Chapter17: Alcohols And Phenols
Section17.SE: Something Extra
Problem 69AP: As a rule, axial alcohols oxidize somewhat faster than equatorial alcohols. Which would you expect...
Related questions
Question
Predict the products of each reaction below. Show stereochemistry where necessary.
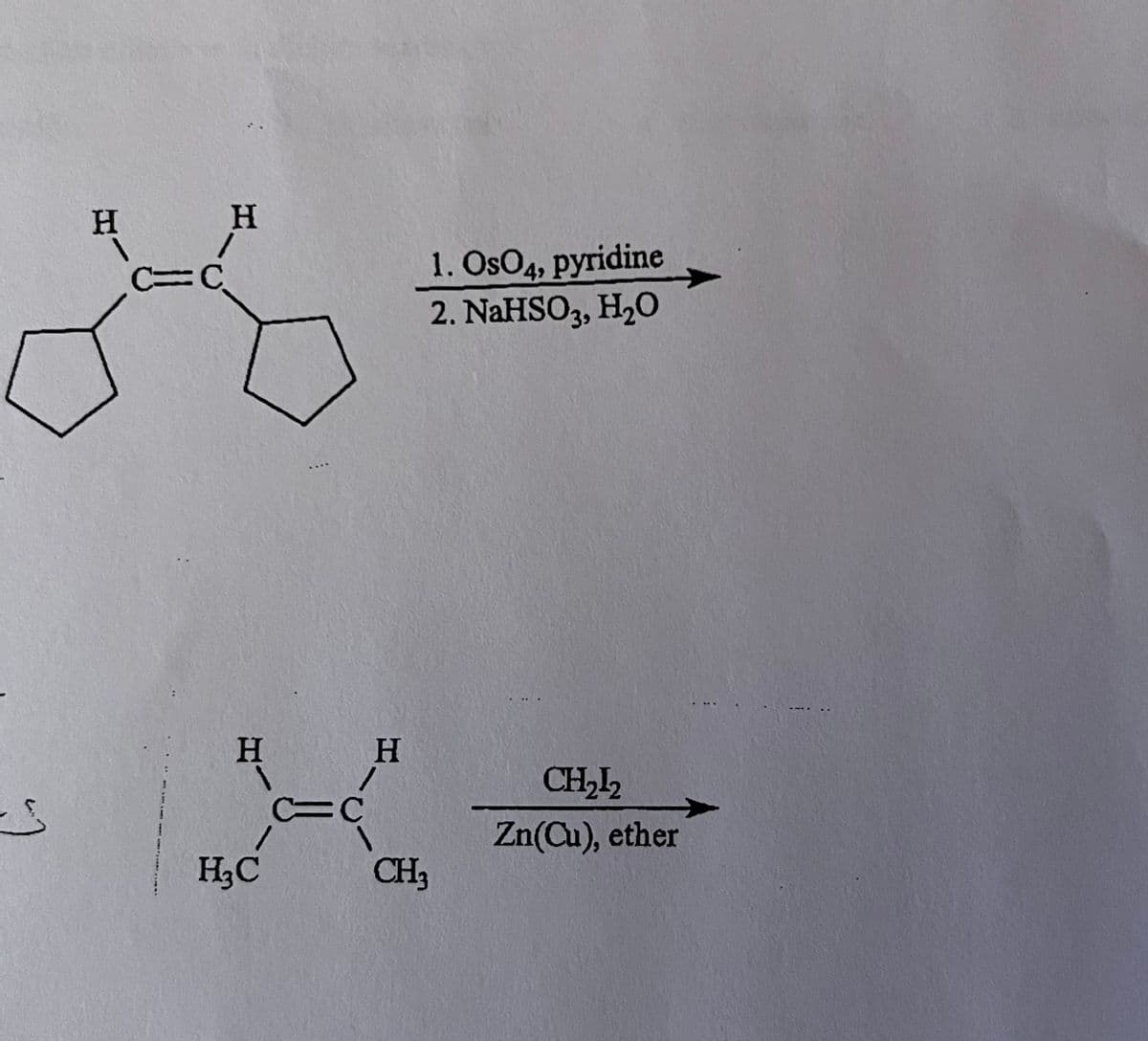
Transcribed Image Text::)
