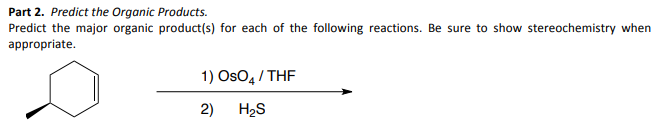
Predict the major organic product(s) for each of the following reactions. Be sure to show stereochemistry when appropriate. 1) OsO4/THF 2) H₂S
Q: Which reagent would be expected to bring about the following conversion? CHỊCHỊCH CH3CH₂CH₂CH₂CHCH₂…
A: We have been given two reactions and we have been asked their reagents.Organic reactants react with…
Q: What is the IUPAC name of the substance shown in the following model?
A:
Q: Which of the monomers shown forms the addition polymer polyethylene? О н Н о н Н Н Н Н Н CI Н Н Н Н…
A:
Q: Determine the net ionic equation for the following reaction (if any) that occurs when the aqueous…
A:
Q: % Transmittance 100 50 a) 0 4000 inH₂ NH₂ b) 3000 g 2000 NH₂ d) hemistry Steps 1500 Wavenumber…
A: With the help of IR we can identify presence of functional group in the molecule.
Q: IZ OH -OH H3O (trace) H2O ? IZ HO HN. -OH но- HN. -OH
A:
Q: 50.00 ml of 0.1000 M Fe(CN)64- solution is titrated with 0.05000 M Tl3+ solution potentiometrically…
A: We can solve this by nernest Equation
Q: 26. Explain the following observations: Me Me Me OH Br₂, heat Me Br Me Me
A: It is from organic reaction mechanism. Here the main thing is decarboxylation and then bromination.…
Q: 9.7 What terminal alkyne and alkyl halide would you use to make the compound shown below?
A: When terminal alkyne is treated with NaNH2 then acetylide is formed which acts as strong nucleophile…
Q: Each value represents a different aqueous solution at 25 °C. Classify each solution as acidic,…
A: pH of a solution is measure of acidic strength If pH > 7 solution will be basic If pH< 7…
Q: The alchemist's dream is to produce gold from cheaper and more abundant elements. This dream was…
A:
Q: Using two equivalents of 1-bromobutane, propose a synthesis to make 4-octanone. Specify the reagents…
A:
Q: Alex Dunphy took a Chemistry exam at CalTech. She was asked to calculate the ionization constant of…
A: Given that, an equilibrium reaction is FAM + H2O FAM- + H3O+. Also, the molar conductance at…
Q: Draw the structure of each of the following molecules. (a) cyclohexyl butanoate; (b)…
A: In this question given nomenclature of three Ester compounds : a) cyclohexyl butanoate ; b)…
Q: A sample of fresh grapefruit juice was filtered and titrated with the above I2 solution. A 100-mL…
A:
Q: Calculate the amount of heat needed to melt 45.3g of solid benzene (C6H6) and bring it to a…
A: Total heat needed to melt benzene is the sum of heat required for melting the benzene and the heat…
Q: The following two-step synthesis gives compound H as the major product. Explain with the aid of a…
A:
Q: Compound A Br₂, H₂O Compound B (C8H15BrO) + enantiomer CH₂O O Compound C + enantiomer Draw the…
A:
Q: Calculate the enthalpy change, A, H, for the formation of 1.00 mol of magnesium carbonate (also used…
A: Here, we have to calculate the enthalpy change for the formation of 1.00 mol of magnesium carbonate…
Q: TRUE OR FALSE: The larger the log Kf of a metal with EDTA, the stronger the bond between the metal…
A: Kf = formation constant of metal EDTA complex. Larger the log Kf value indicate larger the Kf value…
Q: A standard galvanic cell is constructed with Br2|Br- and Sn2+|Sn half cell compartments connected by…
A: In galvanic cell at anode ---> oxidation reaction take place and at cathode -----> reduction…
Q: Explain the method to determine the size of the ring in cellulose.
A: Cellulose is a polysaccharide having a molecular formula (C6H10O5)n. In other words, cellulose is…
Q: 9.6 What terminal alkyne and alkyl halide react to give the following compound? NaNH,
A:
Q: QUESTION 7 In the following gas phase reaction, what is the effect of adding more NO 2 to the…
A:
Q: Compare the de Broglie wavelength of a proton moving at 1.30x107 miles per hour (5.81x106 m/s) to…
A:
Q: Another way to produce gold is through the neutron bombardment of platinum-196. This reaction…
A: Given that Gold can be produced through neutron bombardment of platinum-198. An intermediate,…
Q: Rate Law and Reaction Order Directions: Chloroform (CHCI3) is a known anesthetic. It is also used to…
A:
Q: Carbon tetrachloride reacts at high temperatures with oxygen to produce two toxic gases, phosgene…
A: Note: Since you have posted multiple questions, we will provide the solution only to the first…
Q: a) b) Can these three molecules shown below be distinguished by ¹H NMR spectroscopy when the NMR…
A: Given
Q: Give correct answer
A: calculate the energy released by respiration of 1.00g of sucrose and of myristic acid. Number…
Q: How many seconds are required to deposit 0.137 grams of zinc metal from a solution that contains…
A: Mass of metal = I t × Mole Ratio × Atomic masd of metal / 96500 Where I = current t = time
Q: One mole of a perfect gas expanded isothermally and reversibly at 0 °C from 1 to 1/10 bar. Calculate…
A: GIven , The perfect gas expands isothermally and reversibly .
Q: A technician measures the pressure at 4 locations along a natural gas pipeline, but the gauges at…
A: This is based on the different unit conversions of pressure.
