Q: How do you account for the fact that the dipole moments of fluoromethane and chloromethane are…
A: INTRODUCTION: Dipole moment is defined as the product of charge and the distance of separation…
Q: Which compound gives a molecular ion at m/z = 122: CgHsCH2CH2CH3, CgHsCOCH2CH3, or CHsOCH,CH3? %3D
A: Given: m/z= 122. To find: The ion which has the given m/z value.
Q: CH3 A) aromatic B) anti-aromatic CH3 C) non-aromatic here to search 2:07 82% 54°F 后 小 3/17
A: Aromatic compound properties: 4n+2 delocalised pi- electrons. It should be planar. Cyclic…
Q: Which of the following compounds does not have an alpha hydrogen? CH L. II. IL CH3 O H-C-H IV. CH3C…
A:
Q: Molecular Formula: C7H6N2 4 2 T 4000 3000 2000 1000 CM 9 8 7 6 5 4 3 2 1 0 6 N
A: From the given formula first calculate the degree of unsaturation as shown below, IHD =…
Q: [ 21] which of following compound is predicted to have the LOWEST dipole moment ? (a) (b) (c) (d)…
A:
Q: The dye Tartrazine appears yellow because it absorbs yellow light (590-560 nm) in the visible…
A: Hello. Since your question has multiple parts, we will solve first question for you. If you want the…
Q: SAQ 8-7. Predict the products. но HO, 573 K + 2n
A:
Q: Lewis structures of mescaline, a hallucinogenic compound in peyote cactus, and dopamine, a…
A: The reason why mescaline has the tendency to disrupt the nerve impulses has to be given.
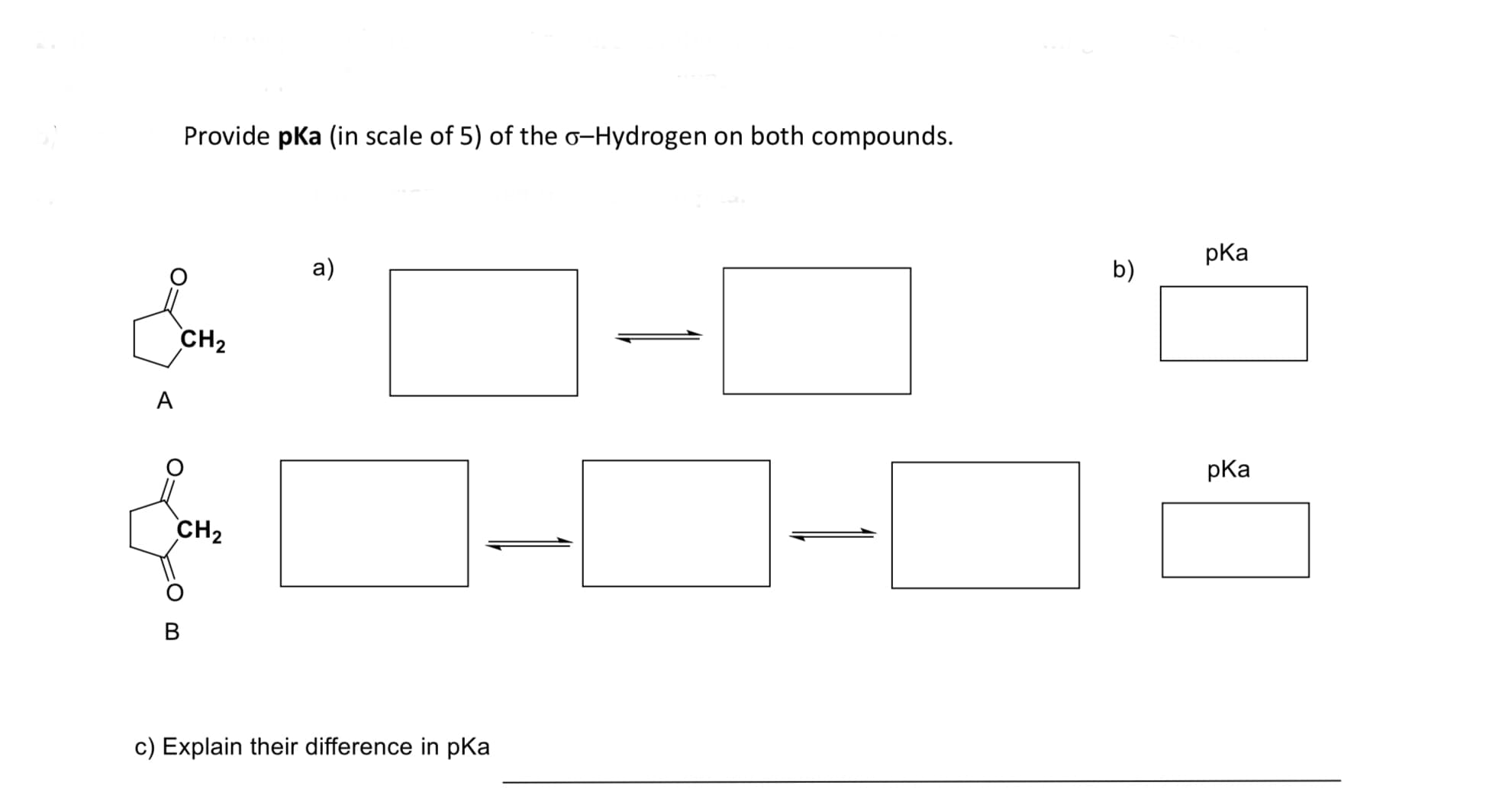
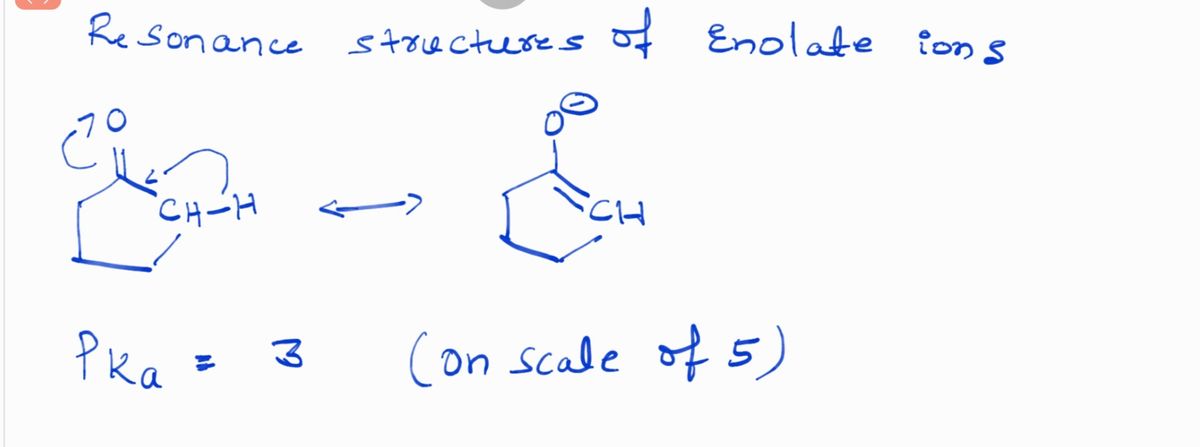
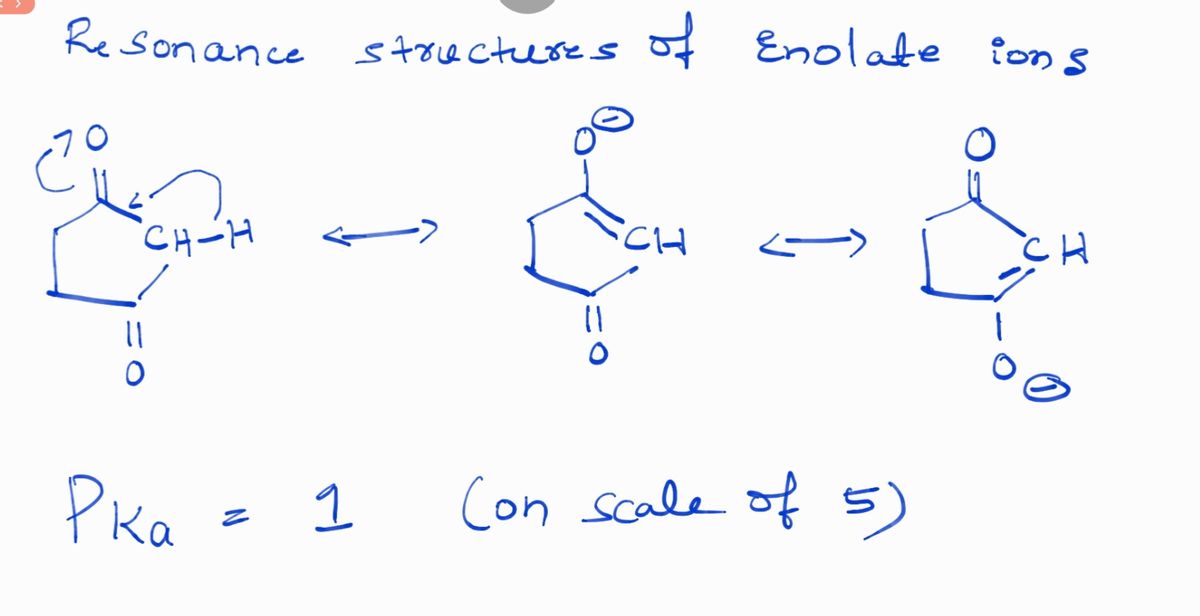
Q: Estimate the pKa of each of the indicated bonds.
A: a.
Q: 1. Which of the following ions is aromatic? IV B. Only Il and III II II A. Only I and |I C. Only III…
A: Q 11) The ion is aromatic when it is cyclic, planar and follows Hückel rule. Hückel rule is that the…
Q: Which is the best description for the indicated bond in the following compound? CH; O a) sp3-sp3 b)…
A: The above bond is given is double bond.
Q: Organize the following four structures in order of increasing pKa. CI CH3 CH3 OH SH OH SH 1 3 4…
A: Acidity is the tendency to loss Hydrogen in an organic compound. It is measured by the value of…
Q: Arrange the molecules according to where you would expect to find their wavelength of maximum…
A:
Q: Propose molecular formulas for: a. m/z = 134 b. M+ = 166
A:
Q: Provide the starting material. LiCu ОН OCH3 CI A B D E = A or B F = B or C G = none of these
A: The reaction given is,
Q: (c)) A partial character table for C3y poin group is given as C30 ? 1 E 2C3 30, 1 ? 1 1 -1 ? 2. -1
A: Character table is a two-dimensional table whose rows correspond to irreducible representations and…
Q: What is the product? NH2 mild acid heat NH2 N. NH2 NH II IV %3D
A: We have given the organic reaction and we have to find the product of reaction.
Q: Arrange the following compounds in the decreasing order of pka values. он он он CH;CH,CH,OH…
A:
Q: `NH2 NH2 :0: NH2 `NH2 A D В O C&D
A: Cross conjugation: Due to cross conjugation, the extent of resonance decreases slightly. therefore,…
Q: What hydrogen atoms in each compound have a pKa ≤ 25?
