Provide the IUPAC name of the product, if any forms from the reaction: in name ex 1,2-dimethylhexane; if no product forms write: N/R) CH3-CH2-CH2-C-H + H₂ Pt
Q: Propose a chemical structure for the name below. Make sure to clearly define the stereochemistry…
A:
Q: Select the correct final major product. Et N NaBH3CN HOAC Et Et H Et. Et NH NH ΝΗ CN COAC
A: Imines (-C=N-) are formed by the condensation of an aldehyde or a ketone with a primary amine. These…
Q: Calculate the following for a 0.225 M solution of a weak acid with Ka=2.1×10^−8. [H3O+] = pH =…
A: Ka of a weak acid(HA) = 2.110-8Molarity of HA = 0.225 MKw = 1.010-14
Q: What Alkyl group does the photo's molecule come from? (K1) CH3 -CH CH3 ○ a methyl ○ b ethyl Ос…
A: The objective of the question is to identify the alkyl group that the given molecule, CH3 -CH CH3,…
Q: 4. Fill in the table 1 2 3 4 5 6 7 8 CH3-CH2-C-CH2-CH2-CH2-CH2-CH3 Proton Chemical Shift (8) in ppm…
A: NMR spectroscopy is a spectroscopic method for the structural elucidation of a molecule. Generally,…
Q: At equilibrium, the concentrations in this system were found to be [N_{2}] = [O_{2}] = 0.300M and…
A: The final concentration of NO be after equilibrium is re-established is 0.5199 MExplanation:
Q: Propose a chemical structure for the name below. Make sure to clearly define the stereochemistry…
A: Given,The name of the molecule is:2-chloro-3-ethyl-2,4-difluoro-4-methylnonane
Q: In the boxes provided below, draw a mechanism to show the formation of the cyanohydrin. Include all…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: OH H2SO4, Δ (d) C-H + OH
A: Acetal-: The compound in which the carbon atom bonded two alkoxy group(OR) i.e a geminal dialkoxy…
Q: 4.36 Give the IUPAC name for each compound.
A: Basic rules for IUPAC nomenclature:1) Select longest chain or block of carbon atoms as parent. 2)…
Q: sample question 2. During further studies on transferrin, the protein was treated with…
A: The above question is about biochemistry and analytical chemistry, specifically the characterisation…
Q: Dinitrogen pentoxide, N_{2}*O_{5} decomposes by first-order kinetics with a rate constant of 3.7 *…
A: The objective of this question is to calculate the time it takes for the concentration of N2O5 to…
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A: The objective of the question is to predict the product formed in the following reaction given.
Q: Lead azide, Pb3(N3)3 has been used as a detonator in automobile airbags. A saturated solution of…
A: Since you have posted multiple questions, we will provide the solution only for the first question…
Q: A 2.7 mg sample of 211 Au decays initially at a rate of 1.08 x 10^18 disintegrations/hr. What is the…
A: The radioactive process generally follows first-order kinetics. The equation for the initial rate of…
Q: Consider the equilibrium system described by the chemical reaction below. For this reaction, Kc =…
A: The objective of the question is to determine the equilibrium concentration of HF in a chemical…
Q: How to calculate the ph? What would be the result if the acid was a di or tripotic? How do the…
A: All of your concerns are discussed; please go through each step.Explanation:Step 1: Let's learn…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. 03 2. Zn, HOAc a
A: Ozonolysis is a chemical reaction in which ozone (O3) is used to cleave carbon-carbon double bonds…
Q: For the compound below please choose the correct set of chair and flipped chair conformations: A 0 D…
A: The cyclohexyl ring goes into rapid inversion which is also called topomerism, ring reversal or…
Q: What is the configuration of the chiral center in the following molecule? OR OS Not a chiral center
A: CIP rule i.e. Chain Ingold Prelog Rule :-The atom which is directly attached to asymmetric centre…
Q: 3 2 CH3-CH2-CH2-OH Proton Chemical Shift (5) in ppm Carbon Chemical Shift (5) in ppm H1 C1 C2 H2 C3…
A: The objective of this question is to identify the proton chemical shifts based on the given molecule…
Q: In making buffer 4, dissolve 5.04g disodium hydrogen phosphate (base) and 3.01g of potassium…
