Q: 4. Provide a mechanism for each of the following reactions. [9 points each] a) H₂O H+ NH2
A:
Q: Give the organic product: + HNO3 H₂SO4 ? I NO2 II III Select one: O A. II OB. IV O C. I OD. III NO₂…
A: In the given question we need to find the product when given aromatic compound shows aromatic…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: Given that, the reaction is:
Q: Using the reagent list, identify the reagents needed to complete the three-step synthesis below.…
A:
Q: What is the IUPAC name of the following compound? ง B OH ::
A: In organic chemistry, IUPAC nomenclature is a method of naming organic chemical compounds as…
Q: Provide the type of the diene in the following compounds
A: Compound having two double bonds are called as Diene. Dienes are classified as conjugated and…
Q: Dinitrogen trioxide decomposes to NO and NO2 in an endothermic process (ΔrH= 40.5 kj/mol-rxn). N2O3…
A: The objective of the question is to predict the effect of lowering the temperature on the position…
Q: a) the enantiomer also forms, write + E. OH b) CI 1. KCN 2. H3O+, A 3. LIAIH4 4. H₂O 5. SOCl2,…
A: BH3, NaBH4 and LiAlH4 all are acting like a reducing agent. But LiAlH4 acts as strong reducing agent…
Q: Which of the following is not an instance where TLC would be used? The solvents used would attack…
A: Thin Layer Chromatography (TLC) is a chromatography technique employed for the isolation of…
Q: What would be the FINAL result of the reaction(s) shown below? H₂O CH3CH2-CEC-H ? cat H2SO4 cat…
A: Option is BCH3-CH2-CO-CH3Explanation:
Q: Consider the following reaction between calcium oxide and carbon dioxide: CaO (s) + CO2 (g)…
A: The objective of this question is to calculate the percent yield of the reaction between calcium…
Q: In a reaction, 1.00 mole of H2 is produced at a constant pressure of 1.00 atm and at 298 K (1 L atm…
A: +481 kJ closest is +483.6 kJ Explanation:Recall that at constant pressure:dU=q+w and q=dHand we can…
Q: Choose all of the correct products of the following substitution reaction. (d) H D CH3 MeOH, H₂O
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: iClicker: To 120.0 ml 2.8 x 10-3 M AgNO3 solution 0.030 mol solid NaCN is added. What is the…
A: The reaction that occurs when NaCN is added to solution can be represented as follows:
Q: Write the major organic products of the following sequences of reactions. Refer to your reaction…
A: 1. KOtBu:When an alkyl halide reacts with t-BuOK (tert-butoxide potassium), it undergoes a…
Q: Each row of the table below describes an aqueous solution at about 25 °C. Complete the table. That…
A:
Q: At 80.35 K, a gas phase reaction has the constant Kp = 3.92x10-4. For this reaction at this…
A:
Q: For the SN2 reaction, draw the major organic product and select the correct (R) or (S) designation…
A: In an SN2 reaction attack of nucleophil occurs from the back side of the leaving group and thus…
Q: E e' G B H Choose the label which best describes the following: C Solution containing Fe2+ and Fe 3+…
A: An electrochemical device that transforms chemical process free energy into electrical energy is…
Q: The following is a diagram of energy states and transitions in the hydrogen atom. T 1 ENERGY 2 3 n =…
A: The objective of the question is to understand the energy states and transitions in the hydrogen…
Q: CH3CHCCI + 2 CH3CH2NH2 CH3 a. Draw the structure of the tetrahedral intermediate INITIALLY FORMED in…
A:
Q: Multiple step cross coupling reactions give me the synthesis step 8-8 요요 OEt H
A: As per the norms, only the first question can be answered.