Q: One mole of a perfect gas expanded isothermally and reversibly at 0 °C from 1 to 1/10 bar. Calculate…
A: Given Initial pressure = 1 bar Final pressure= 0.1 bar Initial temperature= 0°C = 273 K
Q: Sodium chloride solution . . . . . . . . . . . . . . 0.9% w/v Purified water, q.s. . .…
A: Stock NaCl solution concentration, C1 = 1/10 = 0.1 fraction = 0.1 x 100 = 10 % w/v Volume of stock…
Q: d. CH3CH₂CH₂CH₂OH e. он
A: P-tolylsulphonic acid after giving H+ is quite stable due to its resonating structure and hence the…
Q: 10. Explain how it would be possible to distinguish the butanone and buten-1-ol based on FTIR…
A: The two given compounds have two different functionality so their peak in FTIR spectra will come at…
Q: QUESTION 2 6.2 liters of an ideal gas is contained at 3.0 atm and 37 °C. How many moles of this gas…
A:
Q: What volume of a 0.259 M perchloric acid solution is required to neutralize 18.9 mL of a 0.192 M…
A:
Q: Complete the following sequence of reactions, giving structural details of all key intermediates
A: The first step of the reaction is the Diels Alder reaction and the second step of the reaction is a…
Q: Calculate the standard entropy of reaction, ΔS° (in J K−1 mol−1), for: Na2CO3(aq) + 2…
A: we have to calculate the standard entropy of reaction : Na2CO3(aq) + 2 HCl(aq) → 2 NaCl(aq) +…
Q: Calculate the standard enthalpy change for the reaction at 25 °C. Standard enthalpy of formation…
A:
Q: 5. Consider a copper concentration cell. One half of the cell has 1.00 M CUNO3 and the other…
A: We need to calculate the ksp of CuCl. It is given that the cell potential is 0.175 V. One half of…
Q: The answer was off its 1.4 M Thanks for nothing!
A:
Q: Bromine-88 is radioactive and has a half life of 16.3 seconds. Calculate the activity of a 5.8 mg…
A: we have to calculate the activity of the sample
Q: Calculate the hinding energy of C-14, in joules per mole. The measured nuclear mass of 14C is…
A: Given,The measured nuclear mass of 14C = 13.99995 amuRequired, The binding energy…
Q: a Assume that mitochondria are cylinders 8.00 um in length and 0.800 pm in diameter. What is the…
A: According to the bartleby guidelines for the multiple questions. I can solve only first question.
Q: Describe a motor end plate
A: Motor end plates are the structural and functional interfaces between motor Neurons and skeletal…
Q: Aqueous potassium nitrate (KNO3) and solid silver bromide are produced by the reaction of aqueous…
A: When aqueous potassium bromide react with aqueous silver nitrate it forms precipitate of silver…
Q: 9.1 Name the compound shown below.
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- 1) Redraw the structure and assign E or Z configuration to each double bond in the molecule. 2) Draw the organic products obtained from the ozonolysis of the compound, using a reductive work-up with zinc/HAc.Illustrated below is the hydrolysis of a C-C bond (in the first molecule) hydrolyzed by water (2nd molecule). Which pair of open boxes best identifies the location of the (-H) and (-OH) components of water on the products. Choose one from the following: (a) A (b) B (c) C (d) DPredict the major organic product of each reaction or provide the reagents needed to complete each transformation.
- Which one of the statements below is wrong about Zaitsev’s Rule. a. None of the above b. The Zaitsev’s Rule demonstrate the principle of regioselectivity in elimination reactions c. The more substituted alkene is the major product of E1 or E2 elimination d. The more substituted alkane product is obtained when a proton is removed from β-carbon that is bonded to the fewest hydrogens e. The most stable alkene is generally the major productShow how to prepare each alkyne from the given starting material. In part (c), D indicates deuterium. Deuterium-containing reagents such as BD3, D2O, and CH3COOD are available commercially.Indicate how to synthesize the following compounds in two or more stages by showing all required reagents (organic and inorganic). DO NOT SHOW THE MECHANISM. This is an organic chemistry exercise.
- Give reasons for the following :(i) Ethyl iodide undergoes SN2 reaction faster than ethyl bromide.(ii) (±) 2-Butanol is optically inactive.(iii) C—X bond length in halobenzene is smaller than C—X bond length in CH3—X.When 1,2-cyclohexanediol is dehydrated in the presence of concentrated sulfuric acid, the major product is not an alkene. Instead, you get cyclohexanone. Write a reasonable and detailed mechanism for the dehydration of 1,2-cyclohexanediol to form cyclohexanone. Use curved arrows to show the flow of electrons and draw the structures of all intermediates and byproducts formed in the course of this reaction as well as any alternative resonance structures that will help you to account for the formation of the major product observed in this reaction.What is going on from compound B to compound C? Can you please select all the options that apply
- The bicyclo [3.1.0] hexane ring system, highlighted in compound 3, is found in several natural products, including sabinene, a compound partially responsible for the flavor of ground black pepper. One method for preparing this ring system involves the conversion of compound 1 to compound 2, as shown below. Draw the structure of compound 2 and provide a reasonable mechanism for its formation. Add any remaining curved arrows to complete step one of the mechanism, and modify the given drawing as needed to show the intermediate that is formed in this step.Please give a step by step for this problem. Give the major organic products.NaNH2 is typically used instead of KOH to produce alkynes through elimination because NaNH2 is a stronger base. Explain why KOH can be used to create diphenylacetylene.

