Q: The interaction between hexane molecules is: Dispersion force lon-dipole Dipole-Dipole Hydrogen bond
A: The force of attraction present between the molecules of the substance is known as the…
Q: What is the strongest intermolecular force in aniline? A: Ion-dipole forces B: induced dipole -…
A: The strongest intermolecular force in aniline is hydrogen bonding.
Q: Ammonia and hydrogen fluoride both have unusually high boiling points due to _____ A.)dispersion…
A: To determine the reason for high boiling points existing in ammonia and hydrogen fluoride .
Q: Molecules with stronger intermolecular forces will have... Boiling Point Melting Point Surface…
A: Intermolecular forces refer to the type of interactions which are present within the atoms or…
Q: the different between intermolecular forces dipole - induce dipole and Dipole- dipole is А.…
A: Given intermolecular forces are : 1). Dipole-dipole forces 2). Dipole-induce dipole forces…
Q: Cs20 CH3NH2 NF3 CIF C3H3 Dipole- dipole Dispersion forces lonic bonds Hydrogen bonds attractions
A: Ionic bonds is present between highly electropositive metals and highly electronegative non-metals.…
Q: Need help with this problem. Thank you
A: A chemical compound consists of two or more different elements which are bonded with each other…
Q: which of the following will have the strongest dispersion forces? I2 Cl2 F2 or Br2
A: Please find your solution below : Dispersion forces are the type of forces acting between the atoms…
Q: Why are hydrogen bonds considered a special class of dipole- dipole interactions?
A: In solution molecules interacts with each other by molecular interactions. The molecular interaction…
Q: Linear molecules must be nonpolar, thus linear molecules cannot participate in dipole dipole…
A:
Q: hydrogen bonding a. b. ion-dipole ion-ion d. London dispersion force e, dipole-dipole е.
A: When KBr dissolve in water K+ and Br- ions are generated in aqueous solution. Water is a polar…
Q: Ethyl alcohol (CH3CH2OH) easily mixes with water between these two predominantly due to the -…
A: For hydrogen bonding hydrogen must be bonded with a electronegative atom like Fluorine, nitrogen or…
Q: Hydrogen bonding Dipole-dipole forces London dispersion forces ice in glass Roundup herbicide…
A: Hydrogen bonding occurs when hydrogen is attached to Oxygen , Fluorine and Nitrogen .
Q: Sort the intermolecular forces by increasing strength of interaction: Ion-dipole, Dipole-dipole, H…
A: The forces which provide interaction including force of repulsion and attraction acting between the…
Q: What is the intermolecular force of: Part C - C6H5Cl London Dispersion (LD) Hydrogen…
A: The attractive force that withholds two molecules is called as intermolecular force. The influence…
Q: Enter the molecule on each line that has the strongest intermolecular force.CF4, CHF3 _______SO2,…
A: Since the molecule CF4 is completely symmetrical hence its dipole moment will be 0 hence it will be…
Q: Hydrogen bond forms because water has a strong dipole 1. True 2. False
A: Hydrogen bonding :- It is the strongest attractive force among all intermolecular forces. Hydrogen…
Q: ball & stick v labels Indicate with a Y (yes) or an N (no) which apply. dipole forces induced dipole…
A: Applying concept of hydrogen bonding, dipole interaction and induced interaction.