A: A buffer of disodium hydrogen phosphate (base) 3.01g of potassium dihydrogen phosphate (acid)V =…
Q: 11. eauxture. Important: remember to riase and wipe off the mixtures so as not to contaminate them.…
A: The objective of the question is to draw the structures of methyl stearate and propylene glycol and…
Q: Select the correct IUPAC name for the following organic substrate, including approp draw the major…
A: The objective of the question is to-Identify the name of the substrate.Predict the major organic…
Q: Which Newman projection represents correct conformation for the following compound: A HO C H3C. CH3…
A: The two-dimensional representation of a 3D molecule along the C-C bond is known as the Newman…
Q: & 1. NaOH, H2O, heat 2. HCI, H2O (workup) OH OH
A: In nucleophilic addition reactions, a nucleophile attacks an electrophilic center, leading to the…
Q: :0: H. HH © a
A: The reaction between diazomethane(CH2N2) and a carboxylic acid typically results in the formation of…
Q: Identify the most plausible mechanism for the ACID-BASE reaction below: H HH "x H ? A: B: C: D: OA B…
A: An acid-base reaction is also known as a neutralization reaction, In this type of chemical reaction…
Q: Draw the complete mechanism for the acetal formation.
A: see the image belowExplanation:
Q: Use IUPAC nomenclature to name and draw structures for saturated compounds. b) Name the structure:…
A: The IUPAC rules for naming alkanes are given as follows:The longest hydrocarbon chain should be…
Q: 5. Lysozyme is an enzyme that cleaves cell walls. A 0.100-L sample of a solution of lysozyme that…
A: Given,mass of lysozyme = 0.0750 gosmatic pressure = 1.32 x 10-3 atmvolume of the solution = 0.100…
Q: Question 28 For the compound below please identify the position of equilibrium (which chair…
A: At equilibrium, when forward arrow more lengthen than backward arrow then product remain in major…
Q: A B Choose the best description of the kinds of changes that explain the differences between images…
A: Answer:When a system contains only one type of phase that such system is called as homogeneous…
Q: Question 17 Identify the type of hybridization (sp, sp², sp³) for all C (CARBON) atoms of the…
A: Answer:If a carbon atom is forming 4 sigma bonds then it is sp3 hybridized.If a carbon atom is…
Q: igure 1: General reaction scheme. Salicylic acid reacting with acetic anhydride to form…
A: Salicylic acid has -OH and -COOH groups. -OH of one molecule reacts with -COOH of other to produce a…
Q: Please don't provide handwritten solution ....
A: The pressure of CO at equilibrium is 1.31 atm Explanation:You are given the balanced reaction:CO2…
Q: A solution is prepared by dissolving 15.0 g of pure HC2H3O2 and 25.0 g of NaC2H3O2 in 775 mL of…
A: Given,mass of HC2H3O2 = 15.0 gMolar mass of HC2H3O2 = 60.06 g/molmass of NaC2H3O2 = 25.0 gMolar mass…
Q: Question 24 For the compound below please identify the position of equilibrium (which chair…
A: At equilibrium, when forward arrow more lengthen than backward arrow then product remain in major…
Q: There are 48 pairs of students in the following table. Each pair has quantitatively determined the…
A: We have data for caffeine present in a drink, we are to tabulate the data and calculate the…
Q: Please do question 27) and explain/draw out the mechanism with the given reagents
A: The correct answer of the Question no. 27 is :- Option : [ a ] [ (i) H2CrO4 (ii) CH3CH2CH2OH , H+…
Q: The Knoevenagel condensation reaction is a variant of the aldol reaction. A major drawback of the…
A: The Knoevenagel condensation reaction is a version of the aldol reaction in which an aldehyde or…
Q: Identify how many C (CARBON) atoms have sp hybridization for the compound below: :0: N: A B C 0…
A: Hybridization is defined as the intermixing of atomic orbitals which results in the formation of new…
Q: Propose a mechanism that explains the formation of the given byproduct from the starting materials.…
A: Zeolites are crystalline aluminosilicate materials. They are used as catalysts and adsorbents. Their…
Q: Select the correct final major product. A 1) NaBH4/ROH 2) H3O+/H₂O ? -OCH3 H H OH B C Select an…
A: NaBH4 reduces aldehyde and ketone to alcohol.
Q: Question 16 Identify how many N (NITROGEN) atoms have sp hybridization for the compound below: A B 1…
A:
Q: What diene was used to make this compound via a Diels-Alder reaction? O CO₂Me CO₂Me O MeO2C CO₂Me O…
A: Diels-Alder reaction is cycloaddition reaction of the [4π+2π] cyclization .One of the reactant is…
Q: For each compound in the table below, decide whether there would be any hydrogen-bonding force…
A: Answer:When hydrogen atom is directly bonded to oxygen, fluorine or nitrogen atom in a molecule,…
Q: Adjust the structures below to show the resulting intermediates.
A: The resultant intermediate during the reaction of ketone with water is gem diols as two OH group…
Q: 0 H 1.LIAIH 2. H₂O 3. PB. Pyr ? Major Organic Product 0 0 HO
A:
Q: Label each of the following as being a strong base, a weak base, or a species with negligible…
A: Answer:Any specie that loses H+ ion in its solution is called Bronsted-Lowry acid and the specie…
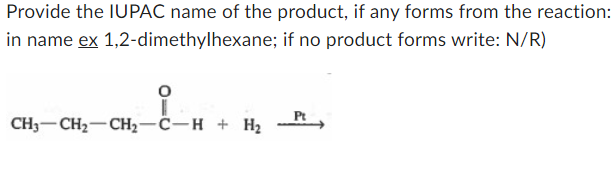
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution
- Complete the following reaction by writing the name of the product. ch3coh + H2 ?When compound CH3-C(CH3)=CH-CH2-CH(CH3)2 is the SUBSTRATE (reactant) in a HALOGENATION reaction with Br2 (and CH2Cl2 as the solvent), a correct condensed structural formula for the MAJOR SUBSTRATE PRODUCT of this reaction is:Draw the products of exo, cis -8 - bromobicyclo (5.1.0) octane, water and heat?
- What is the correct IUPAC name for 2,3,4,5-tetraethylhexane (For Alkene and Alkyne Naming, follow this format "Branch-Carbon Prefix-Functional Group Suffix") e.g. 3-methylhex-2-eneDraw the structures of the following alkenes: a) (Z)-3-methyl-2-hexene, b) (E)-1-chloro-3-ethyl-4-methyl-3-heptene, c) (Z)-5-chloro-3-ethyl-4-hexen-2-olWrite the IUPAC name of the given compounds. For alkenes, write if it is a cis or trans or E or Z if appropriate.
- 1. (a) Draw structures of the seven isomeric alkynes of formula C6H10 - (b) Give the IUPAC and derived name of each. (c) Indicate which ones will react with Ag' or Cu(NH3)2.Please be clear in your writing The following names may have some errors. Correct the name and render the structures corresponding to the following names. g) 1,3-pentadiino h) cyclohexylacetyleneWithout repetitions, how many possible monochloroalkanes are there when 1,1-dimethylcycloheptane reacts with chlorine in the presence of light?
- When cyclopropane is treated with HI, 1-idopropane is formed. A similar type of reaction does not occur with cyclopentene or Cyclohexane. Suggest an explanation for cyclopropane’s reactivity.1. What is the correct IUPAC name for 5-butyl-6-ethyl-4-methylhex-1-ene (For Alkene and Alkyne Naming, follow this format “Branch-Carbon Prefix-Functional Group Suffix”) e.g. 3-methylhex-2-ene 2. What is the correct IUPAC name for 3,4-dimethylhex-5-ene-1-ynepropene + H—OH ------> ?

