PV 1/V (1) 1/V→ (iii) (iv) Boyle's law may be best expressed by O the top left curve any of the curves shown here O either of the curves in the bottom row O the top right curve
PV 1/V (1) 1/V→ (iii) (iv) Boyle's law may be best expressed by O the top left curve any of the curves shown here O either of the curves in the bottom row O the top right curve
Modern Physics
3rd Edition
ISBN:9781111794378
Author:Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Chapter10: Statistical Physics
Section: Chapter Questions
Problem 5P
Related questions
Question
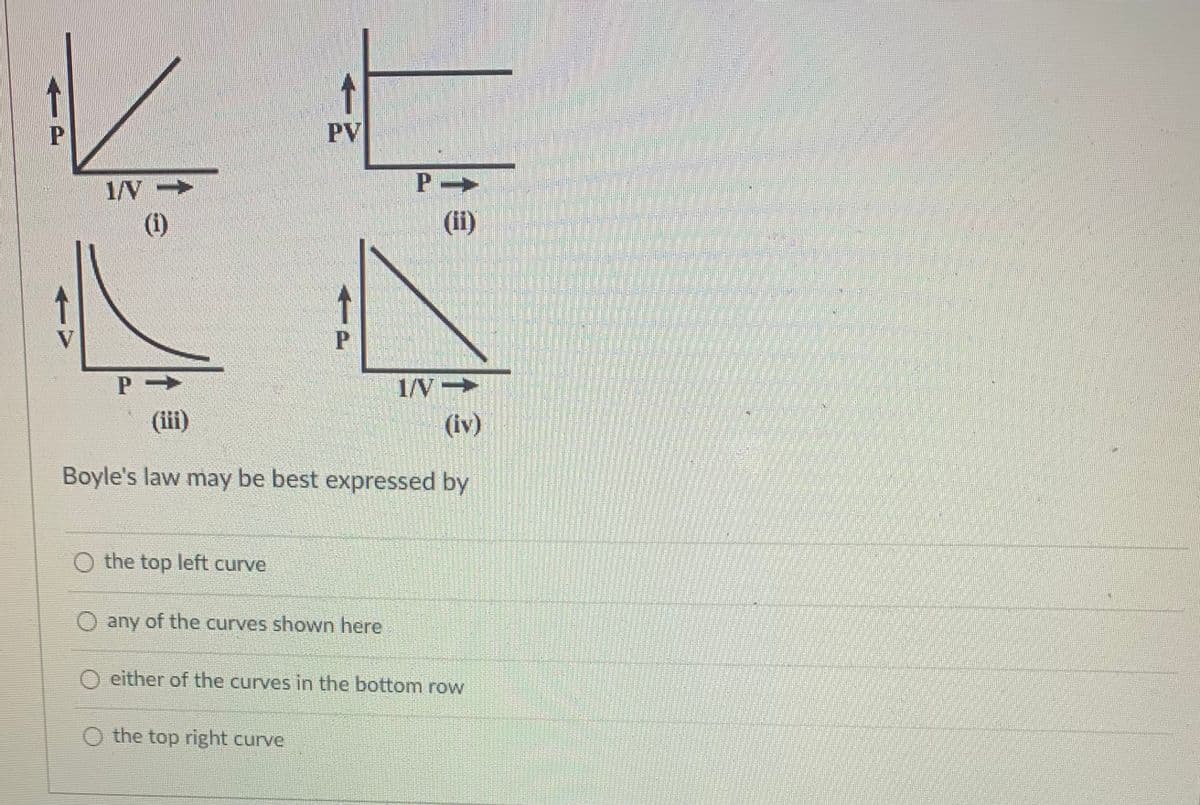
Transcribed Image Text:PV
1/V-
(1)
(ii)
1/V
(iv)
(ii)
Boyle's law may be best expressed by
O the top left curve
any of the curves shown here
either of the curves in the bottom row
O the top right curve
Expert Solution
Step 1
Introduction:
Boyle's law states that the pressure is inversely proportional to the volume of the gas at a constant temperature, and the expression of the ideal gas equation for Boyle's law is given below.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Modern Physics
Physics
ISBN:
9781111794378
Author:
Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:
Cengage Learning


Modern Physics
Physics
ISBN:
9781111794378
Author:
Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:
Cengage Learning
