Possible mechanistic intermediates (A - I) are shown directly below - the five correct missing intermediates are contained within this group of structures.
Possible mechanistic intermediates (A - I) are shown directly below - the five correct missing intermediates are contained within this group of structures.
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter24: Catalytic Carbon-carbon Bond Formation
Section: Chapter Questions
Problem 24.27P
Related questions
Question
Help
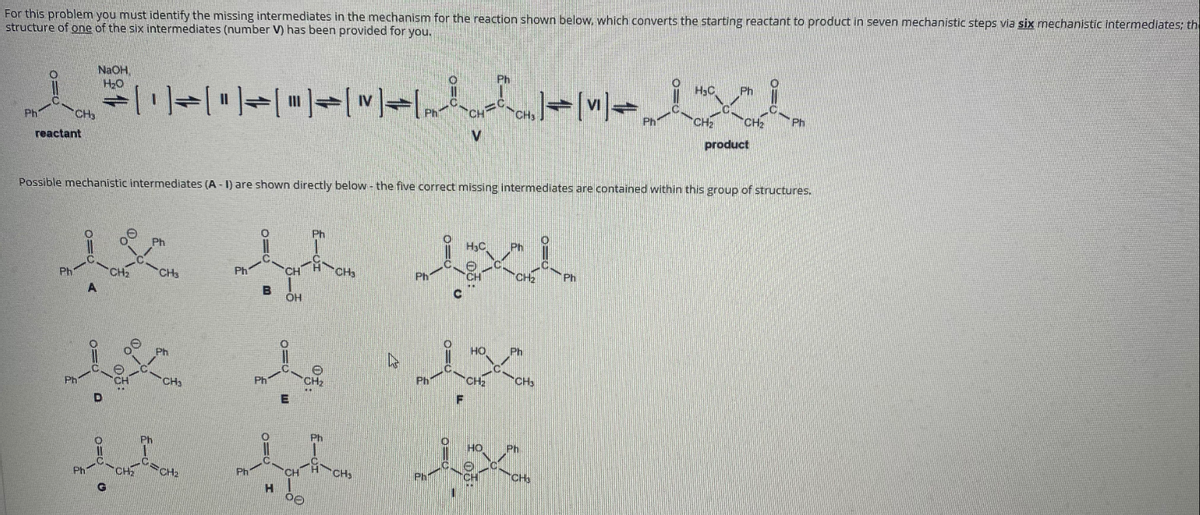
Transcribed Image Text:For this problem you must identify the missing intermediates in the mechanism for the reaction shown below, which converts the starting reactant to product in seven mechanistic steps via six mechanistic intermediates; th
structure of one of the six intermediates (number V) has been provided for you,
NaOH,
H20
Ph
H3C
Ph
Ph
CH
Ph
Ph
CH2
CH
reactant
product
Possible mechanistic intermediates (A - 1) are shown directly below - the five correct missing intermediates are contained within this group of structures.
Ph
HC
Ph
Ph
CH3
Ph
CH
CHa
Ph
CH
CH
Ph
Ph
Ph
CH3
Ph
CH2
CH
Ph
HO
Ph
Ph
CH2
CH
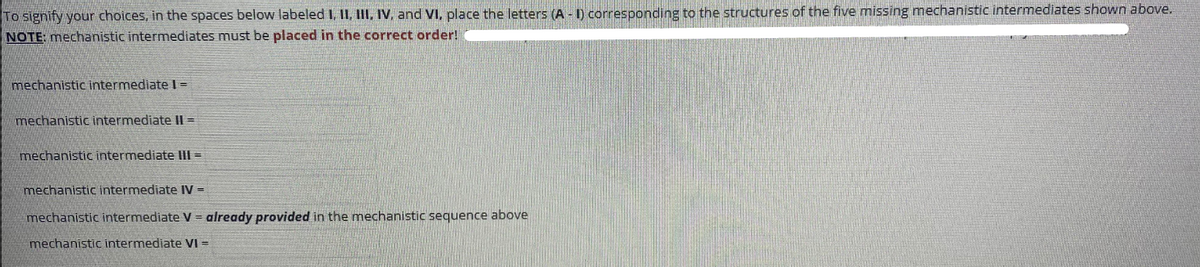
Transcribed Image Text:To signify your choices, in the spaces below labeled I, II, III, IV, and VI, place the letters (A - 1) corresponding to the structures of the five missing mechanistic intermediates shown above.
NOTE: mechanistic intermediates must be placed in the correct order!
mechanistic intermediatel-
mechanistic intermediate II =
mechanistic intermediate III =
mechanistic intermediate IV =
mechanistic intermediate V = already provided in the mechanistic sequence above
mechanistic intermediate VI =
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning