propiophenone. The propiophenon...
Q: Noble gases (Group 18 in the periodic table) have the follow- ing volume concentrations in dry air: ...
A: (a) The ideal gas equation is, PV=nRT. The given pressure is P=1 bar=0.987 atm. Hence, the numb...
Q: Which cation in each set is expected to have the larger (more negative) hydration energy? ...
A: Hydration energy of ion is related to the size and change of the ion. Hydration energy of cation is ...
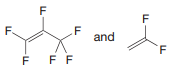
Q: Name the product for the following reaction
A: Addition of H-H across C=C is called hydrogenation. This reaction is facilitated in presence of Pt c...
Q: The equilibrium reaction between dinitrogen tetraoxide, N2O4(g), and nitrogen dioxide, NO2(g), can b...
A: The balanced reaction is as follows: At equilibrium, no. of mol of N2O4 present are 0.24. Thus, it ...
Q: This sketch of a neutral molecule is shaded red or blue wherever the electrostatic potential at the ...
A:
Q: Despite the similarities in the chemical reactivity of elementsin the lanthanide series, their abund...
A: The Oddo-Harkins rule, according to which an element with odd number of proton in the nucleus has a ...
Q: Of the elements Li, K, N, P, and Ne, which (a) is the most electronegative, (b) has the greatest met...
A: (a) Electronegativity of the elements increases along the period from left to right and decrease dow...
Q: please,complete the reaction
A: The nitrating mixture consisting of sulfuric acid and fuming nitric acid react with o-xylene to prod...
Q: Can you please answer all of the sub problems and show all of the steps
A: a) Mg + HNO3 -------> H2 + Mg(NO3)2 Since in RHS we have 2 NO3 ions. Hence making it 2 in LHS al...
Q: Helium occupies a volume of 3.8 L at –45°C. What was its initial temperature when it occupied 8.3 L?...
A: The volume of a gas is directly proportional to its absolute temperature at constant pressure is kno...
Q: Do the following calculations to the correct number of significant figures: a.) 3.8 x 105-8.45 x 104...
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If y...
Q: a5
A:
Q: Why do living systems fight equilibrium
A: SOLUTION: Step 1: The metabolic pathways always try to achieve two things in living system equilibr...
Q: During which of the following processes does the entropy of the system decrease? A. Calcium ...
A: Entropy of any substance is basically the measure of disorder of the substance And the disorder of ...
Q: Write the balanced equation for the neutralization reaction for __H3PO4+__NaOH>>>>___?+_...
A: Since an acid reacts with base to form salt and water hence the products of the above reaction will ...
Q: Draw the products obtained (including stereochemistry) when each compound is treated with LDA, follo...
A: The products obtained when each compound is treated with LDA, followed by CH3I can be drawn as
Q: Thermosetting resins similar to Bakelite (Section 30.7) have also been prepared from renewable feeds...
A: Cardinol and formaldehyde undergoes step growth polymerization in the presence of proton source as s...
Q: Hydrogen sulfide gas (H2S, 34.08 g/mol) can be produced from the reaction of aluminum sulfide (MW = ...
A: The reaction given is Hence from the above reaction we can see that 1 mole of Al2S3 will react to ...
Draw the alternating

Step by step
Solved in 3 steps with 4 images