A: The pKa of alpha hydrogens attached to carbonyl compounds is below 25. The negative charge of the…
Q: Which of the following isomers (all have the same molecular formula of C5H1002) has the highest pka…
A:
Q: Arrange these structures in decreasing order of reactivity towards bromin ation NH NO 3 O A) 1>3>4>2…
A:
Q: S.2. Prove the aromaticity of the molecules given below by discussing them. w.0.00. H a) b) d)
A: a) the given molecule is cyclic and but not planar (all the atoms are sp² hybridised except one…
Q: which of the following isomers will be converted to salt a more rapidly quaternary N(CH3)₂ ~N(CH3)2…
A:
Q: Predict the following products: PCC HBr E A OH F SOCI, в но PCC OH OH HO PBr3 Cro3 OH H Cro, PBr3 OH
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: CH3 CH3 H3C What is the most likely pka for the drug ibuprofen, structure shown? Oa. 4.6 Ob. 2.2 Ос.…
A: The most likely pKa for this drug is 4.6
Q: What is the value of Kb for the formate anion, HCOO-? Ka(HCOOH) = 2.1 × 10-4
A: The relationship between Ka and Kb is shown below:
Q: Which hydrogen in each molecule is most acidic? (See the Attachment)
A: Acidic Hydrogen is the one which is attached to electronegative atom having lesser charge density so…
Q: Draw the resonance structures of [CH3C(OCH3)2]+ and identify its minor and major contributors.
A:
Q: Nicotinic acid, more commonly named niacin, is one of the B vitamins. Show how nico- tinic acid can…
A: The organic reactions help in the conversion of one compound into many other compounds with the help…
Q: НО- + trace HC1 H. но. Он H;C CH3 H;O*, A H;CH,CO OCH,CH3
A:
Q: H. H H. H;CO E
A: ->HCN give nucleophilic addition reaction to carbonyl center.Hence, the carbonyl center which has…
Q: Draw the structure of 2-phenylpropan-1-ol. Draw the molecule on the canvas by choosing buttons from…
A: We are authorized to answer one question at a time, since you have not mentioned which question you…
Q: 3. The pKa1 and pKa.2 of ortho and para isomers of phthalic acid (diprotic acid) are shown below:…
A: The acidity of pthalic acid is higher due to: ● ortho effect ● -ve Mesomeric Effect because…
Q: Consider the three compounds shown below and then answer the question that follow: H H. H •C -C -C…
A: Atoms are bonded to each other in a molecule through two different bonds, a covalent bond or an…
Q: (c) Arrange the following species in order of increasing stability. Explain. CH, H,C CH3 H,C-HC `CH…
A: A question based on stability of carbocation, which is to be accomplished.
Q: Propose a structure for compound B. HC. H H;CO OCH3 EtN H,PO.. calor в *NH2 Br
A: Answer:- Option (2) is the correct answer.
Q: 2b. Arrange the following compounds in order of decreasing pka values. COOH O2N- -COOH -соон O2N II…
A:
Q: Which is the best description for the indicated bond in the following compound? CH3 O a) sp3-sp3 O…
A:
Q: spectroscopic methods, how would you distinguish between the pairs of: a) [CoCl(NH3)5]Cl2 and…
A: (a) Electron spectroscopy can be used to differentiate these complexes.
Q: он OH d. C2H5 e. f. decane-2,2,9-triol 24 diguclonronule clobute 1 2
A: Since you have posted multiple subpart questions, the answer for first three subpart of question is…
Q: 6. Fill in the missing information for the following "road map" problem. 1) LİAIH4 PB13 Mg° B THF…
A:
Q: For the compound below the bonds are primarily : I Linear Formula CH,(CO,H),…
A: The structure is given above. It is clear that, All the 6 carbon atom in the ring are forming…
Q: Arrange the following compounds in the decreasing order of pKa values. OH OH OH CH;CH,CH,OH…
A:
Q: 19) Which of the following species is the most nucleophillIC! A) H2O B) F- C) HS- D) NH3 E) CH3O-
A: Note:- A charged nucleophile is stronger than a neutral nucleophile. So we have to choose only…
Q: Which of the following fluorides would react the most vigorously with water? Select one: a. BF4- b.…
A: From the given, NaF and MgF2 are the salts and do not react with water they simply dissolved in…
Q: Arrange the following in order of increasing acidity: HCN, ClCH2COOH, HClO, C6H5COOH, CH3COOH
A: Electron withdrawing group increases the acidity. Electron releasing group decreases the acidity.…
Q: Consider the following reaction and choose the appropriate response: A. Both halides cis and trans…
A: Alkyl halides reacts with EtONa in presence of EtOH and produces alkenes.
