Provide detailed (arrow pushing) mechanism for the following reaction. Where appropriate indicate Lewis acid and base for each step, and whether they are also Bronsted acids and bases (LB/BA, LA/BA etc.) Indicate the number of steps in the mechanism and the number of intermediates. HO OH H₂O HCI (cat)
Provide detailed (arrow pushing) mechanism for the following reaction. Where appropriate indicate Lewis acid and base for each step, and whether they are also Bronsted acids and bases (LB/BA, LA/BA etc.) Indicate the number of steps in the mechanism and the number of intermediates. HO OH H₂O HCI (cat)
Chapter20: Carboxylic Acids And Nitriles
Section20.SE: Something Extra
Problem 20VC: Electrostatic potential maps of anisole and thioanisole are shown. Which do you think is the...
Related questions
Question
Provide detailed (arrow pushing) mechanism for the following reaction. Where
appropriate indicate Lewis acid and base for each step, and whether they are also
Bronsted acids and bases (LB/BA, LA/BA etc.) Indicate the number of steps in the
mechanism and the number of intermediates.
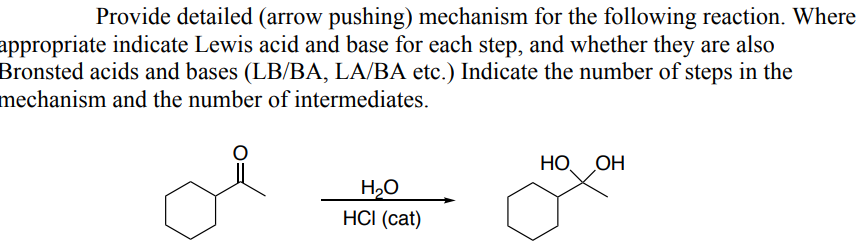
Transcribed Image Text:Provide detailed (arrow pushing) mechanism for the following reaction. Where
appropriate indicate Lewis acid and base for each step, and whether they are also
Bronsted acids and bases (LB/BA, LA/BA etc.) Indicate the number of steps in the
mechanism and the number of intermediates.
HO OH
H₂O
HCI (cat)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 1 steps with 1 images

Recommended textbooks for you

