Chapter4: Organic Compounds: Cycloalkanes And Their Stereochemistry
Section4.SE: Something Extra
Problem 23VC: Name the following compound, identify each substituent as axial or equatorial, and tell whether the...
Related questions
Question
Which is/are the second-most-stable conformer(s) on your list? These are the options to the question I attached below.
|
A. both 3 & 5 |
||
|
B. both 1 & 4 |
||
|
C. 2 only |
||
|
D. 6 only |
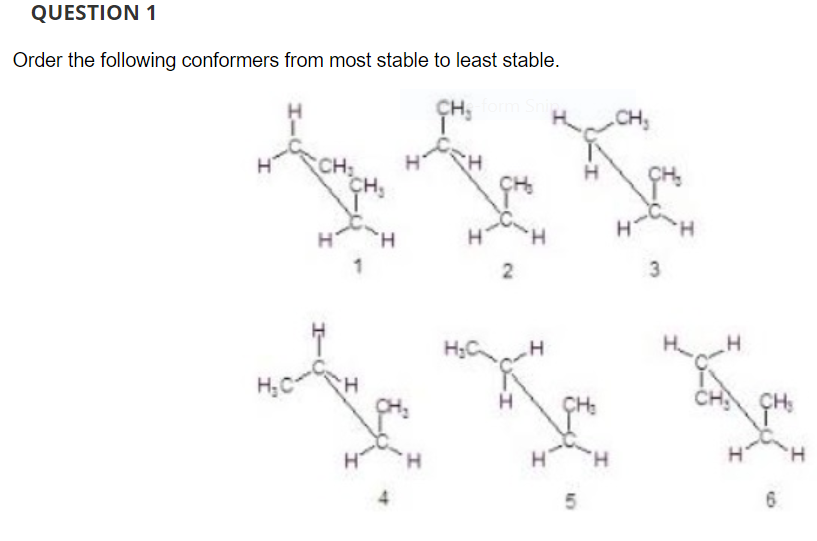
Transcribed Image Text:QUESTION 1
Order the following conformers from most stable to least stable.
CH
Η "Η
2
CH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