Q: Give the major force between acetone and chloroform. dipole-dipole dispersion…
A:
Q: Dispersion only Dipole-dipole only Dispersion, dipole-dipole, and H-bonding Dispersion and…
A: There are various types of intermolecular forces present in the molecule such Vander Waal, hydrogen…
Q: dispersion hydrogen bonding ion-dipole dipole-dipole Motor oil (nonpolar) Lard (nonpolar) Potassium…
A: Motor oil is non-polar in nature. So in between a non-polar solute and non-polar solvent, dispersion…
Q: Match the following IMFS with the species that are present in each type: Dipole-dipole forces V (…
A: Dipole- dipole forces is the force acting between two opposite dipoles . Ion -dipole forces is the…
Q: Indicate with a Y (yes) or an N (no) which apply. dipole forces induced dipole forces hydrogen…
A: The given molecule is methyl ethanoate and it is one of the ester molecules. The type of…
Q: Identify the strongest interparticle force of attraction that can exist in the molecules of the…
A:
Q: Identify the main intermolecular attraction in the following molecule (attached file)…
A: Hydrogen bond is mainly formed by the ability of specific atoms present in a bond to strongly draw…
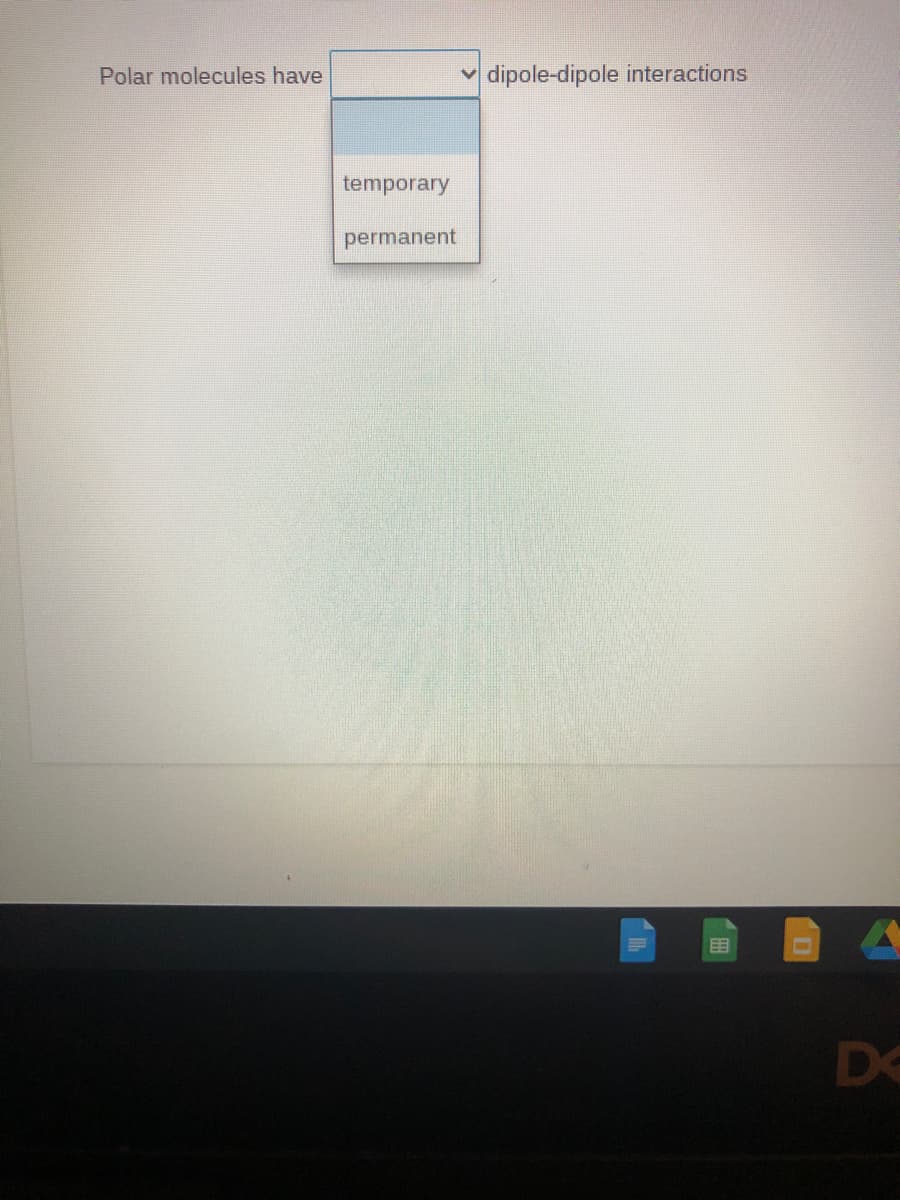
Q: Polar molecules form dipole - dipole intermolecular forces. O False O True
A: If two atoms having significant difference in electronegativity form a bond, then the bonding…
Q: Identify the strongest interparticle force of attraction that can exist in the molecules of the…
A: A question based on intermolecular forces that is to be accomplished.
Q: what intermolecular force is primarily responsible for condensation of Cl2 induced dipole/induced…
A: intermolecular force is primarily responsible for condensation of Cl2
Q: please provide in depth explainations as I will be using this to study.
