Predict the approximate molecular geometry around each carbon atom of acetonitrile: H3C C N: right C atom left C atom linear linear bent bent trigonal planar trigonal planar trigonal pyramidal trigonal pyramidal tetrahedral tetrahedral
Predict the approximate molecular geometry around each carbon atom of acetonitrile: H3C C N: right C atom left C atom linear linear bent bent trigonal planar trigonal planar trigonal pyramidal trigonal pyramidal tetrahedral tetrahedral
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter7: Molecular Structures
Section7.2: Predicting Molecular Shapes: Vsepr
Problem 7.3E
Related questions
Question
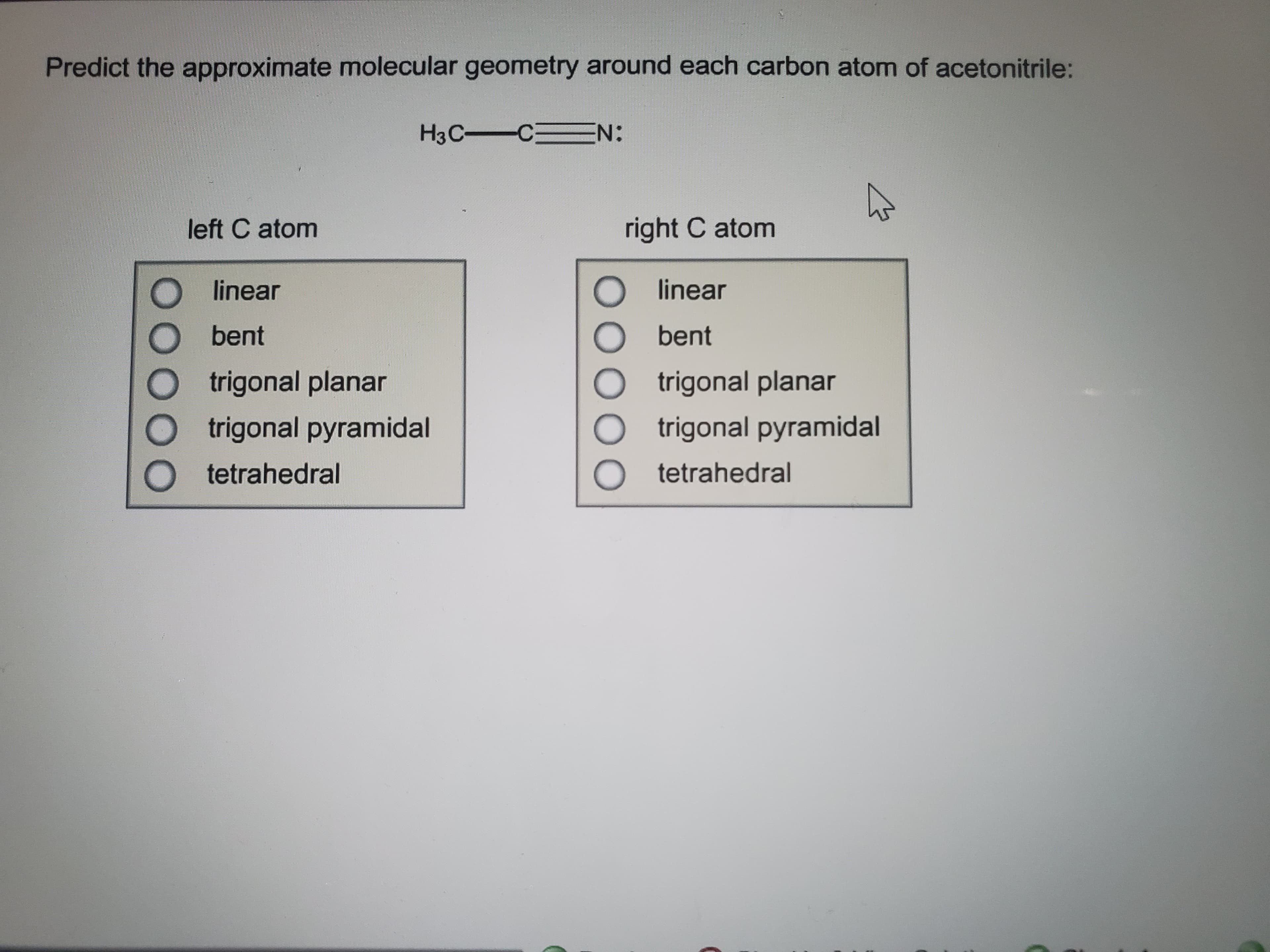
Transcribed Image Text:Predict the approximate molecular geometry around each carbon atom of acetonitrile:
H3C C N:
right C atom
left C atom
linear
linear
bent
bent
trigonal planar
trigonal planar
trigonal pyramidal
trigonal pyramidal
tetrahedral
tetrahedral
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning