Provide the names for the following (3 points each): a) Blank 1 b) Blank 2 c) Blank 3 Last saved 8:04:23 PM ming1 p Questions Filter (8) receint-6935- 20227 CH3 H ||| HIC C C-H 111 CIH H НО. H 8th burk O ACEC C20 Week 12 nelf
Provide the names for the following (3 points each): a) Blank 1 b) Blank 2 c) Blank 3 Last saved 8:04:23 PM ming1 p Questions Filter (8) receint-6935- 20227 CH3 H ||| HIC C C-H 111 CIH H НО. H 8th burk O ACEC C20 Week 12 nelf
Chapter14: Chromatography
Section: Chapter Questions
Problem 6P
Related questions
Question
100%
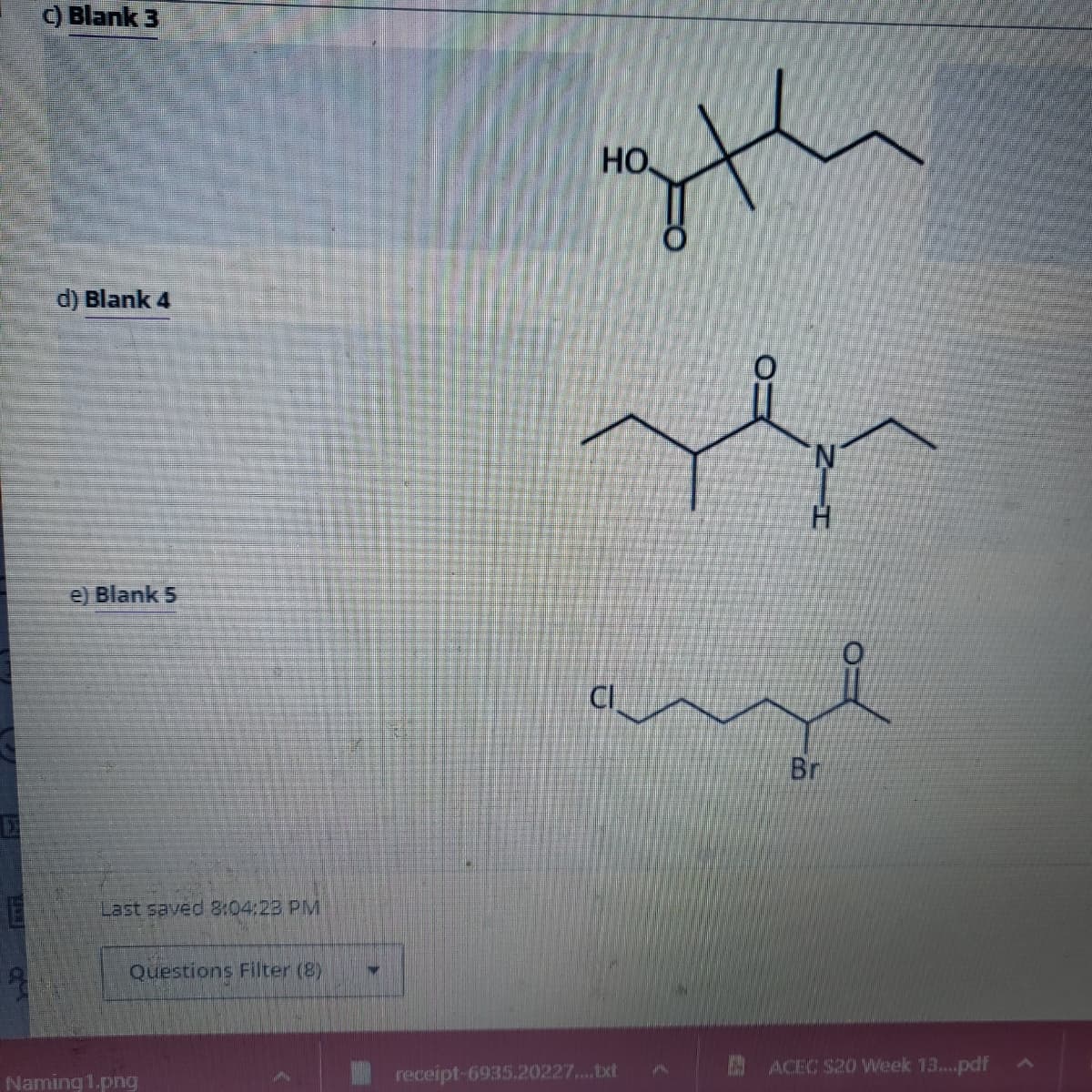
Transcribed Image Text:c) Blank 3
d) Blank 4
e) Blank 5
Last saved 8:04:23 PM
Questions Filter (8)
Naming1.png
НО.
CI
th
receipt-6935.20227....txt
H
Br
ACEC $20 Week 13....pdf
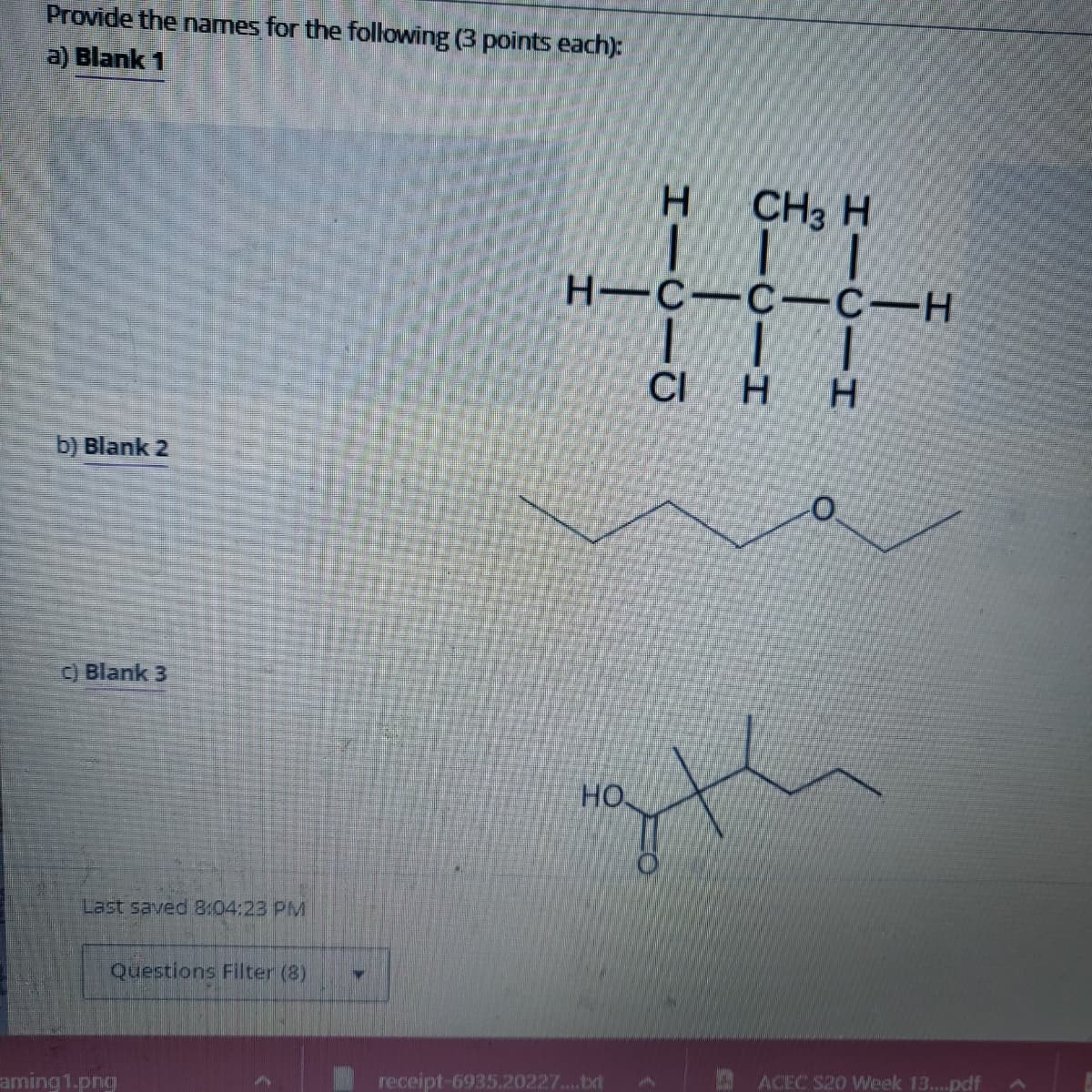
Transcribed Image Text:Provide the names for the following (3 points each):
a) Blank 1
b) Blank 2
c) Blank 3
Last saved 8:04:23 PM
Questions Filter (8)
aming1.png
н-с-с-с-н
| | |
CI H
НО
CH3 H
|||
дн
receipt-6935.20227....book
Н
w
0.
ACEC $20 Week 13....pdf
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