Q: What should be the volume of concentrated ammonium hydroxide that should be measured to prepare 100…
A: Volume of the concentrated ammonium hydroxide to be used is 2.75 mL or approximately 3 mL.This…
Q: Order the compounds made of the following molecules by increasing melting point. HO O= HO molecule…
A: Structure 1: 2ndStructure 2: 1stStructure 3: 3rdStructure 4: 4thExplanation:Melting point principle:…
Q: only typed solution
A: The objective of the question is to determine which among Ethene, Propane, Propene, and Ethane has…
Q: Arrange the compounds from most soluble in water to least soluble in water. Most soluble in water…
A: Understanding the solubility of organic molecules in water is important in many domains, including…
Q: Draw the structure of the major organic product(s) of the reaction. 1. LiAlH4, ether -CH3 2. H₂O • •…
A: Information about the question
Q: After a 664 g iron bar stands in moist air for a month, exactly one–eighth of the iron turns to rust…
A: Initial mass of iron bar = 664 g One-eighth of iron turns into rust . It is given that one-eighth of…
Q: A 0.409 M solution of the salt NaA has a pH of 8.83. Calculate the Ka value for the acid HA. Record…
A: pH = 8.83Molarity of NaA = 0.409 MKw = 1.010-14
Q: Step 1: Fast, reversible HA = H+ + A- Step 2: Fast, reversible X + H+ = HX+ Step 3: Slow HX+ =…
A: The question is asking about the effect of adding A- to the solution on the reaction rate in a…
Q: What is the heat of reaction, ΔH°? 5 Cl2(g) + 4 H2O(l) à 8 HCl(aq) + 2 ClO2(g)…
A: Heat of Reaction(Hrxn)=11.4 kJ/molExplanation:From the table of Standard Heats of formation. The…
Q: Cengage Learnin ✓ Cengage Learnin ✓ Cengage Learnin OWLv2 | Online ✓ b-cnow-owl.ceng X what are the…
A: The objective of this question is to find the rate constant of a gas-phase reaction at a different…
Q: Silver chromate is sparingly soluble in aqueous solutions. The Ksp of Ag2CrO4 is 1.12 × 10-12. What…
A: Given:Calculate the solubility of the given aqueous solutions.Common ion effect states that when a…
Q: 4. Complete the following equations: a) Br₂2 b) KMnO4 c) H2SO4
A: Due to the presence of the double bond, alkenes are electron rich and readily undergoes reaction…
Q: If a given carbon in a cyclic monosaccharide
A: IUPAC Name of Structure:(2R,3S,4S,5R)-4-chloro-5…
Q: A 4.914-g sample of a mixture of cyclohexane (C6H12) and naphthalene (C10H8) is dissolved in 66.40…
A: Upon adding a solute to a pure solvent, it is observed that the freezing point of the solution…
Q: What is the organic product formed by the following sequence of reactions? Select one: O A. II OB.…
A: Option B is correct.Explanation:
Q: When ethylbenzene is treated with NBS and irradiated with UV light, two stereoisomeric compounds are…
A: see the explanation below.Explanation:We have to determine the product of the given reaction:
Q: 8. Provide the products for the following reactions: H CH₂ONa CH2Ph CH3OH OH HCI NaOH Br CO3H H CH3
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: H₂N CO₂H H₂N. CO₂H GABA 2 CO₂Et Modify the structures given below to draw all eight possible allylic…
A: l
Q: Which structure corresponds to the NMR shown above?
A: Given is 1H NMR spectra of organic compounds.In given spectra 3 signals are present.In 1H NMR…
Q: What mass of CO2 is produced when 100.0 g of Fe is produced? 118.2 g…
A: 1) Mass of CO2 produced = 118.2 g2) Mass of Fe produced = 69.94gExplanation:The balanced reaction…
Q: Calculate the pH of the solution in which 0.2M NH4Cl and 0.1M NH3 are present. The pK of ammonia…
A:
Q: What is the missing reactant in the reaction below? CO₂Me 4 I II Select one: O A. IV B. III OCI O D.…
A: Both option II and IV are same starting materialsoption II and IV gives the desired…
Q: Consider three molecules: A, B, and C. Molecule A has a hybridization of sp³. Molecule B has two…
A: Three molecules are given, namely A, B, and CHybridization of Molecule A = sp3Number of electron…
Q: What is the pH of 2.11E-4 M KOH(aq) at 25 °C? (Kw = 1.01E-14) -10.3 9.41 10.3 11.1 3.68
A: Concentration of KOH = 2.1110-4Kw = 1.0110-14
Q: draw the curved electron pushing arrows for the following reaction or mechanisitic step. be sure to…
A: The mechanism for the given reaction has drawn with proper explanation. Please find the attachment…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. CH3CH2CH2C(O)CH2CH₂CH pH 4-5…
A:
Q: By using only amide/Peptide bond forming rxn and a IKylation rxns. Provide all the reaction steps…
A: The objective of this question is to plan and execute a sequence of reactions to achieve a target…
Q: a. Draw the structure of the tetrahedral intermediate INITIALLY-FORMED in the reaction shown.
A: Esters on hydrolysis yield alcohol and carboxylic acid. Hydrolysis takes place in the presence of an…
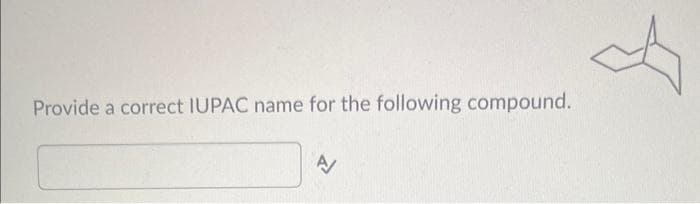
Step by step
Solved in 3 steps