H
C=
C
H
H
H₂C
H
/
=C
1. OsO4, pyridine
2. NaHSO3, H₂O
CH3
CH₂1₂
Zn(Cu), ether
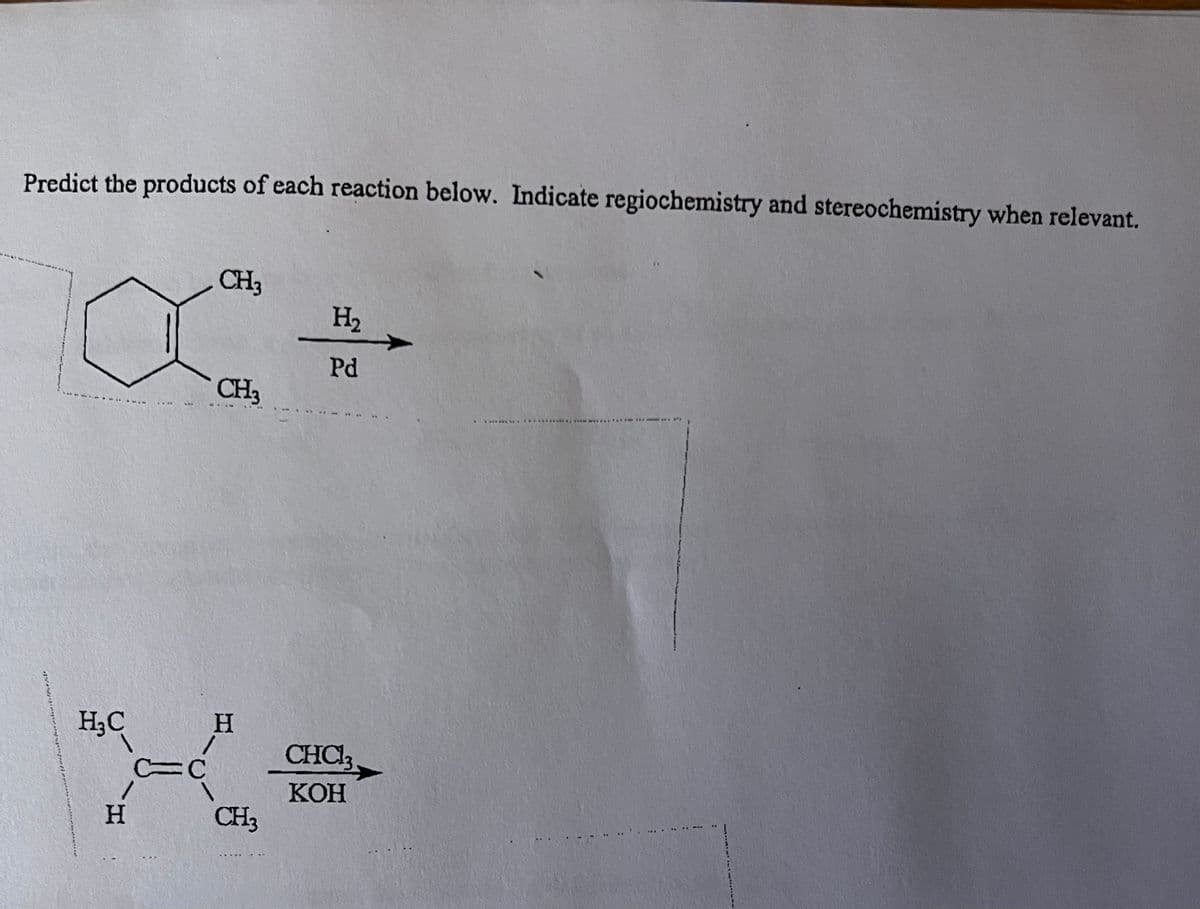
Transcribed Image Text:Predict the products of each reaction below. Indicate regiochemistry and stereochemistry when relevant.
H₂C
H
***
C=C
CH3
CH3
H
CH3
H₂
Pd
CHCl3
KOH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