Q: Arrange the following compounds in the decreasing order of pKa values. OH OH CH;CH,CH,OH…
A: Acidity of a compound directly proportional to the stability of its conjugate base. Higher is the…
Step by step
Solved in 3 steps with 2 images

- Arrange the following groups in order of increasing priority. Q.) -CH3 -CH2OH -CH2NH2 -CH2BrThe compound whose structure is shown here is acetyl acetone. It exists in two forms:the enol form and the keto form The molecule reacts with OH–to form an anion, [CH3COCHCOCH3] (often abbreviatedacac–for acetylacetonate ion). One or the most interesting aspects of this anion is thatone or more of them can react with transition metal cations to give stable, highlycolored compounds (a) Are the keto and enol forms of acetylacetone resonance forms? Explain youranswer.(b) What is the hybridization or each atom (except H) in the enol form? What changesin hybridization occur when it is transformed into the keto form?(c) What are the electron-pair geometry and molecular geometry around each C atomin the keto and enol forms? What changes in geometry occur when the keto formchanges to the enol form?(d) Draw three possible resonance structures for the acac–ion.(e) Is cis-trans isomerism possible in either the enol or the keto form of acetylacetone?(f) Is the enol form of acetylacetone polar?…Having one mole of carbon suboxide (C2O3) means that you have
- Arrange the following compounds in the increasing order of reactivity towards Conc.HNO3 & Conc.H₂SO4 1. Benzene 2. Chlorobenzene 3. phenol 4. Toluene 5. Nitrobenzene A 1.2.3.4.5 (B) 5.1,243 5.1.4.2.3 D 5.2.1.4.3Compound A, C5H10O, is one of the basic building blocks of nature. All steroids ans many other naturally occurring compounds are built from compound A. Spectroscopic analysis of A yields the following information. a) how many double bonds and/or rings does A have? b) From the IR spectrum, what is the identity of the oxygen-conataining functional group? c) what kinds of protons are responsible for the NMR absorptions listed d) propose a structure for ACompound B has molecular formula C9H10. The IR spectrum is shown below. The 1H-NMR spectrum shows a multiplet at 7.2 ppm integrating to 4H, a triplet at 2.9 ppm integrating to 4H, and a triplet at 2.1 ppm integrating to 2 H. Suggest a structure for B and explain your reasoning
- For following substituted benzenes: [1] C6H5Br; [2] C6H5CN; [3] C6H5OCOCH3: On balance, does the substituent make a benzene ring more or lesselectron rich than benzene itself?Rank the following substances in order of their expected SN1 reactivity. A) CH3CH2Br B)CH2=CHCH3Br C) CH2=CHBr D) CH3CHCH3Brwhat is the bond line formula of C6H12O2 with respect to given IR spectra and NMR
- Arrange the following compounds in the increasing order of reactivity towards Conc.HNO3 & Conc.H2SO4 1. Benzene 2. Chlorobenzene 3. phenol 4. Toluene 5. Nitrobenzene 5,2,1,4,3 5,1,2,4,3 5,1,4,2,3 1,2,3,4,5A difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired configuration. a. Label this stereogenic center as R or S. b. A well known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship. c. Suggest a reagent to convert A to the single stereoisomer X.Draw the structure of the compound you would expect from SN2 reaction of the molecule below with NaCN.