A: Hydrogen bonding can take place between a highly electronegative atom and hydrogen. Intramolecular…
Q: Direction: Identify the given substances if it is polar or nonpolar, and which type of…
A: 1.H-F is a polar molecule and have dipole dipole forces 2. CH3-CH2-CH3 is a non polar molecule and…
Q: It is the only force present among symmetrical nonpolar substances such as CH4, CO2, SO3, N2,O2, and…
A:
Q: Identify the main intermolecular attraction in the following molecule: (attached file) ion-dipole…
A: Intermolecular forces are the forces responsible for the interaction between the neighbouring…
Q: Directions: As part of our review of our previous lessons on intermolecular forces, list the type/s…
A: We only answer up to 3 sub-parts. Please resubmit the question with other sub-parts ( up to 3) to be…
Q: TRUE OR FALSE 1.the attractive interaction between an ion and the induced dipole is called…
A: Polar molecule is a molecule in which one end of the molecule is positive, while the other end is…
Q: Which of the following molecules H2CO,CH3COCH3,H2,CO2,CH3Cl has the strongest dipole-dipole…
A: Polar molecules contain polar bonds that form dipoles. The dipole-dipole forces are attractive…
Q: Indicate the strongest intermolecular force (IF) in each molecule. Select the correct rank from…
A: A question based on intermolecular force, which is to be accomplished.
Q: Which intermolecular force correlates with polarizability?
A: Polarizability generally refers to the tendency of matter, when exposed to an electrical field, to…
Q: Dispersion force, Dipole-Dipole, and/or Hydrogen bonding? 1) HBr 2)H3C-CH3 3) CO 4) NH3
A: ->Dispersion force-> it is a weak intermolecular force between two atoms and molecule in close…
Q: Directions: As part of our review of our previous lessons on intermolecular forces, list the type/s…
A: Intermolecular forces are the force of attraction of repulsion which applied between the molecules…
Q: Which of the following molecules will have the greatest dispersion forces? A. H2O B. CH4 C.…
A: London dispersion force is a weak intermolecular force between two atoms or molecules in close…
Q: For the molecule below, what is the predominat intermolecular force of attraction? CI mufa London…
A: There are various type of intermolecular forces exist in the molecule such as dipole-dipole,…
Q: Which of the following is the predominate type of intermolecular force that occurs between an ion…
A: Given : An ion and a non-polar molecule.
Q: 1. Write about the molar energies of attraction associated with dipole-dipole interaction using HCI…
A: Note - Since you have asked multiple questions, we will only solve the first question for you. To…
Q: dispersion forces
A:
Q: Polar or London Dispersion Dipole-Dipole Hydrogen Nonpolar Forces Forces Bonding 1.C,H 2.CH,OH 3.H;S…
A: The electrostatic forces that exist between the molecules are termed as intermolecular forces. These…
Q: Dipole-dipole interactions are hydrogen bonds. weaker than stronger than equal to
A: While intermolecular forces are strong enough to keep molecules together in the solid and liquid…
Q: o ion-dipole > hydrogen bonding > dipole-dipole > dispersion
A: Molecules have weak wander vaals force of attraction. Ion-dipole is strongest intermolecular forces…
Q: In which of the following substances the molecules will have dipole-dipole interactions as their…
A: Given substances, Carbon disulfide, Bromine, ammonia and chloromethane molecules.
Q: List all types of intermolecular forces that exist between particles in each of the following pure…
A:
Q: Intermolecular force of H2O? is it non polar or polar?
A: The forces which are acting between the molecules is known as intermolecular forces
Step by step
Solved in 2 steps with 2 images

- Define the term Ion–Dipole Interactions?A certain gaseous molecular compound is 29.7% S and 70.3%F. A 50.0 g sample of this gas occupies 11.3 L at 1.00 atm and 298 K,. Are the molecules of this compound polar?At 0 °C, the molar polarization of liquid chlorine trifluoride is 27.18 cm3 mol−1 and its mass density is 1.89 g cm−3. Calculate the relative permittivity of the liquid.
- Which one of the following molecules is the most polarizable? F2 H2 Br2 Cl2 I2Define this "Torsional strain is an increase in energy caused by eclipsinginteractions" ?(a) What atoms must a molecule contain to participatein hydrogen bonding with other molecules of the samekind? (b) Which of the following molecules can formhydrogen bonds with other molecules of the same kind:CH3F, CH3NH2, CH3OH, CH3Br?
- What physical concept forms the premise of VSERP theory?Which of the following molecules, when viewed in three dimensions, is nonpolar, with zero net dipole? (Hint: It may help to sketch 3-D structures first.) Carbon dioxide (CO2), where C is the only central atom 3 of these 4 molecules are nonpolar CHF3, where C is the only central atom CF4, where C is the only central atom CH4, where C is the only central atomEthylene glycol, HOCH2CH2OH, may look nonpolar when drawn, but an internal hydrogen bond results in an electric dipole moment, Explain.



