Possible compounds: p-chlorobenzoic acid acetaldehyde trimethylamine styrene glucose benzoic acid 4-bromosalicylic acid ethylamine formaldehyde ethyl acetate tert-butanol p-chlorophenol n-propanol acetone sucrose
Possible compounds: p-chlorobenzoic acid acetaldehyde trimethylamine styrene glucose benzoic acid 4-bromosalicylic acid ethylamine formaldehyde ethyl acetate tert-butanol p-chlorophenol n-propanol acetone sucrose
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter19: Enolate Anions And Enamines
Section: Chapter Questions
Problem 19.57P
Related questions
Question
Determine the plausible identity of the compound given the possible compounds.
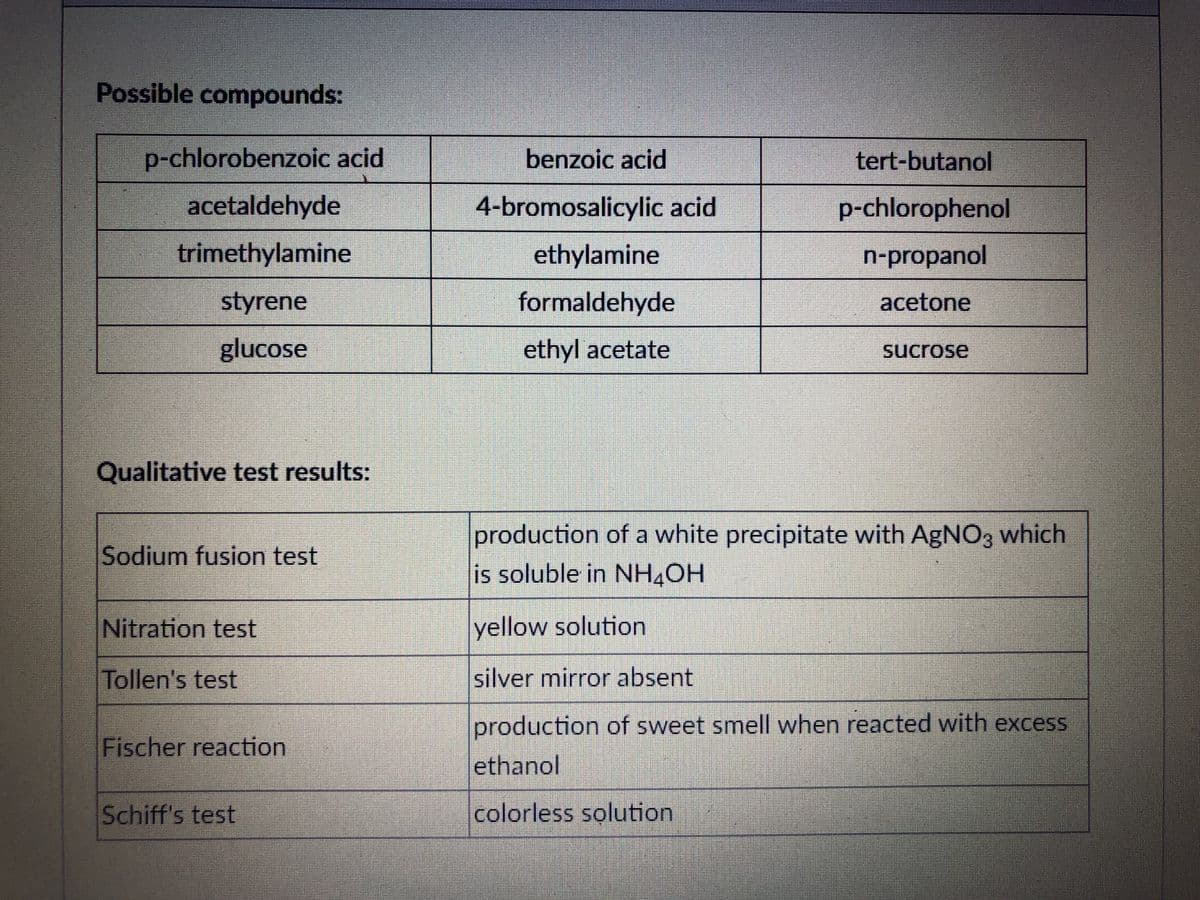
Transcribed Image Text:Possible compounds:
p-chlorobenzoic acid
acetaldehyde
trimethylamine
styrene
glucose
Qualitative test results:
Sodium fusion test
Nitration test
Tollen's test
Fischer reaction
Schiff's test
benzoic acid
4-bromosalicylic acid
ethylamine
formaldehyde
ethyl acetate
tert-butanol
p-chlorophenol
n-propanol
acetone
sucrose
production of a white precipitate with AgNO3 which
is soluble in NH4OH
yellow solution
silver mirror absent
production of sweet smell when reacted with excess
ethanol
colorless solution
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning