Q: (2) jubai what is the Normal concentration of a solution that is 4 percent KHSO4 by weight and has…
A: Normality: The number of grams equivalent of the solute dissolved in 1 L of the solution is known as…
Q: A 250 mL of 1% m/v Na2CO3 (MW = 105.99 g/mol) w
A: Given that 250 mL of 1% m/v Na2CO3 (MW = 105.99 g/mol) was found to have a density of 1.05 g/mL…
Q: الاسبوع القادم Prepare 0.8 N of H2SO4 Specific gravity=(1.84 gm/cm3), assay(40%) in 100 ml D.W. if…
A: Normality of solution = 0.8 N
Q: Percent by Mass mass of solute_ × 100 mass of solution Percent by mass equals the mass of the solute…
A: Given : Mass of solute i.e DTN = 25 mg And mass of solution = 1000.0 mg
Q: 3. Ms Lauren R. Davis prepared three aqueous solutions of sucrose (C12H22O11) in proportions as…
A: Molarity of a solution is the number of moles of solute dissolved in one litre of the solution…
Q: Commercial perchloric acid (100.46 g/mol) 71% (w/w) with specific gravity of 1.67 was diluted by…
A:
Q: A solution of NaNO3 contains 120 grams of solute dissolved in 100 grams of water. At 50°C this…
A: From the graph , Solubility of NaNO3 at 50°C = 113 g/100 g water (less than 120 g) If the amount…
Q: A certain liquld X has a normal bolling polnt of 140.40 °C and a bolling polnt elevation constant…
A: Given : Normal boiling point of liquid X = 140.40 oC Mass of solvent i.e liquid X = 300 g = 0.300 Kg…
Q: A student used 10 mL water instead of 30 mL for extraction of salt from mixture. How may this may…
A: Solubility of substance is defined as mass of solute dissolve per liter of the solution.
Q: An automobile antifreeze mixture is made by mixing equal volumes of ethylene glycol (SG= 1.114; MW =…
A: Since you have posted multiple sub-parts, the answer for first three sub-parts are given below.…
Q: Asolution of cthanol Ld=0.795glmL) and water (H20) is Prefacd IS Placing30.0 ML of ethanol in a…
A: We are given that the density of ethanol as 0.795 g/mL and 30 mL of ethanol is taken to make a…
Q: From 0.8 av lb of 20% w/w KOH solution 142 86.2 121 72.6 g of 12 percent w/w KOH may be produced.
A:
Q: Extraction is a process of purifying immiscible liquids. O TRUE FALSE
A: The purification of any two immiscible liquids can be carried out by a number of methods.
Q: scribe the preparation of 150 mL of 6.00 M H3PO4 f at is 86% H3PO4 (w/w) and has the specific…
A: Given data set: Volume of H3PO4 = 150 ml Molarity of H3PO4 = 6.00 M Specific gravity= 1.71 W/w% =…
Q: 170 milliliters are required to prepare 900 mL of 2.00 M HCI (36.5g/mol) from the commercial reagent…
A: Concentration of commercial HCl = 37.2 % (w/w)
Q: Prepare 0.1N H2SO4 solution of 250ml volume. (Density of H2SO4 is 1.84 and purity is 98 %)
A: We are givenH2SO4 purity=98% i.e in 100 gm of solution H2SO4 present =98 gm
Q: d 630 mL of 2.90% (w/v) aqueous CuSO4 from a 0.385 M CuSO4 solution. Take mL of the 0.385 M CuSO4…
A:
Q: mL of H2SO4 solution 75 (specificgravity =1.18) containing 49% H2SO4 (w/w) is diluted to 590 mL.…
A:
Q: A chemistry student wishes to extract 2.34 g of compound X from 15 mL of an aqueous mixture. She has…
A: Given that : The mass of compound X to be extracted = 2.34 g The volume of the aqueous mixture = 15…
Q: Find the mass of sodium formate that must be dissolved in 430.0 cm' of a 2.0 M solution of fommic…
A: Given, Molarity of HCOOH = [HCOOH] = 2.0 M Volume of HCOOH = 430.0 cm3 pH of the buffer solution…
Q: Prepare 1500mL of 0.5N H2SO4 solution with specific gravity of 1.84g/mL and assay of 97%. Write…
A:
Q: Mass Na CO3 Vi HC1,mL 0.00 0.10 Vf HC1,mL 36.75 Trial VHC) used, ml MHCI 1 0.2135 0.2025 0.2112…
A: Since you have asked a question with multiple sub-parts, as per our company guidelines we are…
Q: Q2. 1000 lb of 10% NaOH solution at100°F is to be fortified to 30% NAOH by adding 73% NaOH at 200°F.…
A: A solution is a homogeneous mixture of two or more components in a solvent. Sodium hydroxide…
Q: how many mL of a 0.555 M KNO3 (Mw=101.1g/mol) solution would contain 30.5 g of KNO3?
A: Given Molarity of KNO3 = 0.555 M Molecular weight of KNO3 = 101.1 gm/mole Mass of…
Q: stoic A Exactly 600.0 mL of a solution that contained 447.7 ppm of Ba (NO3), were mixed with 250.0…
A: The mass and number of moles of a compound can be calculated from a balanced chemical reaction. A…
Q: Supplement tablets containing 50 mg of Mg-" each are sold to bolster bone health and muscle…
A: Given: Amount of Mg2+ in each supplement tablet = 50 mg Hardness value of water in terms of CaCO3 =…
Q: What is the Normality of a sulfuric acid solution containing 75% w/v of H2SO4 (98.0) and having a…
A: Normality: In 1000 mL or 1L of the solution, the gram equivalent of the solute present is called the…
Q: To prepare 100ml of 0.2M from the stock solution (10M) we need to dilute 4ml of the stock solution…
A: Provided final volume(V2) of solution=100 ml Provided final concentration(M2) =0.2M Initial…
Q: 6.30 L of 0.262 M NaOH from the concentrated commercial reagent [50% NaOH (w/w), sp gr 1.525]. Take…
A:
Q: What is the level of the liquid in the gatuted oede Volume mL. 70 60
A: Everything that has and occupies space is called matter. Solids, liquids, gases are the three states…
Q: An unhnown brown liquid is unitorm thronghont ond con be further Seperated by distillation The…
A: Unknown can be determined by given properties.
Q: Preparation of 100 ml 1.0 molar (1.0M) H2SO4 solution (specific gravity, 1.8 g/ml and purity, 98%).…
A: Molarity is defined as the number of moles of solute dissolved per liter of the solution. Molarity,…
Q: 780 mL of 1.10% (w/v) aqueous BaCl2 from a 0.400 M BaCl2 solution. mL with water. Take mL of the…
A: Volume of 0.40 M BaCl2 solution to make 1.10% (w/v) 780 mL of BaCl2 solution = ? Volume of…
Q: To 500 g of K2Cr207 solution with a mass fraction K2Cr207 5% added 300g of water. Determine the mass…
A: Given :- Mass fraction of K2Cr2O7 = 5% Mass of solution = 500 g Mass of water added to the…
Q: I wish to prepare 500.0 mL of a 800.0 ppm w/v fluoride (MW = 18.9984 g/mol) solution. a.)How many…
A: a) First calculate the mass fluoride in 500mL solution then it is used to determine the moles…
Q: 50g of nitrogen (N2) has a volume of ___ liters at STP. Show the complete solution
A:
Q: mL of H2SO4 solution 75 (specificgravity =1.18) containing 49% H2SO4 (w/w) is diluted to 590 mL.…
A: Given information-: Volume of solution = 75 ml ( V1) Specific gravity = 1.18 H2SO4 (w/w) = 49 %…
Q: True solution is transparent. * O True O False
A: True solution is when solute is dissolved uniformly through out the solution. And is homogeneous…
Q: If I take 13.23 grams of tri-sodium phosphate and dissolve it in 500mL, then subsequently tåke 50 ml…
A: One mole of tri-sodium phosphate contains three moles of sodium ions. Steps to be followed (i)…
Q: Calculate the mass percent of a NaCl solution in which 185 g of NaCl is dissolved in 1.89 L of water…
A: By using this formula we can calculate the mass Percent of NaCl . Mass of NaCl = 185 g Volume of…
Q: Prepared 1 M of NaCl solution in volume ?100 ml, Mwt is 58.5 0.585 gm. O 5.85 gm. O 58.5 gm. O
A: Given Data : 1. Concentration of NaCL = 1 M 2. Volume = 100 mL = 100 ÷1000 =…
Q: Answers should be in 4th decimal places An automobile antifreeze mixture is made by mixing equal…
A: Given data: Density of mixture = 1.070 g/mL Density of ethylene glycol = 1.114 g/mL Molecular weight…
Q: What will be the new boilng point of 491ml benzene (Kb = 2.53 oC•Kg/mol; Bp = 80.1oC) when mixed…
A:
Q: 12 g of unknown organic sample was dissolve in 706 mL of Dicloromethane (DCM). The boiling point of…
A: Given:: mass of unknown= 12 g molecular weight of unknown = x = ? ∆Tb= 2.72℃ Kb of DCM = 2.42 ℃ m-1…
Q: DATA Tute used Bocum Nitrate (BalNoz)z Imel BaCNSs)z 261.337glnol 2.0 261.337g/mol 20g 0077moles ams…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is the freezing point of radiator fluid that is 60% antifreeze by mass?Kf for water is 1.96…
A: 60% antifreeze by mass means 60 g antifreeze is present in 100 g of water.Ethylene glycol is used as…
Q: If 10.0 mL of H2SO4 (sp. Gr. 1.50, containing 48.7% of combined SO3 by weight) is diluted to 400 mL,…
A: A numerical problem based on concentration terms that is to be accomplished.
Q: 1.) How many grams of a 75% v/v hydroalcoholic solution should be used in preparing 16 fl.oz. of a…
A: Since you have posted multiple questions, we are entitled to answer the first only. Given:…
Q: 250 mL of 1% m/v Na2CO3 (MW = 105.99 g/mol) was found to have a density of 1.05 g/mL at 25oC. Find…
A:
Q: How would you prepare 75 mL of an H2SO4 solution at 20% in weight (density=1.1394 g/mL,…
A: X % of an acid solution means its Xg in 100g of solution. Volume of solution can be calculated from…
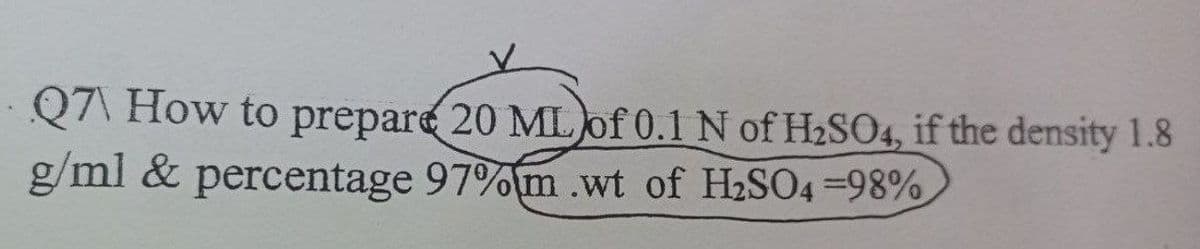
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- You were tasked to prepare 590 ml of 3.00 M HNO3. If you were provided a stock solution of 70.5% (w/w) HNO3 (MW = 63.0 g) with a specific gravity of 1.42, what volume of this solution (in ml) will you be using?A core sample is saturated with an oil (?o = 35oAPI), gas and water. The initial weight of the sample is 224.14 g. After the gas is displaced by water (?w = 1 g/cm3 ), the weight is increased to 225.90 g. The sample is the placed in a Soxhlet distillation apparatus, and 4.4 cm3 water is extracted. After drying the core sample, the weight is now 209.75 g. The sample bulk volume, 95 cm3 is measured in a mercury pycnometer. Find the porosity, water saturation, oil saturation, gas saturation and lithology of the core sample.The ethyl alcohol content of beers produced is reported as ABV or alcohol by volume (vol/vol) If the density of ethyl alcohol at room temperature is 0.7893 grams/mL, what is the percent of ethyl alcohol in 4.50% beer expressed as percent (wt/vol)
- Given the following data forMass of test tube, beaker and cyclohexane = 100.17 gMass of test tube and beaker = 84.07 gFreezing point of cyclohexane = 6.59 oCMass of weighing paper + naphthalene =1.080 gMass of weighing paper = 0.928 gFreezing point solution = 5.11oCKf = 20.8oC/mDetermine the followinga. mass of cyclohexane in g (2 decimal places); _____b. mass of naphthalene in g (4 decimal places); _____c. freezing point depression (2 decimal places); _____d. molality of solution (3 significant figures); _____e. moles of naphthalene (3 significant figures); _____f. molar mass of naphthalene, experimentally (3 significant figures); _____g. % error if theoretical molar mass of naphthalene is 128.17 g/ mole, USE ABSOLUTE VALUE (3 significant figure); ____Stock iron(II) solution (200Ug mL-1 Fe) ferrous ammonium sulfate hexahydrate mass= 0.1437g, transfer it to a 100 ml beaker. add 15 ml approx of water and 15m1 'approx of dilute sulphuric acid (2M H2SO.). then transfer FeII to 100 ml flask makeup to the mark with water. calculate the moles of ferrous ammonium sulfate hexahydrate solution in unit ug/mL.When concentrated nitric is sold, the label contains no mention of the molarity of the acid. Instead, the label normally lists the concentration of nitric acid as a wt/wt percent and gives the specific gravity of the solution. If the solution is 68.0 (wt/wt %) and nitric acid and has a specific gravity of 1.41 kg/L, calculate the molarity of 68.0 % concentrated nitric acid. FW HNO3=63.01g/mol
- 50g of nitrogen (N2) has a volume of ___ liters at STP. Show the complete solution a. V=49.0L b. V=40.0L c. V=44.9L d. V=47.70LSum of coefficients C7H8 + O2 --> CO2 + H2O after balancingA mixture of ethanol (ethyl alcohol) and water contains 40.0% water by mass.(a) Assuming volume additivity of the components, estimate the specific gravity of themixture at 20°C. What volume (in liters) of this mixture is required to provide 150 mol ofethanol?(b) Repeat Part (a) with the additional information that the specific gravity of the mixture at20°C is 0.89045 (making it unnecessary to assume volume additivity). What percentage errorresults from the volume-additivity assumption?



